Abstract
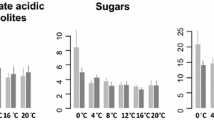
The male part of the spadix of Dracunculus vulgaris exhibits a degree of temperature regulation by inversely controlled heat production over a 20–35 °C range of tissue temperature. To clarify the effects of temperature on cellular metabolism, comparative analysis was performed using 51 metabolites from two distinct tissues (florets and pith) of thermogenic male spadices that had been temperature clamped at either 20 (to produce high respiration) or 35 °C (to produce low respiration). Principal component analysis and hierarchical clustering analysis showed that changes in metabolites in the florets, but not in the pith, were associated with temperature change. The energy charge in the florets treated at 20 °C was significantly higher than that of the florets treated at 35 °C. This indicated the presence of an increased energy-producing pathway that ultimately led to an increased level of thermogenesis at 20 °C. Intriguingly, succinate, a direct substrate for complex II in the mitochondrial respiratory chain, was the metabolite most significantly affected in our analysis, with its concentration in the florets 3.5 times higher at 20 than at 35 °C. However, the mitochondria fed with succinate showed that state 2 and 3 respirations and the capacity of the alternative and cytochrome pathways were all significantly higher at 35 than at 20 °C. Taken together, the results show that the male florets are the primary sites for temperature-induced changes in metabolomic pathways, although succinate-stimulated mitochondrial respiration, per sé, is not the control mechanism for thermoregulation in D. vulgaris.





Similar content being viewed by others
References
Albury, M. S., Elliott, C., & Moore, A. L. (2009). Towards a structural elucidation of the alternative oxidase in plants. Physiologia Plantarum, 137, 316–327.
Atkinson, D. E. (1977). Cellular energy metabolism and its regulation. New York: Academic Press.
Chance, B., & Williams, G. R. (1955). Respiratory enzymes in oxidative phosphorylation. III. The steady state. Journal of Biological Chemistry, 217, 409–428.
Clarke, A., & Pörtner, H. O. (2010). Temperature, metabolic power and the evolution of endothermy. Biological Reviews, 85, 703–727.
Djajanegara, I., Holtzapffel, R., Finnegan, P. M., Hoefnagel, M. H. N., Berthold, D. A., Wiskich, J. T., et al. (1999). A single amino acid change in the plant alternative oxidase alters the specificity of organic acid activation. FEBS Letters, 454, 220–224.
Elthon, T. E., Nickels, R. L., & McIntosh, L. (1989). Monoclonal antibodies to the alternative oxidase of higher plant mitochondria. Plant Physiology, 89, 1311–1317.
Hardie, D. G. (2011). AMP-activated protein kinase: An energy sensor that regulates all aspects of cell function. Genes & Development, 25, 1895–1908.
Hardie, D. G., Carling, D., & Carlson, M. (1998). The AMP-activated/SNF1 protein kinase subfamily: Metabolic sensors of the eukaryotic cell? Annual Review of Biochemistry, 67, 821–855.
Hoefnagel, M., Rich, P. R., Zhang, Q., & Wiskich, J. T. (1997). Substrate kinetics of the plant mitochondrial alternative oxidase and the effects of pyruvate. Plant Physiology, 115, 1145–1153.
Ito, K., Ogata, T., Kakizaki, Y., Elliott, C., Albury, M. S., & Moore, A. L. (2011). Identification of a gene for pyruvate-insensitive mitochondrial alternative oxidase expressed in the thermogenic appendices in Arum maculatum. Plant Physiology, 157, 1721–1732.
Ito, K., Onda, Y., Sato, T., Abe, Y., & Uemura, M. (2003). Structural requirements for the perception of ambient temperature signals in homeothermic heat production of skunk cabbage (Symplocarpus foetidus). Plant, Cell and Environment, 26, 783–788.
Ito, K., & Seymour, R. S. (2005). Expression of uncoupling protein and alternative oxidase depends on lipid or carbohydrate substrates in thermogenic plants. Biology Letters, 1, 427–430.
Kakizaki, Y., Moore, A. L., & Ito, K. (2012). Different molecular bases underlie the mitochondrial respiratory activity in the homoeothermic spadices of Symplocarpus renifolius and the transiently thermogenic appendices of Arum maculatum. Biochemical Journal, 445, 237–246.
Knutson, R. M. (1974). Heat production and temperature regulation in eastern skunk cabbage. Science, 186, 746–747.
Miyagi, A., Takahashi, H., Takahara, K., Hirabayashi, T., Nishimura, Y., Tezuka, T., et al. (2009). Principal component and hierarchical clustering analysis of metabolites in destructive weeds; polygonaceous plants. Metabolomics, 6, 146–155.
Motulsky, H., & Christopoulos, A. (2004). Fitting models to biological data using linear and nonlinear regression: A practical guide to curve fitting. New York: Oxford University Press.
Onda, Y., Kato, Y., Abe, Y., Ito, T., Ito-Inaba, Y., Morohashi, M., et al. (2007). Pyruvate-sensitive AOX exists as a non-covalently associated dimer in the homeothermic spadix of the skunk cabbage, Symplocarpus renifolius. FEBS Letters, 581, 5852–5858.
Polge, C., & Thomas, M. (2007). SNF1/AMPK/SnRK1 kinases, global regulators at the heart of energy control? Trends in Plant Science, 12, 20–28.
Pradet, A., & Raymond, P. (1983). Adenine nucleotide ratios and adenylate energy charge in energy metabolism. Annual Review of Plant Physiology, 34, 199–224.
Rajjou, L., Duval, M., Gallardo, K., Catusse, J., Bally, J., Job, C., et al. (2012). Seed germination and vigor. Annual Review of Plant Biology, 63, 507–533.
Ruben, J. (1995). The evolution of endothermy in mammals and birds: From physiology to fossils. Annual Review of Physiology, 57, 69–95.
Seymour, R. S. (1999). Pattern of respiration by intact inflorescences of the thermogenic arum lily Philodendron selloum. Journal of Experimental Botany, 50, 845–852.
Seymour, R. S. (2004). Dynamics and precision of thermoregulatory responses of eastern skunk cabbage Symplocarpus foetidus. Plant, Cell and Environment, 27, 1014–1022.
Seymour, R. S. (2010). Scaling of heat production by thermogenic flowers: Limits to floral size and maximum rate of respiration. Plant, Cell and Environment, 33, 1474–1485.
Seymour, R. S., & Blaylock, A. J. (1999). Switching off the heater: Influence of ambient temperature on thermoregulation by eastern skunk cabbage Symplocarpus foetidus. Journal of Experimental Botany, 50, 1525–1532.
Seymour, R. S., Gibernau, M., & Pirintsos, S. A. (2009). Thermogenesis of three species of Arum from Crete. Plant, Cell and Environment, 32, 1467–1476.
Seymour, R. S., Lindshau, G., & Ito, K. (2010). Thermal clamping of temperature-regulating flowers reveals the precision and limits of the biochemical regulatory mechanism. Planta, 231, 1291–1300.
Seymour, R. S., & Schultze-Motel, P. (1996). Thermoregulating lotus flowers. Nature, 383, 305.
Seymour, R. S. & Schultze-Motel, P. (1999). Respiration, temperature regulation and energetics of thermogenic inflorescences of the dragon lily Dracunculus vulgaris (Araceae). Proceedings of the Royal Society of London. Series B: Biological Sciences, 266, 1975–1983.
Seymour, R. S., White, C. R., & Gibernau, M. (2003). Environmental biology: Heat reward for insect pollinators. Nature, 426, 243–244.
Silva, J. E. (2006). Thermogenic mechanisms and their hormonal regulation. Physiological Review, 86, 435–464.
Takahashi, H., Imamura, T., Miyagi, A., & Uchimiya, H. (2012). Comparative metabolomics of developmental alterations caused by mineral deficiency during in vitro culture of Gentiana triflora. Metabolomics, 8, 154–163.
Umbach, A. L., Ng, V. S., & Siedow, J. N. (2006). Regulation of plant alternative oxidase activity: A tale of two cysteines. Biochimica et Biophysica Acta, 1757, 135–142.
Vanlerberghe, G. C., & McIntosh, L. (1997). Alternative oxidase: From gene to function. Annual Review of Plant Physiology and Plant Molecular Biology, 48, 703–734.
Vanlerberghe, G. C., Yip, J. Y. H., & Parsons, H. L. (1999). In organello and in vivo evidence of the importance of the regulatory sulfhydryl/disulfide system and pyruvate for alternative oxidase activity in tobacco. Plant Physiology, 121, 793–803.
Wagner, A. M., Krab, K., Wagner, M. J., & Moore, A. L. (2008). Regulation of thermogenesis in flowering Araceae: The role of the alternative oxidase. Biochimica et Biophysica Acta, 1777, 993–1000.
Acknowledgments
This study was partly supported by the Japan Society for the Promotion of Science (JSPS) (Grant-in-Aid for Scientific Research (B) (#22405001 and #24380182), by the FY 2010 Researcher Exchange Program between JSPS and Australian Academy of Science, and by the Prime Minister’s Education Assistance Program for Japan. Support from the Australian Research Council Discovery Grant (DP0771854) is appreciated. Y.K. is supported by a Research Fellowship of the JSPS for Young Scientists. We also thank for Mick Brew who raised D. vulgaris in his garden.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ito, K., Takahashi, H., Umekawa, Y. et al. Metabolite profiling reveals tissue- and temperature-specific metabolomic responses in thermoregulatory male florets of Dracunculus vulgaris (Araceae). Metabolomics 9, 919–930 (2013). https://doi.org/10.1007/s11306-013-0509-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11306-013-0509-x