Abstract
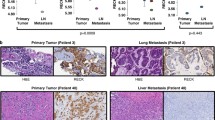
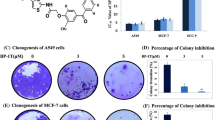
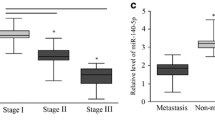
A significant proportion of breast cancer patients harbor clinically undetectable micrometastases at the time of diagnosis. If left untreated, these micro-metastases may lead to disease relapse and possibly death. Hence, there is significant interest in the development of novel anti-metastatic agents that could also curb the growth of pre-established micrometastases. Like primary tumor, the growth of metastases also is driven by angiogenesis. Although the role of cysteine protease Cathepsin L (CTSL) in metastasis associated tumor cell functions such as migration and invasion is well recognized, its role in tumor angiogenesis remains less explored. The present study examines the contribution of CTSL to breast cancer angiogenesis and evaluates the anti-angiogenic efficacy of CTSL inhibitor KGP94. CTSL semi-quantitative RT-PCR analysis on breast tissue panels revealed significant upregulation of CTSL in breast cancer patients which strongly correlated with increased relapse and metastatic incidence and poor overall survival. Preclinically, CTSL ablation using shRNA or KGP94 treatment led to a significant reduction in MDA-MB-231 tumor cell induced angiogenesis in vivo. In-vitro assessments demonstrated a significant decrease in various angiogenic properties such as endothelial cell sprouting, migration, invasion, tube formation and proliferation in the presence of KGP94. Microarray analyses revealed a significant upregulation of cell cycle related genes by CTSL. Western blot analyses further confirmed upregulation of members of the cyclin family by CTSL. Collectively, these data indicate that CTSL is an important contributor to tumor angiogenesis and that the CTSL inhibition may have therapeutic utility in the treatment of breast cancer patients.





Similar content being viewed by others
References
Siegel R et al (2014) Cancer statistics, 2014. CA Cancer J Clin 64(1):9–29
Hanahan D, Weinberg RA (2011) Hallmarks of cancer: the next generation. Cell 144(5):646–674
Siemann DW (2011) The unique characteristics of tumor vasculature and preclinical evidence for its selective disruption by tumor-vascular disrupting agents. Cancer Treat Rev 37(1):63–74
Weidner N et al (1991) Tumor angiogenesis and metastasis–correlation in invasive breast carcinoma. N Engl J Med 324(1):1–8
Burden RE et al (2009) Antibody-mediated inhibition of cathepsin S blocks colorectal tumor invasion and angiogenesis. Clin Cancer Res 15(19):6042–6051
Lakka SS et al (2004) Inhibition of cathepsin B and MMP-9 gene expression in glioblastoma cell line via RNA interference reduces tumor cell invasion, tumor growth and angiogenesis. Oncogene 23(27):4681–4689
Chauhan SS, Goldstein LJ, Gottesman MM (1991) Expression of cathepsin L in human tumors. Cancer Res 51(5):1478–1481
Sudhan DR, Siemann DW (2015) Cathepsin L targeting in cancer treatment. Pharmacol Ther 155:105–116
Chapman HA, Riese RJ, Shi GP (1997) Emerging roles for cysteine proteases in human biology. Annu Rev Physiol 59:63–88
Gottesman MM, Sobel ME (1980) Tumor promoters and Kirsten sarcoma virus increase synthesis of a secreted glycoprotein by regulating levels of translatable mRNA. Cell 19(2):449–455
Rabin MS, Doherty PJ, Gottesman MM (1986) The tumor promoter phorbol 12-myristate 13-acetate induces a program of altered gene expression similar to that induced by platelet-derived growth factor and transforming oncogenes. Proc Natl Acad Sci USA 83(2):357–360
Denhardt DT et al (1986) Close relationship of the major excreted protein of transformed murine fibroblasts to thiol-dependent cathepsins. Cancer Res 46(9):4590–4593
Jordans S et al (2009) Monitoring compartment-specific substrate cleavage by cathepsins B, K, L, and S at physiological pH and redox conditions. BMC Biochem 10:23
Novinec M, Lenarcic B, Turk B (2014) Cysteine cathepsin activity regulation by glycosaminoglycans. Biomed Res Int 2014:309718
Everts V et al (2006) Osteoclastic bone degradation and the role of different cysteine proteinases and matrix metalloproteinases: differences between calvaria and long bone. J Bone Miner Res 21(9):1399–1408
Goretzki L et al (1992) Effective activation of the proenzyme form of the urokinase-type plasminogen activator (pro-uPA) by the cysteine protease cathepsin L. FEBS Lett 297(1–2):112–118
Ishidoh K, Kominami E (1995) Procathepsin L degrades extracellular matrix proteins in the presence of glycosaminoglycans in vitro. Biochem Biophys Res Commun 217(2):624–631
Laurent-Matha V et al (2006) Processing of human cathepsin D is independent of its catalytic function and auto-activation: involvement of cathepsins L and B. J Biochem 139(3):363–371
Mason RW et al (1986) Elastinolytic activity of human cathepsin L. Biochem J 233(3):925–927
Frade R et al (1998) Procathepsin-L, a proteinase that cleaves human C3 (the third component of complement), confers high tumorigenic and metastatic properties to human melanoma cells. Cancer Res 58(13):2733–2736
Gocheva V et al (2006) Distinct roles for cysteine cathepsin genes in multistage tumorigenesis. Genes Dev 20(5):543–556
Chavarria GE et al (2012) Initial evaluation of the antitumour activity of KGP94, a functionalized benzophenone thiosemicarbazone inhibitor of cathepsin L. Eur J Med Chem 58:568–572
Sudhan DR, Siemann DW (2013) Cathepsin L inhibition by the small molecule KGP94 suppresses tumor microenvironment enhanced metastasis associated cell functions of prostate and breast cancer cells. Clin Exp Metastasis 30(7):891–902
Sudhan DR et al (2016) Cathepsin L inactivation leads to multimodal inhibition of prostate cancer cell dissemination in a preclinical bone metastasis model. Int J Cancer 138:2665–2677
Kumar GD et al (2010) Functionalized benzophenone, thiophene, pyridine, and fluorene thiosemicarbazone derivatives as inhibitors of cathepsin L. Bioorg Med Chem Lett 20(22):6610–6615
Gyorffy B et al (2010) An online survival analysis tool to rapidly assess the effect of 22,277 genes on breast cancer prognosis using microarray data of 1,809 patients. Breast Cancer Res Treat 123(3):725–731
Siemann DW et al (2009) Impact of tumor cell VEGF expression on the in vivo efficacy of vandetanib (ZACTIMA; ZD6474). Anticancer Res 29(6):1987–1992
Smyth GK (2005) Limma: linear models for microarray data, vol. 397–420. Springer, New York
de Hoon MJL, Imoto S, Nolan J, Miyano S (2004) Open source clustering software. Bioinform Appl Notes 20(9):1453–1454
Subramanian A et al (2005) Gene set enrichment analysis: a knowledge-based approach for interpreting genome-wide expression profiles. Proc Natl Acad Sci USA 102(43):15545–15550
Shannon P et al (2003) Cytoscape: a software environment for integrated models of biomolecular interaction networks. Genome Res 13(11):2498–2504
Montojo J et al (2010) GeneMANIA Cytoscape plugin: fast gene function predictions on the desktop. Bioinformatics 26(22):2927–2928
Nishida Y et al (1995) Increased cathepsin L levels in serum in some patients with ovarian cancer: comparison with CA125 and CA72-4. Gynecol Oncol 56(3):357–361
Siewinski M et al (2004) Determination of cysteine peptidases-like activity and their inhibitors in the serum of patients with ovarian cancer treated by conventional chemotherapy and vitamin E. J Exp Ther Oncol 4(3):189–193
Svatek RS et al (2008) Role of urinary cathepsin B and L in the detection of bladder urothelial cell carcinoma. J Urol 179(2):478–484 discussion 84
Tumminello FM et al (1996) Cathepsin D, B and L circulating levels as prognostic markers of malignant progression. Anticancer Res 16(4B):2315–2319
van Hinsbergh VW, Koolwijk P (2008) Endothelial sprouting and angiogenesis: matrix metalloproteinases in the lead. Cardiovasc Res 78(2):203–212
Arnaoutova I, Kleinman HK (2010) In vitro angiogenesis: endothelial cell tube formation on gelled basement membrane extract. Nat Protoc 5(4):628–635
Zhang J et al (2010) Plasma cathepsin L and its related pro/antiangiogenic factors play useful roles in predicting rich coronary collaterals in patients with coronary heart disease. J Int Med Res 38(4):1389–1403
Sun J et al (2011) Cathepsin L activity is essential to elastase perfusion-induced abdominal aortic aneurysms in mice. Arterioscler Thromb Vasc Biol 31(11):2500–2508
Keerthivasan S et al (2007) Transcriptional upregulation of human cathepsin L by VEGF in glioblastoma cells. Gene 399(2):129–136
Jain M et al (2010) Cathepsins B and L in peripheral blood mononuclear cells of pediatric acute myeloid leukemia: potential poor prognostic markers. Ann Hematol 89(12):1223–1232
Samaiya M et al (2011) Epigenetic regulation of cathepsin L expression in chronic myeloid leukaemia. J Cell Mol Med 15(10):2189–2199
Tamada Y et al (2000) Involvement of cysteine proteases in bFGF-induced angiogenesis in guinea pig and rat cornea. J Ocul Pharmacol Ther 16(3):271–283
Chung JH et al (2011) Cathepsin L derived from skeletal muscle cells transfected with bFGF promotes endothelial cell migration. Exp Mol Med 43(4):179–188
Asanuma K et al (2002) Selective modulation of the secretion of proteinases and their inhibitors by growth factors in cultured differentiated podocytes. Kidney Int 62(3):822–831
Eilken HM, Adams RH (2010) Dynamics of endothelial cell behavior in sprouting angiogenesis. Curr Opin Cell Biol 22(5):617–625
Frade R, Rousselet N, Jean D (2008) Intratumoral gene delivery of anti-cathepsin L single-chain variable fragment by lentiviral vector inhibits tumor progression induced by human melanoma cells. Cancer Gene Ther 15(9):591–604
Dennemarker J et al (2010) Deficiency for the cysteine protease cathepsin L promotes tumor progression in mouse epidermis. Oncogene 29(11):1611–1621
Urbich C et al (2005) Cathepsin L is required for endothelial progenitor cell-induced neovascularization. Nat Med 11(2):206–213
Handsley MM, Edwards DR (2005) Metalloproteinases and their inhibitors in tumor angiogenesis. Int J Cancer 115(6):849–860
Rebbaa A et al (2009) The anti-angiogenic activity of NSITC, a specific cathepsin L inhibitor. Anticancer Res 29(11):4473–4481
Yu Q, Stamenkovic I (2000) Cell surface-localized matrix metalloproteinase-9 proteolytically activates TGF-beta and promotes tumor invasion and angiogenesis. Genes Dev 14(2):163–176
Yu WH et al (2002) CD44 anchors the assembly of matrilysin/MMP-7 with heparin-binding epidermal growth factor precursor and ErbB4 and regulates female reproductive organ remodeling. Genes Dev 16(3):307–323
Paez-Ribes M et al (2009) Antiangiogenic therapy elicits malignant progression of tumors to increased local invasion and distant metastasis. Cancer Cell 15(3):220–231
Cairns RA, Hill RP (2004) Acute hypoxia enhances spontaneous lymph node metastasis in an orthotopic murine model of human cervical carcinoma. Cancer Res 64(6):2054–2061
Keith B, Simon MC (2007) Hypoxia-inducible factors, stem cells, and cancer. Cell 129(3):465–472
Horsman MR, Siemann DW (2006) Pathophysiologic effects of vascular-targeting agents and the implications for combination with conventional therapies. Cancer Res 66(24):11520–11539
Siemann DW, Horsman MR (2015) Modulation of the tumor vasculature and oxygenation to improve therapy. Pharmacol Ther 153:107–124
Acknowledgments
The authors thank Dr. Kevin Pinney of Baylor University and OXiGENE Inc. for providing KGP94. These studies were supported in part by a grant from the National Cancer Institute (US Public Health Service Grant R01 CA169300).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors have no conflict of interest to disclose.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sudhan, D.R., Rabaglino, M.B., Wood, C.E. et al. Cathepsin L in tumor angiogenesis and its therapeutic intervention by the small molecule inhibitor KGP94. Clin Exp Metastasis 33, 461–473 (2016). https://doi.org/10.1007/s10585-016-9790-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10585-016-9790-1