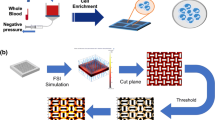
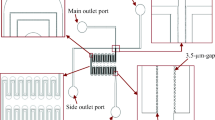
Abstract
Fibrous filters are used widely in transfusion medicine to remove leukocytes (white cells) from platelet-rich plasma (PRP), which is a suspension of leukocytes and platelets in plasma (water and proteins). The interaction of injected leukocytes and platelets with fibers in a filter controls the effectiveness of the filter to selectively retain leukocytes and to allow platelets to pass through. We present comprehensive experimental results at three different spatial scales—cellular, pore, and macroscale—to study PRP filtration in fibrous filters. We used atomic force microscopy (AFM) to measure and compare adhesive forces of the cells brought in contact with clean and protein-coated materials. We then extended cellular-scale experiments to pore-scale flow systems using transparent microfluidic platforms (filter chip) to visualize cell movement in interstitial space and cell capture to microposts under precisely controlled laminar and low-Stokes’-number conditions. We tested filter chips with different surface wettabilities; at the macroscale, we used commercial PRP filters. AFM and filter-chip experiments revealed that plasma proteins play important roles in modifying surface properties to adhere incoming cells. Filter-chip and commercial-filter experimental results showed that adhesion of both leukocytes and platelets decreases at higher shear rates. This reduction was more pronounced for platelets than for leukocytes, which means that results can be used to optimize the selective filtration process.
Similar content being viewed by others
Abbreviations
- AFM:
-
Atomic force microscopy
- CP2D:
-
Citrate phosphate double dextrose buffer
- MPV:
-
Mean particle volume
- PDMS:
-
Poly(dimethylsiloxane)
- PRP:
-
Platelet-rich plasma
- PPP:
-
Platelet-poor plasma
- vdW:
-
van der Waals forces
- z :
-
Distance, nm
- η :
-
Relative cell adhesiveness or cell-capture yield, %
- η L :
-
Leukocyte capture yield, %
- η P :
-
Platelet capture yield, %
References
Baier R.E.: Initial events in interactions of blood with a foreign surface. J. Biomed. Mater. Res. 3, 91–206 (1969)
Balasburamanian V., Grusin N.K., Bucher R.W., Turitto V.T., Slack S.M.: Residence-time dependent changes in fibrinogen adsorbed to polymeric biomaterials. J. Biomed. Mater. Res. 44, 253–260 (1999)
Bhattacharya S., Datta A., Berg J.M., Gangopadhyay S.: Studies on surface wettability of Poly(dimethyl) siloxane (PDMS) and glass under oxygen-plasma treatment and correlation with bond strength. J. Microelectromech. Syst. 14(3), 590–597 (2005)
Bohnert J.L., Horbertt T.A.: Changes in adsorbed fibrinogen and albumin interactions with polymers indicated by decreases in detergent elutability. J. Colloid Interface Sci. 111, 363–377 (1986)
Brenner H., Edwards D.A: Macrotransport Processes. Butterworth-Heinemann, Boston (1993)
Bruil, A.: Leukocyte filters, filtration mechanisms and material design. Ph.D. dissertation, University of Twente, Enschede (1993)
Butt H.J., Cappella B., Kappl M.: Force measurements with the atomic force microscope: technique, interpretation and applications. Surf. Sci. Rep. 59, 1–152 (2005)
De La Rosa C., Tilley P.A., Fox J.D., Kaler K.V.I.S.: Microfluidic device for dielectrophoresis manipulation and electrodisruption of respiratory pathogen Bordetella pertussis. IEEE T. Biomed. Eng. 55(10), 2426–2432 (2008)
Dzik S.: Leukodepletion blood filters: filter design and mechanisms of leukocyte removal. Transfus. Med. Rev. VII, 2–65 (1993)
Freedman J.J., Blajchman M.A., McCombie N.: Canadian Red Cross Society symposium on leukodepletion: report of proceedings. Transfus. Med. Rev. VIII(1), 1–14 (1994)
Garvin J.E.: Factors affecting the adhesiveness of human leucocytes and platelets in vitro. J. Exp. Med. 114, 51–73 (1961)
Goldman M., Delage G.: The role of leukodepletion in the control of transfusion-transmitted disease. Transfus. Med. Rev. IX, 1–9 (1995)
Hull J.R., Tamura G.S., Castner D.G.: Structure and reactivity of adsorbed fibrinonectin films on mica. Biophys. J. 93(8), 2852–2860 (2007)
Israel, L.G., Israel, E.D.: Mechanisms in Hematology, 1st edn. Core Health Services, Inc, Concord (2002)
Israellachvilli J.N.: Intermolecular and Surface Forces. Academic Press, London (2006)
Javadpour F.: CO2 injection in geological formations: determining macroscale coefficients from pore scale processes. Transp Porous Med. 79, 87–105 (2009)
Javadpour, F.: Dispersion and adhesion of blood components in porous media. Ph.D. dissertation, University of Calgary, Calgary (2006)
Javadpour F., Fisher D.: Nanotechnology-based micromodels and new image analysis to study transport in porous media. J. Can. Pet. Technol. 47(2), 30–37 (2008)
Javadpour, F., Jeje, A.: Modeling filtration of platelet-rich plasma in fibrous filters. Transp. Porous. Med. (2011). doi:10.1007/s11242-011-9866-y
Lewis A.L., Freeman R.N.T., Redman R.P., Tolhurst L.A., Kirkwood L.C., Grey D.M., Vick T.A.: Wettable phosphorylcholine-containing polymers useful in blood filtration. J. Mater. Sci. 14, 39–45 (2003)
Nagrath S., Sequist L.V., Maheswaran S., Bell D.W., Irimia D., Ulkus L., Smith M.R., Kwak E.L., Digumarthy S., Muzikansky A., Ryan P., Balis U.J., Tompkins R.G., Haber D.A., Toner M.: Isolation of rare circulating tumor cells in cancer patients by microchip technology. Nature 450, 1235–1239 (2007)
Nightingle S.D.: Universal WBC reduction. Transfusion 1, 1306–1309 (2001)
Parsegian V.A.: Van der Waals Forces, A Handbook for Biologists, Chemists, Engineers, and Physicists. Cambridge University Press, Cambridge (2006)
Retzinger G.S., Cook B.C., Deanglis A.P.: The binding of fibrinogen to surfaces and the identification of two distinct surface-bound species of the protein. J. Colloid Interface Sci. 168, 514–521 (1994)
Retzinger G.S., McGinnis M.C.: A turbidimetric method for measuring fibrin formation and fibrinolysis at solid-liquid interfaces. Anal. Biochem. 186, 169–178 (1990)
Robertson C.R., Chang H.N.: Platelet retention in columns packed with glass beads. Ann. Biomed. Eng. 2, 361–391 (1974)
Rothen A.: Films of protein in biological processes. Adv. Protein Chem. 3, 123–137 (1947)
Scott K.L., Lecak J., Acker J.P.: Biopreservation of red blood cells: past, present, and future. Transfus. Med. Rev. 19(2), 127–142 (2005)
Tang L., Sheu M.S., Chu T.Y., Huang H.: Anti-inflammatory properties of triblock siloxane copolymer-blended materials. Biomaterials 20, 1365–1370 (1999)
Thorslund S., Klett O., Nikolajeff F., Markides K., Bergquist J.: A hybrid poly(dimethylsiloxane) microsystem for on-chip whole blood filtration optimized for steroid screening. Biomed. Microdevices 8, 73–79 (2006)
Tien C.: Granular Filtration of Aerosols and Hydrosols. Butterworths, Boston (1989)
Whitlock P.W., Clarson S.J., Retzinger G.S.: Fibrinogen adsorbs from aqueous media to microscopic droplets of poly(dimethylsiloxane) and remains coagulable. J. Biomed. Mater. Res. 45, 55–61 (1999)
Yazer M.H., Podlosky L., Clarke G., Nahirniak S.M.: The effect of prestorage WBC reduction on the rates of febrile nonhemolytic transfusion reactions to platelet concentrates and RBC. Transfusion 44, 10–15 (2004)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Javadpour, F., Amrein, M. & Jeje, A. Multiscale Experimental Study of Selective Blood-Cell Filtration in Fibrous Porous Media. Transp Porous Med 91, 913–926 (2012). https://doi.org/10.1007/s11242-011-9880-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11242-011-9880-0