Abstract
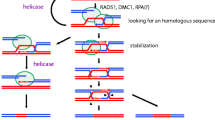
cDNA corresponding to OsRad51 protein was isolated from cDNA library of rice flowers (Oryza sativa, Indica cultivar group) and cloned in to pET28a expression vector. The protein was over expressed in E. coli BL21 (DE3) and purified. Purified OsRad51 could bind single and double stranded DNA, however it showed higher affinity for single stranded DNA. Transmission Electron Microscopy (TEM) studies of OsRad51–DNA complexes showed that this protein formed ring like structures and bound DNA forming filaments. OsRad51 protein promoted renaturation of complementary single strands in to duplex DNA molecules and also showed ATPase activity, which was stimulated by single strand DNA. Fluorescence resonance energy transfer (FRET) assays revealed that OsRad51 promoted homology dependent renaturation as well as strand exchange reactions. Renaturation activity was ATP dependent; however strand exchange activity was ATP independent. This is the first report on in vitro characterization of Rad51 protein from crop plants.








Similar content being viewed by others
Abbreviations
- FRET:
-
Fluorescence resonance energy transfer
- IPTG:
-
Isopropyl thio-galactoside
- MALDI-TOF:
-
Matrix assisted laser desorption and time of flight
- Ni-CAM:
-
Nickel chelating affinity matrix
- ss:
-
Single-stranded
- ds:
-
Double-stranded
- RF:
-
Replicative form
- TEM:
-
Transmission Electron Microscopy
References
Abe K, Osakabe K, Nakayama S, Endo M, Tagiri A, Todoriki S et al (2005) Arabidopsis RAD51C gene is important for homologous recombination in meiosis and mitosis. Plant Physiol 139:896–908. doi:10.1104/pp.105.065243
Amme S, Matros A, Schlesier B, Mock HP (2006) Proteome analysis of cold stress response in Arabidopsis thaliana using DIGE-technology. J Exp Bot 57:1537–1546. doi:10.1093/jxb/erj129
Ayora S, Piruat JI, Luna R, Reiss B, Russo VEA, Aguilera A et al (2002) Characterization of two highly similar Rad51 homologs of Physcomitrella patens. J Mol Biol 316:35–49. doi:10.1006/jmbi.2001.5336
Baumann P, West SC (1998) Role of human Rad51 protein in homologous recombination and double strand break repair. Trends Biochem Sci 23:247–251. doi:10.1016/S0968-0004(98)01232-8
Benson FE, Stasiak A, West SC (1994) Purification and characterization of the human Rad51 protein, an anologue of E. coli RecA. EMBO J 13:5764–5771
Bianco PR, Tracy RB, Kowalczykowski SC (1998) DNA strand exchange proteins: a biochemical and physical comparison. Front Biosci 3:570–603
Bishop DK, Park D, Xu L, Kleckner N (1992) DMC1: a meiosis-specific yeast homolog of E coli recA required for recombination, synaptonemal complex formation, and cell cycle progression. Cell 69:439–456. doi:10.1016/0092-8674(92)90446-J
Bleuyard JY, Gallego ME, Savigny F, White CI (2005) Differing requirements for the Arabidopsis Rad51 paralogs in meiosis and DNA repair. Plant J 41:533–545. doi:10.1111/j.1365-313X.2004.02318.x
Chi P, Van-Komen S, Sehorn MG, Sigurdsson S, Sung P (2006) Roles of ATP binding and ATP hydrolysis in human Rad51 recombinase function. DNA Repair (Amst) 5:381–391. doi:10.1016/j.dnarep.2005.11.005
Conway AB, Lynch TW, Zhang Y, Fortin GS, Fung CW, Symington LS et al (2004) Crystal structure of a Rad51 filament. Nat Struct Mol Biol 11:791–796. doi:10.1038/nsmb795
Doutriaux MP, Couteau F, Bergounioux C, White C (1998) Isolation and characterization of the RAD51 and DMC1 homologues from Arabidopsis thaliana. Mol Gen Genet 257:283–291. doi:10.1007/s004380050649
Dray E, Siaud N, Dubois E, Doutriaux MP (2006) Interaction between Arabidopsis BRCA2 and its partners Rad51, Dmc1, and Dss1. Plant Physiol 140:1059–1069. doi:10.1104/pp.105.075838
Dudas A, Chovanec M (2004) DNA double-strand break repair by homologous recombination. Mutat Res 566:131–167. doi:10.1016/j.mrrev.2003.07.001
Franklin AE, McElver J, Sunjevaric I, Rothstein R, Bowen B, Cande WZ (1999) Three-dimensional microscopy of the Rad51 recombination protein during meiotic prophase. Plant Cell 11:809–824
Franklin AE, Golubovskaya IN, Bass HW, Cande WZ (2003) Improper chromosome synapsis is associated with elongated RAD51 structures in the maize desynaptic2 mutant. Chromosoma 112:17–25. doi:10.1007/s00412-003-0242-8
Gupta RC, Bazemore LR, Golub EI, Radding CM (1997) Activities of human recombination protein Rad51. Proc Natl Acad Sci USA 94:463–468. doi:10.1073/pnas.94.2.463
Hohn B, Puchta H (1999) Gene therapy in plants. Proc Natl Acad Sci USA 96:8321–8323. doi:10.1073/pnas.96.15.8321
Iida S, Terada R (2004) A tale of two integrations, transgene and T-DNA: gene targeting by homologous recombination in rice. Curr Opin Biotechnol 15:132–138. doi:10.1016/j.copbio.2004.02.005
Kant CR, Rao BJ, Sainis JK (2005) DNA binding and pairing activity of OsDmc1, a recombinase from rice. Plant Mol Biol 57:1–11. doi:10.1007/s11103-004-5828-x
Kawabata M, Kawabata T, Nishibori M (2005) Role of recA/RAD51 family proteins in mammals. Acta Med Okayama 59:1–9
Kim HK, Morimatsu K, Nordén B, Ardhammar M, Takahashi M (2002) ADP stabilizes the human Rad51-single stranded DNA complex and promotes its DNA annealing activity. Genes Cells 7:1125–1134. doi:10.1046/j.1365-2443.2002.00588.x
Li W, Chen C, Markmann MU, Timofejeva L, Schmelzer E, Ma H et al (2004) The Arabidopsis AtRAD51 gene is dispensable for vegetative development but required for meiosis. Proc Natl Acad Sci USA 101:10596–10601. doi:10.1073/pnas.0404110101
Li W, Yang X, Lin Z, Timofejeva L, Xiao R, Makaroff CA et al (2005) The AtRAD51C gene is required for normal meiotic chromosome synapsis and double-stranded break repair in Arabidopsis. Plant Physiol 138:965–976. doi:10.1104/pp.104.058347
Markmann MU, Hadi MZ, Koepchen K, Alonso JC, Russo VEA, Schell J et al (2002) The organization of Physcomitrella patens RAD51 genes is unique among eukaryotic organisms. Proc Natl Acad Sci USA 99:2959–2964. doi:10.1073/pnas.032668199
Masson JY, West SC (2001) The Rad51 and Dmc1 recombinases: a non-identical twin relationship. Trends Biochem Sci 26:131–136. doi:10.1016/S0968-0004(00)01742-4
Morita T, Yoshimura Y, Yamamoto A, Murata K, Mori M, Yamamoto H et al (1993) A mouse homolog of the Escherichia coli recA and Saccharomyces cerevisiae RAD51 genes. Proc Natl Acad Sci USA 90:6577–6580. doi:10.1073/pnas.90.14.6577
Osakabe K, Abe K, Yamanouchi H, Takyuu T, Yoshioka T, Ito Y et al (2005) Arabidopsis Rad51B is important for double-strand DNA breaks repair in somatic cells. Plant Mol Biol 57:819–833. doi:10.1007/s11103-005-2187-1
Pawlowski WP, Golubovskaya IN, Cande WZ (2003) Altered nuclear distribution of recombination protein RAD51 in maize mutants suggests the involvement of RAD51 in meiotic homology recognition. Plant Cell 15:1807–1816. doi:10.1105/tpc.012898
Pittman DL, Schimenti JC (2000) Midgestation lethality in mice deficient for the RecA- related gene, Rad51d/Rad51l3. Genesis 26:167–173. doi :10.1002/(SICI)1526-968X(200003)26:3<167::AID-GENE1>3.0.CO;2-M
Puchta H (2005) The repair of double-strand breaks in plants: mechanisms and consequences for genome evolution. J Exp Bot 56:1–14. doi:10.1093/jxb/eri123
Rajanikant C, Kumbhakar M, Pal H, Rao BJ, Sainis JK (2006) DNA strand exchange activity of rice recombinase OsDmc1 monitored by fluorescence resonance energy transfer and the role of ATP hydrolysis. FEBS J 273:1497–1506. doi:10.1111/j.1742-4658.2006.05170.x
Rice KP, Eggler AL, Sung P, Cox MM (2001) DNA pairing and strand exchange by the Escherichia coli RecA and yeast Rad51 proteins without ATP hydrolysis. On the importance of not getting stuck. J Biol Chem 276:38570–38581. doi:10.1074/jbc.M105678200
Schuermann D, Molinie J, Fritsch O, Hohn B (2005) The dual nature of homologous recombination in plants. Trends Genet 21:172–181. doi:10.1016/j.tig.2005.01.002
Sehorn MG, Sigurdsson S, Bussen W, Unger VM, Sung P (2004) Human meiotic recombinase Dmc1 promotes ATP-dependent homologous DNA strand exchange. Nature 429:433–437. doi:10.1038/nature02563
Shinohara A, Ogawa H, Matsuda Y, Ushio N, Ikeo K, Ogawa T (1993) Cloning of human, mouse and fission yeast recombination genes homologous to RAD51 and recA. Nat Genet 4:239–243. doi:10.1038/ng0793-239
Sogo JA, Stasiak W, De Bernadin R, Losa R, Koller T (1987) Binding of proteins to nucleic acids as studied by electron microscopy. In: Sommerville J, Scheer U (eds) Electron microscopy in molecular biology. IRL Press Oxford, England, pp 61–79
Stassen NY, Logsdon JM, Vora GJ, Offenberg HH, Palmer JD, Zolan ME (1997) Isolation and characterization of rad51 orthologs from Coprinus cinereus and Lycopersicon esculentum, and phylogenetic analysis of eukaryotic recA homologs. Curr Genet 31:144–157. doi:10.1007/s002940050189
Symington LS (2002) Role of RAD52 epistasis group genes in homologous recombination and double strand break repair. Microbiol Mol Biol Rev 66:630–670. doi:10.1128/MMBR.66.4.630-670.2002
Terada R, Urawa H, Inagaki Y, Tsugane K, Iida S (2002) Efficient gene targeting by homologous recombination in rice. Nat Biotechnol 20:1030–1034. doi:10.1038/nbt737
Tombline G, Fishel R (2002) Biochemical characterization of human Rad51 protein. 1. ATP hydrolysis. J Biol Chem 222:14417–14425. doi:10.1074/jbc.M109915200
Tsuzuki T, Fujii Y, Sakumi K, Tominaga Y, Nakao K, Sekiguchi M et al (1996) Targeted disruption of the Rad51 gene leads to lethality in embryonic mice. Proc Natl Acad Sci USA 93:6236–6240. doi:10.1073/pnas.93.13.6236
Acknowledgements
We would like to thank all the members of Structural Cell Biology group for TEM work and Dr. Hans-Peter Mock, Dr. Andrea Matros, Applied Biochemistry group for MALDI –TOF analysis at Leibniz Institute of Plant Genetics and Crop Plant Research (IPK), Gatersleben, Germany. Thanks are also due to Professor A. K. Tyagi, University of Delhi, New Delhi, India for providing the rice cDNA library. The International Bureau of Federal Ministry of Education and Research, Germany (BMBF) and the Department of Atomic Energy, India, funded this project under Indo-German Collaborative project IND05/009.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Rajanikant, C., Melzer, M., Rao, B.J. et al. Homologous recombination properties of OsRad51, a recombinase from rice. Plant Mol Biol 68, 479–491 (2008). https://doi.org/10.1007/s11103-008-9385-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11103-008-9385-6