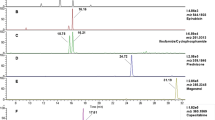
Abstract
The results of an inter-laboratory comparison exercise to determine cytostatic anticancer drug residues in surface water, hospital wastewater and wastewater treatment plant effluent are reported. To obtain a critical number of participants, an invitation was sent out to potential laboratories identified to have the necessary knowledge and instrumentation. Nine laboratories worldwide confirmed their participation in the exercise. The compounds selected (based on the extent of use and laboratories capabilities) included cyclophosphamide, ifosfamide, 5-fluorouracil, gemcitabine, etoposide, methotrexate and cisplatinum. Samples of spiked waste (hospital and wastewater treatment plant effluent) and surface water, and additional non-spiked hospital wastewater, were prepared by the organising laboratory (Jožef Stefan Institute) and sent out to each participant partner for analysis. All analytical methods included solid phase extraction (SPE) and the use of surrogate/internal standards for quantification. Chemical analysis was performed using either liquid or gas chromatography mass (MS) or tandem mass (MS/MS) spectrometry. Cisplatinum was determined using inductively coupled plasma mass spectrometry (ICP-MS). A required minimum contribution of five laboratories meant that only cyclophosphamide, ifosfamide, methotrexate and etoposide could be included in the statistical evaluation. z-score and Q test revealed 3 and 4 outliers using classical and robust approach, respectively. The smallest absolute differences between the spiked values and the measured values were observed in the surface water matrix. The highest within-laboratory repeatability was observed for methotrexate in all three matrices (CV ≤ 12 %). Overall, inter-laboratory reproducibility was poor for all compounds and matrices (CV 27–143 %) with the only exception being methotrexate measured in the spiked hospital wastewater (CV = 8 %). Random and total errors were identified by means of Youden plots.







Similar content being viewed by others
References
Česen M, Kosjek T, Laimou-Geraniou M, Kompare B, Širok B, Lambropolou D, Heath E (2015) Occurrence of cyclophosphamide and ifosfamide in aqueous environment and their removal by biological and abiotic wastewater treatment processes. Sci Total Environ 527–528:465–473
Crauste-Manciet S, Sessink Pj Fau - Ferrari S, Ferrari S Fau - Jomier JY, Jomier Jy Fau - Brossard D, Brossard D (2005) Environmental contamination with cytotoxic drugs in healthcare using positive air pressure isolators
Dehouck P, Vander Heyden Y, Smeyers-Verbeke J, Massart DL, Marini RD, Chiap P et al (2003) Interlaboratory study of a liquid chromatography method for erythromycin: determination of uncertainty. J Chromatogr A 1010:63–74
European Food Safety Authority. Management of left-censored data in dietary exposure assessment of chemical substances. EFSA Journal 2010; 8: Article 1557
Farré M, Petrovic M, Gros M, Kosjek T, Martinez E, Heath E et al (2008) First interlaboratory exercise on non-steroidal anti-inflammatory drugs analysis in environmental samples. Talanta 76:580–590
Ferrando-Climent L, Rodriguez-Mozaz S, Barceló D (2013) Development of a UPLC-MS/MS method for the determination of ten anticancer drugs in hospital and urban wastewaters, and its application for the screening of human metabolites assisted by information-dependent acquisition tool (IDA) in sewage samples. Anal Bioanal Chem 405:5937–5952
Ferrando-Climent L, Rodriguez-Mozaz S, Barcelo D (2014) Incidence of anticancer drugs in an aquatic urban system: from hospital effluents through urban wastewater to natural environment
Heath E, Kosjek T, Andersen HR, Holten Lützhøft HC, Adolfson Erici M, Coquery M et al (2010a) Inter-laboratory exercise on steroid estrogens in aqueous samples. Environ Pollut 158:658–662
Heath E, Kosjek T, Farre M, Quintana JB, de Alencastro LF, Castiglioni S et al (2010b) Second interlaboratory exercise on non-steroidal anti-inflammatory drug analysis in environmental aqueous samples. Talanta 81:1189–1196
Hund E, Massart DL, Smeyers-Verbeke J (2000) Inter-laboratory studies in analytical chemistry. Anal Chim Acta 423:145–165
Jones TL, Betowski LD, Lopez-Avila V (1994) Validation of liquid chromatography—mass spectrometry methods: interlaboratory studies. TrAC Trends Anal Chem 13:333–338
Kosjek T, Heath E (2011) Occurrence, fate and determination of cytostatic pharmaceuticals in the environment. TrAC Trends Anal Chem 30:1065–1087
Kosjek T, Perko S, Žigon D, Heath E (2013) Fluorouracil in the environment: analysis, occurrence, degradation and transformation. J Chromatogr A 1290:62–72
Kosjek T, Negreira N, de Alda ML, Barceló D (2015) Aerobic activated sludge transformation of methotrexate: identification of biotransformation products. Chemosphere 119(Supplement):S42–S50
Llewellyn N, Lloyd P, Jürgens MD, Johnson AC (2011) Determination of cyclophosphamide and ifosfamide in sewage effluent by stable isotope-dilution liquid chromatography–tandem mass spectrometry. J Chromatogr A 1218:8519–8528
Negreira N, López de Alda M, Barceló D (2013a) On-line solid phase extraction–liquid chromatography–tandem mass spectrometry for the determination of 17 cytostatics and metabolites in waste, surface and ground water samples. J Chromatogr A 1280:64–74
Negreira N, Mastroianni N, López de Alda M, Barceló D (2013b) Multianalyte determination of 24 cytostatics and metabolites by liquid chromatography–electrospray–tandem mass spectrometry and study of their stability and optimum storage conditions in aqueous solution. Talanta 116:290–299
Negreira N, de Alda ML, Barceló D (2014a) Cytostatic drugs and metabolites in municipal and hospital wastewaters in Spain: filtration, occurrence, and environmental risk. Sci Total Environ 497–498:68–77
Negreira N, López de Alda M, Barceló D (2014b) Study of the stability of 26 cytostatic drugs and metabolites in wastewater under different conditions. Sci Total Environ 482–483:389–398
Odraska P, Dolezalova L, Kuta J, Oravec M, Piler P, Blaha L (2013) Evaluation of the efficacy of additional measures introduced for the protection of healthcare personnel handling antineoplastic drugs. Ann Occup Hyg 57:240–250
Parrella A, Lavorgna M, Criscuolo E, Russo C, Fiumano V, Isidori M (2014) Acute and chronic toxicity of six anticancer drugs on rotifers and crustaceans
Van den Bossche L, Lodi A, Schaar J, Shaakov S, Zorzan M, Tranquillini ME et al (2010) An interlaboratory study on the suitability of a gradient LC-UV method as a compendial method for the determination of erythromycin and its related substances. J Pharm Biomed Anal 53:109–112
Vander Heyden Y, Smeyers-Verbeke J (2007) Set-up and evaluation of interlaboratory studies. J Chromatogr A 1158:158–167
Vander Heyden Y, Saevels J, Roets E, Hoogmartens J, Decolin D, Quaglia MG et al (1999) Interlaboratory studies on two high-performance liquid chromatographic assays for tylosin (tartrate). J Chromatogr A 830:3–28
Yin J, Shao B, Zhang J, Li K (2010) A preliminary study on the occurrence of cytostatic drugs in hospital effluents in Beijing, China. Bull Environ Contam Toxicol 84:39–45
Youden WJ (1959) Graphical diagnosis of interlaboratory test results. 15, Industrial Quality Control, p 24–28
Web References
Chemoth 2013; http://chemoth.com/economics, (Accessed 1 Aug 2013)
CytoThreat Project 2014; http://www.cytothreat.eu/ (Accessed 10 Aug 2013)
Cytotoxic Safety 2013; http://www.cytotoxicsafety.org/protect-the-environment/ (Accessed 1 Aug 2013)
Cytotoxic Safety 2013; http://cytotoxicsafety.org/wp-content/uploads/2014/03/Cytotoxic-Drug-Contamination-in-Surface-Water-Theresa-OKeefe.pdf (Accessed 5 Aug 2013)
ISO TC 69/SC 6 2011; http://www.iso.org/iso/iso_technical_committee.html%3Fcommid=49808 (Accessed 20 Jun 2013)
ISO 13528, 2009; http://www.iso.org/iso/catalogue_detail.htm?csnumber=35664 (Accessed 2 Aug 2013)
MedCalc Software 2015; http://www.medcalc.org/manual/youdenplot.php (Accessed 10 Jul 2013)
Reuters 2008; http://www.reuters.com/article/2008/02/19/idUS97258+19-Feb-2008+BW20080219, (Accessed 25 Nov 2014)
World Cancer Report 2014; B.W. Stewart, C.P. Wild (Eds.), IARC, WHO (2014). http://edition.cnn.com/2014/02/04/health/who-world-cancer-report/ (Accessed 12 Oct 2024)
Acknowledgement
This work was financially supported by the EU through the EU FP7 projects CytoThreat (Fate and effects of cytostatic pharmaceuticals in the environment and the identification of biomarkers for an improved risk assessment on environmental exposure project; grant agreement No.: 265264) and SOLUTIONS (Solutions for present and future emerging pollutants in land and water resources management; grant agreement No. 603437), by the Slovenian Research Agency (Program groups P1-0143, Project L1-5457, Project J1-6744 and a Young researcher grant to M. Č.), and by the Generalitat de Catalunya (Consolidated Research Groups “2014SGR418-Water and Soil Quality Unit” and “2014SGR291-ICRA”). The work reflects only the authors’ views. The Community is not liable for any use that may be made of the information contained therein. The authors would also like to thank Prof. Walter Giger and Dr. Valeria Dulio for their support with networking and Dr. Francesco Busetti for his willingness to participate in this exercise and Dr. David Heath for providing language help.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Philippe Garrigues
Electronic supplementary material
Below is the link to the electronic supplementary material.
Figure S1
(DOCX 113 kb)
Figure S2
(DOCX 84 kb)
Figure S3
(DOCX 84 kb)
Figure S4
(DOCX 71 kb)
Figure S5
(DOCX 55 kb)
Table S1
(DOCX 21 kb)
Table S2
(DOCX 19 kb)
Table S3
(DOCX 24 kb)
Table S4
(DOCX 22 kb)
Table S5
(DOCX 15 kb)
Table S6
(DOCX 18 kb)
Table S8
(DOCX 23 kb)
Table S9
(DOCX 20 kb)
Table S10
(DOCX 24 kb)
Table S11
(DOCX 22 kb)
Table S12
(DOCX 13 kb)
Rights and permissions
About this article
Cite this article
Heath, E., Česen, M., Negreira, N. et al. First inter-laboratory comparison exercise for the determination of anticancer drugs in aqueous samples. Environ Sci Pollut Res 23, 14692–14704 (2016). https://doi.org/10.1007/s11356-015-4982-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-015-4982-9