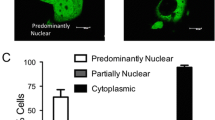
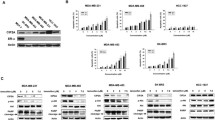
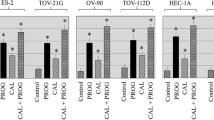
Abstract
Estrogens stimulate growth of hormone-dependent breast cancer but paradoxically induce tumor regress under certain circumstances. We have shown that long-term estrogen deprivation (LTED) enhances the sensitivity of hormone dependent breast cancer cells to estradiol (E2) so that physiological concentrations of estradiol induce apoptosis in these cells. E2-induced apoptosis involve both intrinsic and extrinsic pathways but precise mechanisms remain unclear. We found that exposure of LTED MCF-7 cells to E2 activated AMP activated protein kinase (AMPK). In contrast, E2 inhibited AMPK activation in wild type MCF-7 cells where E2 prevents apoptosis. As a result of AMPK activation, the transcriptional activity of FoxO3, a downstream factor of AMPK, was up-regulated in E2 treatment of LTED. Increased activity of FoxO3 was demonstrated by up-regulation of three FoxO3 target genes, Bim, Fas ligand (FasL), and Gadd45α. Among them, Bim and FasL mediate intrinsic and extrinsic apoptosis respectively and Gadd45α causes cell cycle arrest at the G2/M phase. To further confirm the role of AMPK in apoptosis, we used AMPK activator AICAR in wild type MCF-7 cells and examined apoptosis, proliferation and expression of Bim, FasL, and Gadd45α. The effects of AICAR on these parameters recapitulated those observed in E2-treated LTED cells. Activation of AMPK by AICAR also increased expression of Bax in MCF-7 cells and its localization to mitochondria, which is a required process for apoptosis. These results reveal that AMPK is an important factor mediating E2-induced apoptosis in LTED cells, which is implicative of therapeutic potential for relapsing breast cancer after hormone therapy.






Similar content being viewed by others
References
Yue W, Wang J-P, Conaway MR, Li Y, Santen RJ (2003) Adaptive hypersensitivity following long-term estrogen deprivation: involvement of multiple signal pathways. J Steroid Biochem Mol Biol 86:265–274
Masamura S, Santner SJ, Heitjan DF, Santen RJ (1995) Estrogen deprivation causes estradiol hypersensitivity in human breast cancer cells. J Clin Endocrinol Metab 80(10):2918–2925
Song RX-D, Mor G, Naftolin F, McPherson RA, Song J, Zhang Z, Yue W, Wang J-P, Santen RJ (2001) Effect of long-term estrogen deprivation on apoptotic responses of breast cancer cells to 17β-estradiol. J Natl Cancer Inst 93(22):1714–1723
Lewis JS, Meeke K, Osipo C, Ross EA, Kidawi N, Li T, Bell E, Chandel NS, Jordan VC (2005) Intrinsic mechanism of estradiol-Induced apoptosis in breast cancer cells resistant to estrogen deprivation. J Natl Cancer Inst 97(23):1746–1759
Carter AC, Sedransk N, Kelley RM et al (1977) Diethylstilbestrol: recommended dosages for different categories of breast cancer patients: report of the cooperative breast cancer group. JAMA 237(19):2079–2085. doi:10.1001/jama.1977.03270460065023
Yue W, Wang J-P, Zhang L, Wang W, Santen RJ, Aiyar SE (2013) Induction of apoptosis in hormone refractory breast cancer: horizontal modulation is superior to vertical. J Exp Ther Oncol 10:16–179
Greer EL, Brunet A (2005) FOXO transcription factors at the interface between longevity and tumor suppresion. Oncogene 24:7410–7425
Zhang Y, Gan B, Liu D, J-h Paik (2011) FoxO family members in cancer. Cancer Biol Ther 12:253–259
Brunet A, Bonni A, Zigmond MJ, Lin M, Juo ZP, Hu LS, Anderson MJ, Arden KC, Blenis J, Greenberg ME (1999) Akt promotes cell survival by phosphorylating and inhibiting a Forkhead transcription factor. Cell Cycle 96:857–868
Greer EL, Oskoui PR, Banko MR, Maniar JM, Gygi MP, Gygi SP, Brunet A (2007) The energy sensor AMP-activated protein kinase directly regulates the mammalian FOXO3 transcription factor. J Biol Chem 282:30107–30119
Hu MCT, Lee D-F, Xia W, Golfman LS, Ou-Yang F, Yang J-Y, Zou Y, Bao S, Hanada N, Saso H, Kobayashi R, Hung M-C (2004) IκB kinase promotes tumorigenesis through inhibition of forkhead FOXO3a. Cell 117(2):225–237. doi:10.1016/S0092-8674(04)00302-2
Yang J-Y, Zong CS, Xia W, Yamaguchi H, Ding Q, Xie X, Lang J-Y, Lai C-C, Chang C-J, Huang W-C, Huang H, Kuo H-P, Lee D-F, Li L-Y, Lien H-C, Cheng X, Chang K-J, Hsiao C-D, Tsai F-J, Tsai C-H, Sahin AA, Muller WJ, Mills GB, Yu D, Hortobagyi GN, Hung M-C (2008) ERK promotes tumorigenesis by inhibiting FOXO3a via MDM2-mediated degradation. Nat Cell Biol 10(2):138–148. http://www.nature.com/ncb/journal/v10/n2/suppinfo/ncb1676_S1.html
Kim J-H, Choi S-Y, Kang B-H, Lee S-M, Park HS, Kang G-Y, Bang JY, Cho E-J, Youn H-D (2013) AMP-activated protein kinase phosphorylates CtBP1 and down-regulates its activity. Biochem Biophys Res Commun 431:8–13
Jiang S-y, Wolf DM, Yingling JM, Chang C, Jordan VC (1992) An estrogen receptor positive MCF-7 clone that is resistant to antiestrogens and estradiol. Mol Cell Endocrinol 90(1):77–86. doi:10.1016/0303-7207(92)90104-E
Pathiraja TN, Nayak SR, Xi Y, Jiang S, Garee JP, Edwards DP, Lee AV, Chen J, Shea MJ, Santen RJ, Gannon F, Kangaspeska S, Jelinek J, Issa J-PJ, Richer JK, Elias A, McIlroy M, Young LS, Davidson NE, Schiff R, Li W, Oesterreich S (2014) Epigenetic reprogramming of HOXC10 in endocrine-resistant breast cancer. Sci Transl Med 6(229):229ra241. doi:10.1126/scitranslmed.3008326
Chan CM, Martin LA, Johnston SR, Ali S, Dowsett M (2002) Molecular changes associated with the acquisition of oestrogen hypersensitivity in MCF-7 breast cancer cells on long-term oestrogen deprivation. J Steroid Biochem Mol Biol 81:333–341
Hardie DG, Scott JW, Pan DA, Hudson ER (2003) Management of cellular energy by the AMP-activated protein kinase system. FEBS Lett 546(1):113–120. doi:10.1016/S0014-5793(03)00560-X
Kemp BE, Stapleton D, Campbell DJ, Chen ZP, Murthy S, Walter M, Gupta A, Adams JJ, Katsis F, van Denderen B, Jennings IG, Iseli T, Michell BJ, Witters LA (2003) AMP-activated protein kinase, super metabolic regulator. Biochem Soc Trans 31:162–168
Towler MC, Hardie DG (2007) AMP-activated protein kinase in metabolic control and insulin signaling. Circ Res 100:328–341
Meisse D, Van de Casteele M, Beauloye C, Hainault I, Kefas BA, Rider MH, Foufelle F, Hue L (2002) Sustained activation of AMP-activated protein kinase induces c-Jun N-terminal kinase activation and apoptosis in liver cells. FEBS Lett 526:38–42
Sauer H, Engel S, Milosevic N, Sharifpanah F, Wartenberg M (2012) Activation of AMP-kinase by AICAR induces apoptosis of DU-145 prostate cancer cells through generation of reactive oxygen species and activation of c-Jun N-terminal kinase. Int J Oncol 40:501–508
Lee K-H, Hsu E-C, Guh J-H, Yang H-C, Wang D, Kulp SK, Shapiro CL, Chen C-S (2011) Targeting energy metabolic and oncogenic signaling pathways in triple-negative breast cancer by a novel adenosine monophosphate-activated protein kinase (AMPK) activator. J Biol Chem 286(45):39247–39258
Brown KA, McInnes KJ, Takagi K, Ono K, Hunger N, Wang L, Sasano H, Simpson ER (2011) LKB1 expression is inhibited by estradiol-17β in MCF-7 cells. J Steroid Biochem Mol Biol 127:439–443
Gilley J, Coffer PJ, Ham J (2003) FOXO transcription factors directly activate bim gene expression and promote apoptosis in sympathetic neurons. J Cell Biol 162(4):613–622
Dijkers PF, Medema RH, Lammers J-WJ, Koenderman L, Coffer PJ (2000) Expression of the pro-apoptotic Bcl-2 family member Bim is regulated by the forkhead transcription factor FKHR-L1. Curr Biol 10:1201–1204
Sunters A, de Mattos SF, Stahl M, Brosens JJ, Zoumpoulidou G, Saunders CA, Coffer PJ, Medema BH, Coombes RC, Lam EW-F (2003) FoxO3a transcriptional regulation of Bim controls apoptosis in paclitaxel-treated breast cancer cell lines. J Biol Chem 278:49795–49805
Song RX-D, Chen Y, Zhang Z, Bao Y, Yue W, Wang J, Fan P, Santen RJ (2010) Estrogen utilization of IGF-1-R and EGF-R to signal in breast cancer cells. J Steroid Biochem Mol Biol 118:219–230
Grooteclaes M, Deveraux Q, Hildebrand J, Zhang Q, Goodman RH, Frisch SM (2003) C-terminal-binding protein corepresses epithelial and proapoptotic gene expression programs. Proc Natl Acad Sci USA 100:4568–4573
Cook KL, Shajahan AN, Wärri A, Jin L, Hilakivi-Clarke LA, Clarke R (2012) Glucose-regulated protein 78 controls cross-talk between apoptosis and autophagy to determine antiestrogen responsiveness. Cancer Res 72:3337–3349
Cook KL, Wärri A, Soto-Pantoja DR, Clarke PA, Cruz MI, Zwart A, Clarke R (2014) Hydroxychloroquine inhibits autophagy to potentiate antiestrogen responsiveness in ER+ breast cancer. Clin Cancer Res 20(12):3222–3232. doi:10.1158/1078-0432.CCR-13-3227
Yue W, Fan P, Wang J-P, Li Y, Santen RJ (2007) Mechanisms of acquired resistance to endocrine therapy in hormone-dependent breast cancer cells. J Steroid Biochem Mol Biol 106:102–110
Acknowledgments
The current studies were supported by Department of Defense Grant (W81XWH-10-1-0030). The authors acknowledge Dr. Craig Jordan for providing MCF-7-5C cells.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Chen, H., Wang, Jp., Santen, R.J. et al. Adenosine monophosphate activated protein kinase (AMPK), a mediator of estradiol-induced apoptosis in long-term estrogen deprived breast cancer cells. Apoptosis 20, 821–830 (2015). https://doi.org/10.1007/s10495-015-1111-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10495-015-1111-7