Abstract
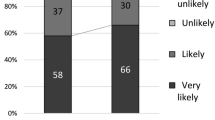
We evaluated the adherence and acceptability of a vaginal ring containing dapivirine, maraviroc, or both drugs for 28 days during a Phase I placebo-controlled trial in 48 HIV-negative sexually abstinent U.S. women aged 18–40. Adherence was assessed weekly by clinical interview and computer-assisted self-interviewing; acceptability assessment occurred at the last product-use visit. Study retention was 98 % (47/48); 94 % (45/48) reported being fully adherent with ring use during the 28-day period. Two participants experienced the ring partially coming out. Analysis was blinded and behavioral data were combined across study groups. Most women reported being very comfortable having the ring in their vagina; 44 % preferred continuous use, whereas 51 % had no preference compared to episodic use. Although a range of minor ring concerns were expressed, few were actually experienced. High adherence to and acceptability of this vaginal ring in this Phase I trial contributes to its promise as a sustained mechanism for multidrug vaginal microbicide delivery.


Similar content being viewed by others
Notes
The link to the vaginal ring use instructions sheet provided to participants is http://www.mtnstopshiv.org/sites/default/files/attachments/Section%209%20-Study%20Product%20Considerations%20-%20Version%201.0%2002Sept11.pdf (Section. 9.1).
References
van der Straten A, Van Damme L, Haberer JE, Bangsberg DR. Unraveling the divergent results of pre-exposure prophylaxis trials for HIV prevention. AIDS. 2012;26(7):F13–9.
Baeten J, Celum C. Systemic and topical drugs for the prevention of HIV infection: antiretroviral pre-exposure prophylaxis. Annu Rev Med. 2013;64:219.
Hendrix CW, Andrade A, Kashuba A, et al. Tenofovir-Emtricitabine directly observed dosing: 100 % adherence concentrations (HPTN 066). Paper presented at: 21st Conference on Retorviruses and Opportunistic Infections. Boston; 2014.
Amico K. Adherence to preexposure chemoprophylaxis: the behavioral bridge from efficacy to effectiveness. Curr Opin HIV AIDS. 2012;7:542–8.
Creinin MD, Meyn LA, Borgatta L, et al. Multicenter comparison of the contraceptive ring and patch: a randomized controlled trial. Gynecol Oncol. 2008;111(2, Part 1):267–77. doi:10.1097/01.AOG.0000298338.58511.d1.
UNAIDS. Global Report: UNAIDS report on the global AIDS epidemic 2013. Geneva, Switzerland: Joint United Nations Programme on HIV/AIDS (UNAIDS). 2013.
Friend D. Intravaginal rings: controlled release systems for contraception and prevention of transmission of sexually transmitted infections. Drug Deliv Transl Res. 2011;1(3):185–93.
Woodsong C, Montgomery E, Masenga G, et al. Safety and acceptability of vaginal ring as microbicide delivery method in African women. Paper presented at Microbicides, Pittsburg; Accessed 23–26 May 2010.
van der Straten A, Woodsong C, ET M, Nel A. High adherence and acceptability of a monthly Dapivirine vaginal ring for HIV prevention in Africa. Paper presented at International Microbicides Conference, Sydney, Australia; 2012.
Nel A, Kamupira M, Woodsong C, van der Straten A, ET M, van Niekerk N. Safety, acceptability, and pharmacokinetics assessment (adherence of monthly Dapivirine Vaginal Microbicide Rings (Ring-004) for HIV prevention. Paper presented at Conference on Retrovirus and Opportunistic Infections, Seattle; 2012.
Montgomery E, van der Straten A, Cheng H, et al. Vaginal ring adherence in Sub-Saharan Africa: expulsion, removal, and perfect use. AIDS Behav. 2012;16:1787–98.
Phase III trial of dapivirine ring begins in Africa: ASPIRE testing new HIV prevention approach for women. http://www.mtnstopshiv.org/node/4546: Microbicides Trial Network; 2012.
First efficacy trial of a microbicide ring to prevent HIV is underway: the ring study to assess IPM’s monthly ARV ring for women. http://www.ipmglobal.org/publications/first-efficacy-trial-microbicide-ring-prevent-hiv-underway: International Partnership for Microbicides, Inc.; 2012.
NuvaRing. Registration In South Africa, MSD pharmaceuticals, 2012.
Novak A, de la Loge C, Abetz L, van der Meulen EA. The combined contraceptive vaginal ring, NuvaRing: an international study of user acceptability. Contraception. 2003;67(3):187–94.
Brache V, Faundes A. Contraceptive vaginal rings: a review. Contraception. 2010;82(5):418–27.
Brache V, Payán LJ, Faundes A. Current status of contraceptive vaginal rings. Contraception. 2013;87(3):264–72.
Stewart FH, Brown BA, Raine TR, Weitz TA, Harper CC. Adolescent and young women’s experience with the vaginal ring and oral contraceptive pills. J Pediatr Adolesc Gynecol. 2007;20(6):345–51.
Chen B, Panther L, Marzinke MA, et al. Phase 1 Safety, pharmacokinetics, and pharmacodynamics of dapivirine and Maraviroc vaginal rings: a double-blind randomized trial. J Acquir Immune Defic Syndr. 2015;70:242–49.
van der Straten A, Montgomery E, Cheng H, et al. High acceptability of a vaginal ring intended as a microbicide delivery method for HIV prevention in African women. AIDS Behav. 2012;16:1775–86.
van den Berg JJ, Rosen RK, Bregman DE, et al. “Set it and forget it”: women’s perceptions and opinions of long-acting topical vaginal gels. AIDS Behav. 2014;18(5):862–70.
Malcolm R, Fetherston S, McCoy C, Boyd P, Major I. Vaginal rings for delivery of HIV microbicides. Int J Women’s Health. 2012;4(1):595–605.
Minnis AM, van der Straten A, Salee P, Hendrix CW. Pre-exposure prophylaxis adherence measured by plasma drug level in MTN-001: comparison between vaginal gel and oral tablets in two geographic regions. AIDS Behav. 2015.
Epstein LB, Sokal-Gutierrez K, Ivey SL, Raine T, Auerswald C. Adolescent experiences with the vaginal ring. J Adolesc Health. 2008;43(1):64–70.
Witte SS, El-Bassel N, Gilbert L, Wu E, Chang M, Hill J. Promoting female condom use to heterosexual couples: findings from a randomized clinical trial. Perspect Sex Reprod Health. 2006;38(3):148–54.
Montgomery E, Blanchard K, Cheng H, et al. Diaphragm and lubricant gel acceptance, skills and patterns of use among women in an effectiveness trial in Southern Africa. Eur J Contracept Reprod Health Care. 2009;14(6):410–9.
Acknowledgements
We would like to acknowledge the women who participated in this study and to extend special thanks to the clinical research sites’ teams in Pittsburgh, PA, Birmingham, AL, and Boston, MA. The full MTN-013/IPM 026 study team can be viewed at http://www.mtnstopshiv.org/studies/2241. The study was designed and implemented by the Microbicide Trials Network (MTN). The MTN is funded by the National Institute of Allergy and Infectious Diseases (UM1AI068633, UM1AI068615, UM1AI106707), with co-funding from the Eunice Kennedy Shriver National Institute of Child Health and Human Development and the National Institute of Mental Health, all components of the U.S. National Institutes of Health. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health. The International Partnership for Microbicides (IPM), IND sponsor, supplied the products used in this study [Dapivirine vaginal ring (VR), maraviroc VR, maraviroc/dapivirine VR, placebo VR].
Author information
Authors and Affiliations
Corresponding author
Additional information
On behalf of the MTN-013/IPM 026 Protocol Team for the Microbicide Trials Network.
Rights and permissions
About this article
Cite this article
van der Straten, A., Panther, L., Laborde, N. et al. Adherence and Acceptability of a Multidrug Vaginal Ring for HIV Prevention in a Phase I Study in the United States. AIDS Behav 20, 2644–2653 (2016). https://doi.org/10.1007/s10461-016-1299-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10461-016-1299-8