Abstract
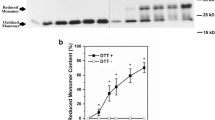
Reactive nitrogen species have been implicated in the pathogenesis of over 40 human diseases, including inflammation. Evidences suggest that reactive nitrogen species such as nitrite/nitrate and halogenated oxidant-HOCl accumulate at the site of inflammation. At physiologically attainable concentrations, HOCl was found to significantly damage the antiproteolytic potential of human α2M and induce subtle changes in conformation as judged by fluorescence analysis. Our studies further suggest that at physiological concentrations, nitrite offered significant protection against HOCl induced α2M inactivation. Our studies suggest that nitrite may act as an antioxidant at physiological concentrations by removing HOCl at sites where both NO2 and HOCl are formed.








Similar content being viewed by others
Abbreviations
- α2M:
-
Alpha-2-Macroglobulin
- HOCl:
-
Hypochlorous acid
- NO2 :
-
Nitrite
- NO2 − :
-
Nitrite anion
- NO:
-
Nitric oxide
References
Bartosz G (1991) Gerontology 37:33–67
Bryan NS, Fernandez BO, Bauer SM, Garcia-Saura MF, Milsom AB, Rassaf T, Maloney RE, Bharti A, Rodriguez J, Feelisch M (2005) Nat Chem Biol 1(5):290–297
Bryan NS, Grisham MB (2007) Free Radic Biol Med 43:645–657
Carr A, Vanderberg JJM, Winterbourn CC (1996) Arch Biochem Biophys 332:63–69
Daugherty A, Dunn JL, Rateri DL, Heinecke JW (1994) J Clin Invest 94:437–444
Dean RT, Fu S, Stocker S, Davies MJ (1997) Biochem J 324:1–18
Eiserich JP, Cross CE, Jones AD, Halliwell B, Van Der Vliet A (1996) J Biol Chem 271:19199–19208
Eiserich JP, Hristova M, Cross CE, Jones AD, Freeman BA, Halliwell B, Van Der Vliet A (1998) Nature 391:393–397
Ganrot PO (1966) Clin Chim Acta 14:493–501
Goaz PW, Biswell HA (1961) J Dent Res 40:355–365
Gonias SL, Sahakian A, Levine RL, Weiss SJ (1993) J Biol Chem 269:4683–4691
Halliwell B (1995) Ann Rheum Dis 54:505–510
Halliwell B, Gutteridge JMC (1984) Biochem J 219:1–14
Halliwell B, Zhao K, Whiteman M (1999) Free Radic Res 31:651–669
Hazell LJ, Arnold L, Flowers D, Waeg G, Malle E, Stocker R (1996) J Clin Invest 97:1535–1544
Hazen SJ, Heinicke JW (1997) J Clin Invest 99:2075–2081
Helallah MIH, Korenaga T (2000) J Chromatogr B 744:433–437
Himmelfarb J, McMenamin ME, Loseto G, Heinecke JW (2001) Free Rad Biol Med 31:1163–1169
Ishiwata H, Tanimura A, Ishidate M (1975) J Food Hyg Soc Jpn 16:89–92
Jasin HE (1993) Inflammation 17:167–181
Johnson DW, Margerum DW (1991) Inorg Chem 30:4845–4851
Kelm M (1999) Biochim Biophys Acta 1411:273–289
Kelm M, Feelisch M, Grube R, Motz W, Strauer BE (1992) In: Moncada S, Marletta MA, Hibbs JRJB, Higgs A (eds) The biology of nitric oxide. Physiological and clinical aspects. Portland Press, London, pp 319–322
Kelm M, Yoshida K (1994) In: Feelisch M, Stamler JS (eds) Metabolic fate of nitric oxide and related oxides. Methods in nitric oxide research. Chichester, Wiley, pp 47–58
Khan FH, Khan SA (2009) Protein Pept Lett 16(1):32–35
Khan FH, Mirza M, Saleemuddin M (1999) J Biochem Mol Biol Biophys 3:109–116
Khan SA, Khan FH (2001) Free Radic Res 34:113–122
Khan SA, Khan FH (2004) Biochim Biophys Acta 1674:139–148
Knight GM, Duncan CW, Leifert C, Golden MH (1999) Brit J Nutr 81:349–358
Laemmli UK (1970) Nature 227:680–685
Leone AM, Francis PL, Rhodes P, Moncada S (1994) Biochem Biophys Res Commun 200:951–957
Lundberg JO, Weitzberg E, Cole JA, Benjamin N (2006) Nature Rev Microbiol 2:593–602
Marnett LJ, Riggins JN, West JD (2003) J Clin Invest 111:583–593
Martinez-Cayuela M (1995) Biochimie 77:147–161
Morris JC (1966) J Phys Chem 70:3798–3805
Panasenko OM, Briviba J, Klotz LO, Sies H (1997) Arch Biochem Biophys 343:245–259
Schaur J, Jerlich A, Stelmaszynska T (1998) Curr Top Biophys 22(Suppl. B):176–185
Sottrup-Jensen L (1989) J Biol Chem 264:11539–11542
Spencer JPE, Whiteman M, Jenner A, Halliwell B (2000) Free Rad Biol Med 28:1039–1050
Sud A, Khullar M, Wanchu A, Bambert P (2000) Nitric Oxide Biol Chem 4:615–619
Torre D, Ferrario G, Speranza F, Fiori GP, Zeroli P (1996) J Clin Pathol 49:574–576
Ueda T, Maekawa T, Sadamitsu D, Ohshita S, Ogino K, Nakamura K (1995) Electrophoresis 16:1002–1004
Uesugi M, Yoshidam K, Jasin HE (2000) J Immunol 165:6532–6537
Van Der Vliet A, Nguyen MN, Shigenaga MK, Eiserich JP, Marelich GP, Cross CE (2000) Am J Physiol Lung Cell Mol Physiol 279:L537–L546
Wanchu A, Agnihotri N, Deodhar SD, Ganguly NK (1996) Ind J Med Res 104:263–268
Wasil M, Halliwell B, Grootveld M, Moorhouse CP, Hutchison DCS, Baum H (1987) Biochem J 243:867
Wasil M, Halliwell B, Hutchison DCS, Baum H (1987) Biochem J 243:219–223
Whiteman M, Hooper DC, Scott GS, Koprowski H, Halliwell B (2002) Proc Natl Acad Sci USA 99:12061–12066
Whiteman M, Jenner A, Halliwell B (1997) Chem Res Toxicol 10:1240–1246
Whiteman M, Jenner J, Halliwell B (1999) Biomarkers 4:303–310
Whiteman M, Rose P, Halliwell B (2003) Biochem Biophys Res Commun 303:1217–1224
Whiteman M, Sialu JL, Halliwell B (2003) J Biol Chem 278:8380–8384
Wink DA, Grisham MB, Mitchell JB, Ford PC (1996) Methods Enzymol 268:12–31
Winterbourn CC, Kettle AJ (2000) Free Rad Biol Med 29:403–499
Wu SM, Pizzo SV (2001) Arch Biochem Biophys 391:119–126
Zavodnik IB, Lapshina EA, Zavodnik LB, Bartosz G, Soszynski M, Bryszcwska M (2001) Free Radic Biol Med 30:363–369
Acknowledgments
MWK would like to thank the Indian Council of Medical Research, New Delhi for a research fellowship. FHK acknowledges the generous financial support provided by UGC-DRS program for this study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wasim Khan, M., Naqshbandi, A., Zubair, H. et al. Nitrite, a Reactive Nitrogen Species, Protects Human Alpha-2-Macroglobulin from Halogenated Oxidant, HOCl. Protein J 29, 276–282 (2010). https://doi.org/10.1007/s10930-010-9249-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10930-010-9249-1