Abstract
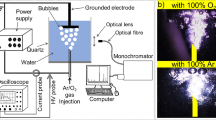
The objective of this work was to study the degradation and mineralization of ethylenethiourea (ETU) in water by ozonation at different pH values and in the presence of hydrogen peroxide. Degradation experiments were performed using an initial ETU concentration of 50 ppm for 180 min with a gas flux of 0.25 dm3 min−1 and an O3 production rate of 12.1 mg min−1. Degradation of by-products was monitored by direct injection electrospray ionization mass spectrometry (ESI-MS), ETU concentration was determined by HPLC-UV, and its mineralization was detected by total organic carbon (TOC) analysis. Optimum degradation of ETU in water was observed at pH = 11, whereas at pH = 3, the degradation of ETU was slowest, indicating that the reaction occurred through different mechanisms. The additional effects of hydroxyl radicals formed at the highest pH can be used to explain the results obtained in this study. Peroxone experiments were carried out in the presence of 400 and 800 mg L−1 H2O2; the degradation of ETU was faster at 400 mg L−1 H2O2. This was attributed to the scavenging effect of the excess H2O2. ETU treatment by ozonation produced several by-products of degradation such as ethylene urea and 2-imidazoline.




Similar content being viewed by others
References
Amorim CC, Bottrel SE, Costa EP, Teixeira AP, Leão MM (2013a) Removal of ethylenthiourea and 1,2,4-triazole pesticide metabolites from water by adsorption in commercial activated carbons. J Environ Sci Health B 48(3):183–190. doi:10.1080/03601234.2013.730287
Amorim CC, Leão MMD, Moreira RFPM, Fabris JD, Henriques AB (2013b) Performance of blast furnace waste for azo dye degradation through photo-Fenton-like processes. Chem Eng J 224:59–66. doi:10.1016/j.cej.2013.01.053
Ballesteros MM, Sánchez PJ, Casas LJ, Oller I, Malato RS (2009) Degradation of a four-pesticide mixture by combined photo-Fenton and biological oxidation. Water Res 43(3):653–660. doi:10.1016/j.watres.2008.11.020
Bottrel SE, Amorim CC, Leao MM, Costa EP, Lacerda IA (2014) Degradation of ethylenethiourea pesticide metabolite from water by photocatalytic processes. J Environ Sci Health B 49(4):263–270. doi:10.1080/03601234.2014.868280
Chelme-Ayala P, El-Din MG, Smith DW (2010) Degradation of bromoxynil and trifluralin in natural water by direct photolysis and UV plus H2O2 advanced oxidation process. Water Res 44(7):2221–2228. doi:10.1016/j.watres.2009.12.045
De Witte B, Van Langenhove H, Demeestere K, Saerens K, De Wispelaere P, Dewulf J (2010) Ciprofloxacin ozonation in hospital wastewater treatment plant effluent: effect of pH and H2O2. Chemosphere 78(9):1142–1147. doi:10.1016/j.chemosphere.2009.12.026
Dong X, Meng J, Yang B, Zhang Y, Gan J, Shu X, Shu J (2011) Experimental studies on ozonation of ethylenethiourea. J Environ Sci 23(1):65–69. doi:10.1016/s1001-0742(10)60374-9
Farré MJ, Franch MI, Malato S, Ayllón JA, Peral J, Doménech X (2005) Degradation of some biorecalcitrant pesticides by homogeneous and heterogeneous photocatalytic ozonation. Chemosphere 58(8):1127–1133
Gago-Ferrero P, Demeestere K, Silvia Díaz-Cruz M, Barceló D (2013) Ozonation and peroxone oxidation of benzophenone-3 in water: effect of operational parameters and identification of intermediate products. Sci Total Environ 443:209–217. doi:10.1016/j.scitotenv.2012.10.006
Hoigné J, Bader H (1983) Rate constants of reactions of ozone with organic and inorganic compounds in water—I: non-dissociating organic compounds. Water Res 17(2):173–183. doi:10.1016/0043-1354(83)90098-2
Hwang E-S, Cash JN, Zabik MJ (2003) Determination of degradation products and pathways of mancozeb and ethylenethiourea (ETU) in solutions due to ozone and chlorine dioxide treatments. J Agric Food Chem 51(5):1341–1346
Marshall WD, Singh J (1977) Oxidative inactivation of ethylenethiourea by hypochlorite in alkaline medium. J Agric Food Chem 25(6):1316–1320. doi:10.1021/jf60214a051
Meijers R, Oderwald-Muller E, Nuhn P, Kruithof J (1995) Degradation of pesticides by ozonation and advanced oxidation. Ozone Sci Eng 17(6):673–686
Nogueira RF, Oliveira MC, Paterlini WC (2005) Simple and fast spectrophotometric determination of H2O2 in photo-Fenton reactions using metavanadate. Talanta 66(1):86–91. doi:10.1016/j.talanta.2004.10.001
Poole AJ, Cord-Ruwisch R (2004) Treatment of strong flow wool scouring effluent by biological emulsion destabilisation. Water Res 38(6):1419–1426. doi:10.1016/j.watres.2003.11.034
Santos MS, Alves A, Madeira LM (2011) Paraquat removal from water by oxidation with Fenton’s reagent. Chem Eng J 175:279–290. doi:10.1016/j.cej.2011.09.106
Soares AS, Leão M, Neto MV, da Costa E, de Oliveira M, Amaral N (2013) Efficiency of conventional drinking water treatment process in the removal of endosulfan, ethylenethiourea, and 1,2,4-triazole. J Water Supply Res Technol 62(6):367–376
Srivastava AK, Ali W, Singh R, Bhui K, Tyagi S, Al-Khedhairy AA, Srivastava PK, Musarrat J, Shukla Y (2012) Mancozeb-induced genotoxicity and apoptosis in cultured human lymphocytes. Life Sci 90(21–22):815–824. doi:10.1016/j.lfs.2011.12.013
USEPA (2005) Reregistration eligibility decision for mancozeb. Prevention, pesticides and toxic substances (7508C). EPA 738-R-04-012
von Gunten U (2003) Ozonation of drinking water: part I. Oxidation kinetics and product formation. Water Res 37(7):1443–1467. doi:10.1016/S0043-1354(02)00457-8
Zapata A, Velegraki T, Sánchez-Pérez J, Mantzavinos D, Maldonado M, Malato S (2009) Solar photo-Fenton treatment of pesticides in water: effect of iron concentration on degradation and assessment of ecotoxicity and biodegradability. Appl Catal B Environ 88(3):448–454. doi:10.1016/j.apcatb.2008.10.024
Acknowledgments
The authors thank the Foundation for Research Support of the State of Minas Gerais (FAPEMIG), the Coordination for the Improvement of Higher Education Personnel (CAPES), and the National Counsel of Technological and Scientific Development (CNPq) for their financial support.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Philippe Garrigues
Rights and permissions
About this article
Cite this article
Bottrel, S., Amorim, C., Ramos, V. et al. Ozonation and peroxone oxidation of ethylenethiourea in water: operational parameter optimization and by-product identification. Environ Sci Pollut Res 22, 903–908 (2015). https://doi.org/10.1007/s11356-014-3616-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-014-3616-y