Abstract
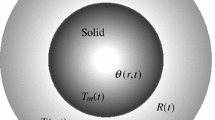
In this paper, we study the melting of a spherical nanoparticle. The model differs from previous ones in that a number of features have been incorporated to match experimental observations. These include the size dependence of the latent heat and a cooling condition at the boundary (as opposed to the fixed temperature condition used in previous studies). Melt temperature variation and density change are also included. The density variation drives the flow of the outer fluid layer. The latent heat variation is modelled by a new relation, which matches experimental data better than previous models. A novel form of Stefan condition is used to determine the position of the melt front. This condition takes into account the latent heat variation, the energy required to create new surface and the kinetic energy of the displaced fluid layer. Results show that melting times can be significantly faster than predicted by previous theoretical models; for smaller particles, this can be around a factor 3. This is primarily due to the latent heat variation. The previously used fixed temperature boundary condition had two opposing effects on melt times: the implied infinite heat transfer led to faster melting but also artificially magnified the effect of kinetic energy, which slowed down the process. We conclude that any future models of nanoparticle melting must be based on the new Stefan condition and account for latent heat variation.







Similar content being viewed by others
References
Ahmad F, Pandey AK, Herzog AB, Rose JB, Gerba CP, Hashsham SA (2012) Environmental applications and potential health implications of quantum dots. J Nanoparticle Res 14:1038
Alexiades V, Solomon AD (1992) Mathematical modeling of melting and freezing processes. Hemisphere, Washington DC
Bachels T, Güntherodt H-J, Schäfer R (2000) Melting of isolated tin nanoparticles. Phys Rev Lett 85:1250–1253
Back JM (2014) Stefan Problems for Melting Nanoscaled Particles, PhD thesis, U. Queensland. http://eprints.qut.edu.au/79905/1/Julian_Back_Thesis. Accessed 12 Aug 2016
Back JM, McCue SW, Hsieh MH-N, Moroney TJ (2014) The effect of surface tension and kinetic undercooling on a radially-symmetric melting problem. Appl Math Comput 229:41–52
Back JM, McCue SW, Moroney TJ (2014) Including nonequilibrium interface kinetics in a continuum model for melting nanoscaled particles. Sci Rep 4:7066
Buffat P, Borel J-P (1976) Size effect on the melting temperature of gold particles. Phys Rev A 13:2287–2298
David TB, Lereah Y, Deutscher G, Kofman R, Cheyssac P (1995) Solid–liquid transition in ultra-fine lead particles. Philos Mag A 71:1135–1143
Delogu F (2005) Structural and energetic properties of unsupported Cu nanoparticles from room temperature to the melting point: molecular dynamics simulations. Phys Rev B 72:205418
Dieringer JA, McFarland AD, Shah NC, Stuart DA, Whitney AV, Yonzon CR, Young MA, Zhang X, Van Duyne RP (2006) Introductory lecture: surface enhanced Raman spectroscopy: new materials, concepts, characterization tools, and applications. Faraday Discuss 132:9–26
Dragomirescu FD, Eisenschmidt K, Rohde C, Weigand B (2016) Perturbation solutions for the finite radially symmetric Stefan problem. Int J Therm Sci 104:386–395
Ercolessi F, Andreoni W, Tosatti E (1991) Melting of small gold particles: mechanism and size effects. Phys Rev Lett 66:911–914
Fedorov AV, Shulgin AV (2011) Mathematical modeling of melting of nano-sized metal particles. Combust Explos Shock Waves 47(2):147–152
Florio BJ, Myers TG (2016) The melting and solidification of nanowires. J Nanoparticle Res 18(6):1–12
Font F, Myers TG (2013) Spherically symmetric nanoparticle melting with a variable phase change temperature. J Nanoparticle Res 15:2086
Font F, Myers TG, Mitchell SL (2014) A mathematical model for nanoparticle melting with density change. Microfluid Nanofluidics 18:233–243
Garg S, Bansal R, Ghosh C (1993) Thermal physics. Tata McGraw-Hill Education, New Delhi
Gröhn F, Kim G, Bauer BJ, Amis EJ (2001) Nanoparticle formation within dendrimer-containing polymer networks: route to new organic-inorganic hybrid materials. Macromolecules 34:2179–2185
Jiang H, Moon K-S, Dong H, Hua F, Wong C (2006) Size-dependent melting properties of tin nanoparticles. Chem Phys Lett 429:492–496
Jou D, Casas-Vázquez J, Lebon G (1996) Extended irreversible thermodynamics. Springer, Berlin
Lai S, Guo J, Petrova V, Ramanath G, Allen L (1996) Size-dependent melting properties of small tin particles: nanocalorimetric measurements. Phys Rev Lett 77:99–102
Latent Heat of Melting of some common Materials. http://www.engineeringtoolbox.com/latent-heat-melting-solids-d_96.html. Accessed 19 Nov 2015
Lim HS, Ong CK, Ercolessi F (1993) Surface effects in vibrational and melting properties of Pb clusters. Zeitschrift für Phys D Atoms Mol Clust 26:45–47
McCue SW, Wu B, Hill JM (2009) Micro/nanoparticle melting with spherical symmetry and surface tension. IMA J Appl Math 74:439–457
Mornet S, Vasseur S, Grasset F, Duguet E (2004) Magnetic nanoparticle design for medical diagnosis and therapy. J Mater Chem 14:2161
Myers TG, Mitchell SL, Font F (2012) Energy conservation in the one-phase supercooled Stefan problem. Int Commun Heat Mass Transf 39(10):1522–1525
Myers TG, MacDevette MM, Font F, Cregan V (2014) Continuum mathematics at the nanoscale. J Math Ind 4(1):1–13
Myers TG (2016) Mathematical modelling of phase change at the nanoscale. Int Commun Heat Mass Transf 76:59–62
Salata O (2004) Applications of nanoparticles in biology and medicine. J Nanobiotechnol 2:3
Sharafat S, Ghoniem N (2000) Summary of thermo-physical properties of Sn, and compounds of Sn-H Sn-O, Sn-C, Sn-Li, Sn-Si Comp. Prop. Sn, Sn-Li, Pb-Li, Rep. UCLA-UCMEP-00-31, University of California, Los Angeles
Shin J-H, Deinert MR (2014) A model for the latent heat of melting in free standing metal nanoparticles. J Chem Phys 140:164707
Sun J, Simon S (2007) The melting behavior of aluminum nanoparticles. Thermochim Acta 463:32–40
Tanabe K (2007) Optical radiation efficiencies of metal nanoparticles for optoelectronic applications. Mater Lett 61:4573–4575
Thermal Conductivity of Materials and Gases. http://www.engineeringtoolbox.com/thermal-conductivity-d_429.html. Accessed 19 Nov 2015
Tolman RC (1949) The effect of droplet size on surface tension. J Chem Phys 17:333
Wu B, Tillman P, McCue SW, Hill JM (2009) Nanoparticle melting as a Stefan moving boundary problem. J Nanosci Nanotechnol 9(2):885–888
Wu B, McCue SW, Tillman P, Hill JM (2009) Single phase limit for melting nanoparticles. Appl Math Model 33(5):2349–2367
Xiong S, Qi W, Cheng Y, Huang B, Wang M, Li Y (2011) Universal relation for size dependent thermodynamic properties of metallic nanoparticles. Phys Chem Chem Phys 13:10652–10660
Yih TC, Al-Fandi M (2006) Engineered nanoparticles as precise drug delivery systems. J Cell Biochem 97:1184–1190
Zhang M, Efremov M, Schiettekatte F, Olson E, Kwan A, Lai S, Wisleder T, Greene J, Allen L (2000) Size-dependent melting point depression of nanostructures: nanocalorimetric measurements. Phys Rev B 62:10548–10557
Zhang Y, Zhang HL, Wu JH, Wang XT (2011) Enhanced thermal conductivity in copper matrix composites reinforced with titanium-coated diamond particles. Scr Mater 65(12):1097–1100
Acknowledgments
The authors acknowledge that the research leading to these results has received funding from ‘la Caixa’ Foundation. TM acknowledges financial support from the Ministerio de Ciencia e Innovación grant MTM2014-56218.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ribera, H., Myers, T.G. A mathematical model for nanoparticle melting with size-dependent latent heat and melt temperature. Microfluid Nanofluid 20, 147 (2016). https://doi.org/10.1007/s10404-016-1810-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10404-016-1810-6