Abstract
The present study aimed to determine the clinical presentation, the multimodal interdisciplinary treatment strategies and outcome of posterior fossa arterio-venous malformations (AVMs) in our neurovascular centre. Fifty-three patients with a posterior fossa AVM were seen between 1998 and 2012 and analysed retrospectively. Patients were either managed conservatively or treated with endovascular, microsurgical or radiosurgical procedures or in combination. Thirty-nine patients (74 %) presented with intracranial haemorrhage and 14 patients (26 %) with unspecific symptoms. In 22 cases with haemorrhage (56 %), an intracerebellar haematoma was found, whereas 17 patients (44 %) suffered from subarachnoid haemorrhage. AVMs were located in the cerebellum in 44 patients (83 %), in the brainstem in four patients (7.5 %) and the cerebello-pontine angle in another four individuals (7.5 %). Forty-two patients (79 %) were treated either by emboliziation (n = 12, 29 %), surgical resection (n = 16, 38 %), surgical resection with preoperative embolization (n = 12, 29 %) or radiotherapy alone (n = 2, 4 %). A total of eleven patients did not receive any treatment (21 %). Both, morbidity and mortality related to treatment were 12 %, whereas overall morbidity and mortality was 26 and 15 %, respectively. Complete AVM elimination was achieved in 81 % of the treated lesions. A multimodal treatment sequence nowadays represents the gold standard for posterior fossa AVMs. Patients are at high risk for morbidity and mortality, due to the impact of haemorrhage and treatment. Therefore, treatment has to be thoroughly indicated, especially for those patients without bleeding. The initial neurological condition seems to be crucial in terms of clinical outcome.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Although cerebral arterio-venous malformations (AVMs) belong to the most frequent cerebral vascular malformations, AVMs of the posterior fossa are relatively rare comprising only between 10 and 15 % [1, 8]. Based on their location near to important neurovascular structures such as the brainstem and the cranial nerves, these patients are at an increased risk for severe morbidity or even mortality due to bleeding of the AVM or additionally caused by the treatment.
Over the last decades, only a small number of larger series with posterior fossa AVMs have been published, including historical surgical series [2, 22, 7] or series treated inconsistently by different treatment modalities such as surgery, radiotherapy or more recently by endovascular therapy [20, 12, 19, 3]. However, treatment methods have changed over the last decades, especially due to technical developments of endovascular procedures, as well as due to more insights about the natural history of AVMs and their risk for bleeding.
The aim of our study was to analyse a sizable series of patients with posterior fossa AVMs, consistently treated in our neurovascular centre by an interdisciplinary team with individualized multimodal treatment concepts.
Methods
Patients
Between 1998 and 2012, a total of 53 patients with AVMs of the posterior fossa were seen in our neurovascular centre. There were 20 males and 33 females with a mean age of 43.8 years (age ranging between 2 and 87 years). Basic demographic data, clinical presentation and outcome were evaluated. Initial clinical presentation on admission was graded according to the Glasgow Coma Scale (GCS). Thirty-nine patients (74 %) presented with intracranial haemorrhage, either from the AVM or an AVM-associated aneurysm. Haemorrhage was defined as the acute onset of clinical and neurological symptoms as well as detection of blood revealed by computed tomography (CT) or magnetic resonance imaging (MRI). Patients with unspecific symptoms leading to the diagnosis of the posterior fossa AVM or incidental AVMs were assigned to the non-haemorrhagic group (n = 14, 26 %).
Neuroimaging studies
Patients were examined with MRI and cerebral digital subtraction angiography (DSA) before treatment. MRI was performed to determine the exact location, eloquence, size and configuration of the AVM. AVMs located in the deep cerebellar nuclei, cerebellar peduncles or brainstem were considered eloquent [24].
Cerebral DSA was performed to assess the angiographic characteristics of the AVM with special consideration of number and type of arterial feeders (e.g. en passant) and absence or presence of associated aneurysms. Aneurysms were divided into feeding artery aneurysms (FAA), intranidal and venous aneurysms.
Characteristics of the AVM nidus (plexiform, high-flow/fistulous) and type of venous drainage were also determined by this procedure. Superficial venous drainage was only assumed in those cases with direct drainage into the straight and transverse sinus [24].
Based on the neuroradiological findings, AVMs were classified according to the original 5-tier and the revised 3-tier Spetzler-Martin grading system (SMG) [24, 25].
Decision-making process and treatment indication
Treatment options were evaluated in an interdisciplinary approach in our neurovascular centre by experienced neurosurgeons, neuroradiologists and, if considered necessary, radiosurgeons. Criteria for treatment decision were clinical and neurological symptoms, type of presentation with or without haemorrhage, with special consideration on the potential source of bleeding (e.g. presence of flow-associated aneurysms) and SMG. For patients, who presented with non-haemorrhagic symptoms, the decision was based additionally on the estimated haemorrhage risk as well as on patients’ will.
With exception of emergencies showing signs of decerebration (n = 4), all patients with acute bleeding were admitted to treatment in the subacute stage, giving the opportunity of clinical stabilization. Mean time interval between bleeding onset and AVM treatment was 11 days, ranging from 1 to 21 days. The primary aim was complete AVM elimination by endovascular procedures, microneurosurgery or in a combined approach with one or repetitive preoperative interventional procedures in order to reduce both the flow in the AVM feeding arteries and the size of the AVM nidus. In cases, in which only incomplete AVM occlusion was assumed, or in those cases with significantly increased surgical and endovascular risk, radiosurgery or conservative therapy was recommended.
The treatment of associated aneurysms was mainly addressed to FAA. Thus, nidal and venous aneurysms were incorporated into the treatment strategy of the AVM itself, due to the fact that bleeding from these lesions could not be distinguished conclusively from rupture of the AVM nidus. In case of bleeding from a ruptured FAA, treatment was primarily directed towards the symptomatic aneurysm by either endovascular or microneurosurgical procedures, alone, or in combination with the AVM treatment. FAAs of patients with haemorrhage from ruptured AVM as well as patients without bleeding were treated based on the estimated risk from both FAA and AVM.
Follow-up
Patients were followed up clinically and radiologically. Mean follow-up was 19 months, ranging from 2 to 96 months. Clinical outcome was assessed using the modified Rankin scale (mRS) in order to achieve a good functional grading. Patients’ outcome was defined as favourable, if the mRS was less than three and unfavourable if equal or higher than three. Most treated patients underwent follow-up DSA to assess the obliteration rate of the AVM or the associated aneurysm.
Statistical analysis
To assess the influence of angiographic characteristics on haemorrhagic presentation, odds ratio (OR) and 95 % confidence interval (CI) were calculated using a univariate analysis. A p value of <0.05 was considered as statistically significant.
Results
Clinical presentation
On admission, 39 patients presented with haemorrhage (74 %) and 11 individuals with AVM-related unspecific symptoms. These symptoms were vertigo (n = 6), temporary sensory (n = 2) and cranial nerve (n = 2) disturbances. One patient suffered from a repetitive syncope. In three cases, AVMs were considered incidental with no symptoms. Epilepsy as a primary symptom was not observed. Seizures following a haemorrhagic event were seen in four patients.
A total of 33 of 39 patients (85 %) presenting with haemorrhage were referred to our neurovascular centre in the acute stage, whereas six patients (15 %) were admitted in a delayed stage between 2 months and 3 years after bleeding (mean 18 months). Among these, 20 patients (51.5 %) were in a good clinical condition with a GCS > 13, including all six patients, presenting in a subacute state. Six patients (15.5 %) presented in an intermediate clinical stage according to a GCS 8–12. Thirteen patients (33 %) were in poor clinical condition with unconsciousness and a GCS of <7. Among these, nine individuals were admitted with a GCS = 3. In four comatose patients, signs of decerebration with unilateral or bilateral dilated and non-reactive pupils were present.
Acute hydrocephalus was found in 22 individuals (56 %). All patients except for one in poor and intermediate clinical condition displayed with concomitant hydrocephalus, compared with only four patients in good clinical stage. Five patients suffered from rupture of a FAA, representing 13 % of all bleedings. The clinical presentation is summarized in Table 1.
Radiological findings
In 22 out of 39 patients (56 %) with haemorrhage, a cerebellar haematoma was found, which was located in the cerebellar hemispheres in 12 cases (55 %) and in the vermis in ten cases (45 %). Mean maximal haematoma diameter was 31.5 ± 13.0 mm. Seventeen patients (44 %) suffered from subarachnoid haemorrhage (SAH) of the basal and suboccipital cisterns or the fourth ventricle.
AVMs were located in the cerebellum in 44 patients (83 %), in the brainstem in four patients (7.5 %) and in the cerebello-pontine angle in another four individuals (7.5 %). Among patients with cerebellar AVMs, 13 (30 %) were detected in the vermis and 31 (70 %) in the cerebellar hemispheres, including the tonsils and the cerebellar peduncles. In one individual, the AVM occupied nearly the complete posterior fossa. Eloquent location, as defined above, was found in 29 patients (55 %).
Complete angiographic data were available in 49 patients (92 %). Those four patients with signs of decerebration underwent immediate surgery without preoperative DSA but received CT angiography prior to surgery. According to the SMG, AVM size was graded as small in 29 patients (59 %), intermediate in 17 patients (35 %) and large in three patients (6 %). En passant arterial supply was found in 11 cases (22 %). Twelve AVMs (24 %) had only one single feeder and 15 cases (31 %) two feeders. More than two arterial feeders were found in 22 cases (45 %) and bilateral supply in 11 patients (22 %). A high-flow/fistulous nidus component was present in nine cases (18 %).
Deep venous drainage was identified in 19 patients (39 %), whereas superficial drainage was seen in the majority of cases (n = 30, 61 %). Venous aneurysms or ectasias were seen in four patients (8 %).
A total of 17 FAAs were found in 12 of 49 patients (24 %), with four patients carrying multiple aneurysms. In two patients, an additional intranidal aneurysm was found and one patient harboured a single intranidal aneurysm only. Most frequent location for FAA was the posterior inferior cerebellar artery (PICA) in eight cases (67 %). Rupture from FAA was found in five patients. Thus, 40 % of patients with FAA suffered from aneurysm rupture.
Whereas the AVM size (<3 cm) was associated with a statistical higher risk for haemorrhage (p = 0.04), no other angiographic features showed an influence on clinical presentation or haemorrhage. An overview of the patients’ radiological characteristics in relation to clinical presentation (haemorrhagic vs. non-haemorrrhagic) is shown in Tables 2 and 3.
Treatment
Forty-two patients (79 %) were treated by either embolization, surgical resection, with or without preoperative embolization or radiosurgery. Among this group, 31 patients initially presented with haemorrhage and 11 patients without.
Twelve patients received surgical resection with preoperative embolization. Mean time between endovascular treatment and surgical AVM excision in patients with haemorrhage (n = 8) was 5.6 days (range 2 to 16 days), compared with 14.5 days (range 1 to 32 days) for patients without haemorrhage (n = 4).
Sixteen patients underwent surgery alone without preoperative endovascular treatment, including those four cases with emergency surgery due to life-threatening haemorrhagic presentation.
Endovascular embolization alone was performed in 12 patients, including three patients with sole coiling of the associated aneurysms and without further treatment of the AVM itself. Two of these patients suffered from rupture of the aneurysm. Reasons for leaving the AVM untreated were a poor posthaemorrhagic condition (mRS = 4) without recovery after 19 months in one patient and a significantly increased risk for treatment in the other two cases.
Two patients without bleeding were submitted to stereotactic radiosurgery, because of an increased treatment related risk for both endovascular therapy and surgery.
Eleven patients did not receive any treatment. Three patients died from primarily devastating bleeding within 2 days and 1 week, respectively. Four patients refused therapy, although treatment was recommended. Among these, three patients initially presented with haemorrhage. In four cases, the AVM was classified as untreatable and observation only was recommended, with two patients presenting with haemorrhage and two patients without. An overview of all treatment modalities is shown in Table 4.
Complications
In 12 cases (29 %), major therapy-related complications occurred. Three patients with haemorrhage sustained rupture of the AVM during endovascular embolization with a need of urgent decompressive suboccipital craniectomy, evacuation of the haematoma and excision of the remaining AVM. Among these patients, one patient died and one patient made a good recovery (mRS = 2). The third patient, initially presenting in a poor clinical state (GCS < 7), did not recover and remained in an unfavourable condition (mRS = 4). One patient with complete endovascular obliteration developed progressive cerebellar swelling 2 days after embolization and underwent secondary suboccipital decompression. This patient made an excellent recovery (mRS = 1). Another patient in intermediate condition after initial haemorrhage suffered from midbrain ischemia after embolization and remained in highly disabled clinical condition (mRS = 5) at the time of the last follow-up.
Three individuals suffered from rebleeding after primary surgery. Cause for postoperative haemorrhage in one case was subjacent coagulopathy, due to known hepatic dysfunction. The second patient developed perioperative coagulopathy. Due to significant clinical deterioration, both patients died 15 and 16 days after surgery. The third patient, harbouring a large symptomatic AVM of the cerebellar hemisphere without haemorrhage, died 2 days after surgery because of incomplete subtotal nidus resection and fatal rebleeding. One patient sustained a cardiac arrest with a need of resuscitation 2 days after treatment and did not recover (mRS = 5). Two patients with haemorrhage and good initial clinical state suffered from uni- and bilateral PICA infarction after AVM resection, leading to a mRS 4 and mRS 5 on discharge. One patient with haemorrhage and good initial clinical state showed severe neurological deficits after uneventful surgery and died 2 months after surgery during rehabilitation, due to a pulmonary embolism. Thus, treatment related morbidity was 12 % (n = 5) and mortality 12 % (n = 5), respectively.
Clinical outcome
Of those patients with either treatment of the AVM or an associated aneurysm (n = 42), 25 patients (60 %) were in good or favourable state at the last follow-up, according to a mRS = 2 or less. Among this group, 16 patients showed an excellent recovery (mRS 0–1, 38 %) including five patients initially presenting in a poor and one patient in an intermediate state.
Thirteen patients (31 %) remained in a poor clinical condition with a mRS ≥ 3 in eight cases due to a poor initial state (GCS ≤ 7). However five patients (12 %) sustained worsening of their clinical condition after treatment, due to major complications, as described above.
Of all 53 patients with an AVM of the posterior fossa, 14 patients remained in poor clinical state with severe neurological deficits and eight patients died, accounting an overall morbidity of 26 % and mortality of 15 %.
Radiological outcome
Among the 42 treated patients, 37 patients received radiological follow-up evaluation with DSA. Complete AVM elimination was achieved in 30 individuals (81 %). In four cases, the AVM was incompletely treated. Two patients with subtotal AVM embolization (>95 % embolization rate) refused further treatment, although radiosurgery or surgery was recommended. One patient with initial bleeding remained in poor clinical condition (mRS = 5) after partial embolization (50 % embolization rate), not capable for further surgery. Another patient with haemorrhage, treated with endovascular embolization, followed by surgical subtotal nidus excision, received a postoperative stereotactic radiosurgery.
In all cases of SAH from rupture of an associated aneurysm (n = 5), the bleeding source was treated completely by coiling of the aneurysm or occluding the parent vessel. Three patients received additional treatment of the AVM, whereas two patients did not undergo AVM treatment, as mentioned above. Overall, 10 out of 12 patients with associated aneurysms received additional aneurysm obliteration. Radiological outcome details are shown in Table 5.
Illustrative cases
Case 1
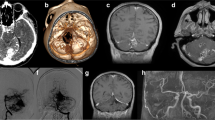
This 16-year-old girl was admitted in comatose state with a GCS of 3 without signs of decerebration. Cranial CT revealed a large left cerebellar haemorrhage with a maximum diameter of 45 mm and consecutive hydrocephalus. Initially, a ventricular drainage was inserted and the patient was transferred to our hospital. Subsequent MRI and DSA demonstrated a left para-vermal cerebellar AVM with a diffuse nidus configuration and a size of 20 mm. Arterial feeders arose from the left superior cerebellar artery (SCA) and the left anterior inferior cerebellar artery (AICA). No associated aneurysms were detectable, and venous drainage was directly into the left sigmoid sinus. According to the 3-tier SMG, the AVM was classified as grade II. A multimodal treatment strategy in the subacute stage was decided, with initial endovascular intervention, followed by surgical excision, if necessary. Partial embolization was performed 12 days after bleeding via the left SCA, whereas further embolization via the left AICA was not feasible, because of diffuse and multiple en passant feeders. Thus, the patient underwent uneventful microsurgical resection 2 days after embolization, via a left suboccipital trans-cortical approach in sitting position. Postsurgical angiography demonstrated complete AVM elimination. Postoperatively, the patient recovered continuously with broadly regained daily independence 3 month after rehabilitation, moderate gait-ataxia and diplopia (mRS = 2) (Fig. 1).
Case 1; a Initial CT scan showing a large right cerebellar haemorrhage with compression of the fourth ventricle. b–d Preinterventional MRI and cerebral DSA revealing a left 20 mm cerebellar para-vermal AVM with arterial supply from the left SCA and AICA, draining into the left sigmoid sinus. e After partial embolization, DSA demonstrates a significant reduction of flow and nidus size, but a still present early draining vein (arrow). f–h Postoperative CT scan after surgical resection of the residual nidus and controlangiography, verifying complete AVM elimination; 219 × 124 mm (300 × 300 DPI)
Case 2
This 45-year-old neurologically intact female patient complained of chronic headache, ear and neck pains, combined with intermittent nausea. After an orthostatic dysregulation, cranial MRI was performed, showing an AVM of 35 mm in the left cerebellar hemisphere. DSA confirmed a plexiforme AVM of the left para-mediane cerebellar hemisphere. This AVM SMG III [25] was supplied by feeders from the left SCA, AICA and PICA, with venous drainage into the left transverse and left inferior petrous sinus. Additionally, an associated distal bilobar saccular PICA aneurysm was present, with a maximum diameter of 8 mm. The situation was discussed extensively, and recommendation was given towards treatment, which was in accordance with the patient’s strong will for treatment.
Endovascular treatment led to complete coil embolization of the aneurysm with preservation of the parent PICA and in the same setting to nearly 50 % occlusion of the AVM nidus, thus, obliterating the caudal portions of the AVM. Further embolization was judged as not possible due to en passant supply from the AICA and SCA. The patient tolerated this intervention without any problems and subsequent microsurgical resection was accomplished 16 days later. Complete AVM resection was proven by postoperative angiography. At the last follow-up 5 years after treatment, she showed no deficits and a full activity at daily life and work (Fig. 2).
Case 2; a, b Initial angiogram in a patient without bleeding and unspecific symptoms showing a plexiforme AVM of the left paremediane hemisphere, feed by branches of the left SCA, AICA and PICA. An additional FAA of the left PICA was found (arrow). c–e Postinterventional angiogram and MRI after partial AVM embolization and coil occlusion of the FAA (arrow). f–h Postoperative DSA and MRI after subsequent surgery, showing complete AVM resection; 173 × 91 mm (300 × 300 DPI)
Discussion
Clinical presentation
The natural history of posterior fossa AVMs is still poorly understood. However, if these lesions become symptomatic, their treatment remains challenging despite significant technological developments over the last two decades. Due to the specific anatomical conditions of the posterior fossa with the presence of highly eloquent neurovascular structures as the cranial nerves and the brainstem, these patients are at an increased risk for severe neurological symptoms and even mortality.
In contrast to supra-tentorial lesions, haemorrhage and subsequent hydrocephalus was the leading clinical presentation for posterior fossa AVMs in our series, which was also reported by others [11, 13, 21]. On admission, 74 % of our patients presented with haemorrhage, most commonly as an intraparenchymal haematoma (56 %). This observation is different to previous reports, emphasizing subarachnoid haemorrhage as the most common presentation in 61–90 % [9, 10, 26]. Haemorrhage and a resulting tight posterior fossa [28, 4] with consecutive brainstem compression and hydrocephalus might be one reason why nearly 50 % of our collective presented in a poor or intermediate neurological condition with impaired consciousness and severe neurological deficits. Similar observations were made by the Toronto Brain Vascular Malformation Study Group [3], accounting a bleeding rate of 62 % and neurological deficits in 48 %.
Interestingly, in our series, only a small AVM nidus size correlated with an increased risk for bleeding, which has been previously reported by others [23, 15]. Despite these findings, we could not identify further potential angiographic features to estimate future haemorrhage. Although we found FAA in 29 % of patients presenting with haemorrhage compared with only 14 % in the non-haemorrhagic group, this finding failed to show statistical difference.
Treatment indication and modalities
With the ongoing development in treatment techniques over the last years, an interdisciplinary board for treatment indication and decision processes nowadays is mandatory. The risks of treatment have to be balanced with the natural history and the risk of bleeding. Treatment appears justified for symptomatic patients with bleeding and potentially treatable lesions [16]. However, therapy indications have to be thoroughly reviewed in patients with non-specific symptoms or incidental AVMs. Reflecting a more conservative approach not only for non-symptomatic posterior fossa AVMs, the number of untreated posterior fossa AVMs constantly grew from about 6–9 % in earlier reports [2, 7] up to 18–37 % in more recent publications [3, 26], with 21 % of untreated lesions in our cohort.
Taking three historical surgical series together [2, 22, 26], only six of 65 treated patients (9 %) underwent an endovascular intervention of their AVM as a single or a combined procedure, thus, endovascular approaches displayed only a marginal role. Kelly and co-workers [12] recently presented a larger series of multimodally treated AVMs of the posterior fossa, reflecting a more interdisciplinary treatment strategy. The growing role of endovascular interventions can also be pointed out by other publications, emphasizing endovascular embolization as an important additional treatment modality, either alone or as part of a multimodal treatment concept for the majority of patients [3, 18].
We only considered radiosurgery alone in two carefully selected cases, thus leading to a 100 % obliteration rate within 3 years. Both individuals harboured a small AVM involving the brain stem. There are several studies dealing with radiosurgery for deep-seated AVMs of the posterior fossa. Nagy et al. [19] presented their large number of 160 pons/medulla sited AVMs with considerable obliteration rates up to 70 % after 4 years in those malformations, smaller than 4 cm3. Adverse radiation effects and clinical outcome were though correlated with AVM size, leading to 50 % permanent morbidity in larger AVMs. In contrast, Kelly et al. [12] reported only 29 % occlusion rates after 3 years, including only SMG III–V and thus certainly larger AVMs. They finally recommended radiosurgery only for brain stem AVMs, whereas all other lesions of the posterior fossa should undergo an interdisciplinary interventional approach.
Clinical outcome
Excellent and good clinical results were reported in up to 67–81 % of treated patients even for earlier series [2, 3, 7, 12]. These results are slightly different from our findings with a favourable outcome in only 60 %. Despite improvement of surgical and endovascular techniques, different patient characteristics and a high number of patients in a poor clinical condition in our cohort might explain this discrepancy. There are numerous reports indicating the initial neurological state as a major predictive factor for clinical outcome for patients with spontaneous posterior fossa bleedings as well as for bleedings due to cerebrovascular malformations [4–6, 27, 29]. Although the impact of haemorrhage from a ruptured AVM or spontaneous cerebellar haemorrhage is different, the local effects with brainstem compression and hydrocephalus are similar, leading to a high number of initially poor-grade patients and thereby to a poor clinical outcome in 31 % in our cohort. Other authors suggested that patients with cerebellar haemorrhage and a comatose state should not be considered for intensive therapy [14]. However, we observed 11/19 patients (58 %) with initially poor or intermediate clinical state improving in clinical condition and six patients (32 %) even making an excellent recovery. Therefore, intensive therapy in our opinion might be justified for these patients. Da Costa et al. [3] reported similar results with 66 % of patients improving in clinical state at the last follow-up. An overview of some series and their results is presented in Table 6.
Despite successful treatment results in most cases, utmost attention has to be drawn to those patients, suffering from severe treatment complications. Major treatment related complications causing permanent and severe neurological disability or death occurred in ten patients (24 %); among these, six patients initially presenting in a good neurological condition. Although the rate of treatment complications in literature is comparable to our results, ranging between 17 and 20 % [2, 12, 26, 22], this issue must be stressed in particular, since data of the ARUBA trial have been recently published [17].
These circumstances underline previous assumption, that “in view of the rarity of these lesions to obtain the optimal outcome, these patients should be treated in a specialized neurovascular centre” [26].
Conclusion
Posterior fossa AVMs are complex and rare lesions, requiring thorough treatment strategies by an experienced interdisciplinary neurovascular treatment board. A multimodal treatment sequence with endovascular embolization, surgical resection, radiotherapy and conservative treatment in selected cases nowadays represents the gold standard for these lesions. The initial neurological condition seems to be crucial in terms of clinical outcome and patients are at high risk for morbidity and mortality, caused by both the impact of haemorrhage and treatment. Therefore, treatment indication in particular for those patients without bleeding remains difficult and needs further to be clarified.
References
Al-Shahi R, Warlow C (2001) A systematic review of the frequency and prognosis of arteriovenous malformations of the brain in adults. Brain: J Neurol 124(Pt 10):1900–1926
Batjer H, Samson D (1986) Arteriovenous malformations of the posterior fossa. Clinical presentation, diagnostic evaluation, and surgical treatment. J Neurosurg 64(6):849–856. doi:10.3171/jns.1986.64.6.0849
da Costa L, Thines L, Dehdashti AR, Wallace MC, Willinsky RA, Tymianski M, Schwartz ML, ter Brugge KG (2009) Management and clinical outcome of posterior fossa arteriovenous malformations: report on a single-centre 15-year experience. J Neurol, Neurosurg, Psychiatry 80(4):376–379. doi:10.1136/jnnp.2008.152710
Dammann P, Asgari S, Bassiouni H, Gasser T, Panagiotopoulos V, Gizewski ER, Stolke D, Sure U, Sandalcioglu IE (2011) Spontaneous cerebellar hemorrhage–experience with 57 surgically treated patients and review of the literature. Neurosurg Rev 34(1):77–86. doi:10.1007/s10143-010-0279-0
Dolderer S, Kallenberg K, Aschoff A, Schwab S, Schwarz S (2004) Long-term outcome after spontaneous cerebellar haemorrhage. Eur Neurol 52(2):112–119. doi:10.1159/000080268
Donauer E, Loew F, Faubert C, Alesch F, Schaan M (1994) Prognostic factors in the treatment of cerebellar haemorrhage. Acta Neurochir 131(1–2):59–66
Drake CG, Friedman AH, Peerless SJ (1986) Posterior fossa arteriovenous malformations. J Neurosurg 64(1):1–10. doi:10.3171/jns.1986.64.1.0001
Fleetwood IG, Steinberg GK (2002) Arteriovenous malformations. Lancet 359(9309):863–873. doi:10.1016/S0140-6736(02)07946-1
Fults D, Kelly DL Jr (1984) Natural history of arteriovenous malformations of the brain: a clinical study. Neurosurgery 15(5):658–662
Hamilton MG, Spetzler RF (1994) The prospective application of a grading system for arteriovenous malformations. Neurosurgery 34(1):2–6, discussion 6–7
Kelly D. Flemming RDBJ (2011) Youmans Neurological Surgery, vol 4. 6th edn. Saunders
Kelly ME, Guzman R, Sinclair J, Bell-Stephens TE, Bower R, Hamilton S, Marks MP, Do HM, Chang SD, Adler JR, Levy RP, Steinberg GK (2008) Multimodality treatment of posterior fossa arteriovenous malformations. J Neurosurg 108(6):1152–1161. doi:10.3171/JNS/2008/108/6/1152
Khaw AV, Mohr JP, Sciacca RR, Schumacher HC, Hartmann A, Pile-Spellman J, Mast H, Stapf C (2004) Association of infratentorial brain arteriovenous malformations with hemorrhage at initial presentation. Stroke: J Cerebral Circulation 35(3):660–663. doi:10.1161/01.STR.0000117093.59726.F9
Kobayashi S, Sato A, Kageyama Y, Nakamura H, Watanabe Y, Yamaura A (1994) Treatment of hypertensive cerebellar hemorrhage—surgical or conservative management? Neurosurgery 34(2):246–250, discussion 250–241
Langer DJ, Lasner TM, Hurst RW, Flamm ES, Zager EL, King JT Jr (1998) Hypertension, small size, and deep venous drainage are associated with risk of hemorrhagic presentation of cerebral arteriovenous malformations. Neurosurgery 42(3):481–486, discussion 487–489
Mohr JP, Moskowitz AJ, Parides M, Stapf C, Young WL (2012) Hull down on the horizon: a randomized trial of unruptured brain arteriovenous malformations (ARUBA) trial. Stroke: J Cerebral Circulation 43(7):1744–1745. doi:10.1161/STROKEAHA.112.653584
Mohr JP, Parides MK, Stapf C, Moquete E, Moy CS, Overbey JR, Salman RA-S, Vicaut E, Young WL, Houdart E, Cordonnier C, Stefani MA, Hartmann A, von Kummer Rd, Biondi A, Berkefeld J, Klijn CJM, Harkness K, Libman R, Barreau X, Moskowitz AJ (2013) Medical management with or without interventional therapy for unruptured brain arteriovenous malformations (ARUBA): a multicentre, non-blinded, randomised trial. The Lancet
Mpotsaris A, Loehr C, Harati A, Lohmann F, Puchner M, Weber W (2010) Interdisciplinary clinical management of high grade arteriovenous malformations and ruptured flow-related aneurysms in the posterior fossa. Interv Neuroradiol: J of Peritherapeutic neuroradiology, Surgical Procedures and related Neurosciences 16(4):400–408
Nagy G, Major O, Rowe JG, Radatz MW, Hodgson TJ, Coley SC, Kemeny AA (2012) Stereotactic radiosurgery for arteriovenous malformations located in deep critical regions. Neurosurgery 70(6):1458–1469. doi:10.1227/NEU.0b013e318246a4d0, discussion 1469–1471
Pollock BE, Gorman DA, Brown PD (2004) Radiosurgery for arteriovenous malformations of the basal ganglia, thalamus, and brainstem. J Neurosurg 100(2):210–214. doi:10.3171/jns.2004.100.2.0210
Schmidt NO, Reitz M, Raimund F, Treszl A, Grzyska U, Westphal M, Regelsberger J (2011) Clinical relevance of associated aneurysms with arteriovenous malformations of the posterior fossa. Acta Neurochir Suppl 112:131–135. doi:10.1007/978-3-7091-0661-7_23
Solomon RA, Stein BM (1986) Management of arteriovenous malformations of the brain stem. J Neurosurg 64(6):857–864. doi:10.3171/jns.1986.64.6.0857
Spetzler RF, Hargraves RW, McCormick PW, Zabramski JM, Flom RA, Zimmerman RS (1992) Relationship of perfusion pressure and size to risk of hemorrhage from arteriovenous malformations. J Neurosurg 76(6):918–923. doi:10.3171/jns.1992.76.6.0918
Spetzler RF, Martin NA (1986) A proposed grading system for arteriovenous malformations. J Neurosurg 65(4):476–483. doi:10.3171/jns.1986.65.4.0476
Spetzler RF, Ponce FA (2011) A 3-tier classification of cerebral arteriovenous malformations. Clinical Article J Neurosurg 114(3):842–849. doi:10.3171/2010.8.JNS10663
Symon L, Tacconi L, Mendoza N, Nakaji P (1995) Arteriovenous malformations of the posterior fossa: a report on 28 cases and review of the literature. Br J Neurosurg 9(6):721–732
van Loon J, Van Calenbergh F, Goffin J, Plets C (1993) Controversies in the management of spontaneous cerebellar haemorrhage. A consecutive series of 49 cases and review of the literature. Acta Neurochir 122(3–4):187–193
Weisberg LA (1986) Acute cerebellar hemorrhage and CT evidence of tight posterior fossa. Neurology 36(6):858–860
Yilmaz A, Musluman AM, Kanat A, Cavusoglu H, Terzi Y, Aydin Y (2011) The correlation between hematoma volume and outcome in ruptured posterior fossa arteriovenous malformations indicates the importance of surgical evacuation of hematomas. Turk Neurosurg 21(2):152–159. doi:10.5137/1019-5149.JTN.3401-10.0
Author information
Authors and Affiliations
Corresponding author
Additional information
Comments
Isabel Cuervo-Arango Herreros, Asturias, Spain and Álvaro Campero, Tucumán, Argentina
In this interesting and well-written article, Klaus-Peter Stein et al. report a retrospective study about clinical presentation, multimodal interdisciplinary treatment strategies and outcome of posterior fossa arterio-venous malformations in 53 patients in the University Hospital Essen, Germany, between 1998 and 2012, as well as a thorough review of the related literature. They make the difference in terms of clinical presentation between haemorrhagic and non-haemorrhagic groups, as well as neurological symptoms, which will determine the treatment planning. In addition, the authors evaluate the treatment options in an interdisciplinary approach, what we believe is the first step that must be taken.
An important finding in their series is that 56 % of patients had intracranial haematoma in the haemorrhagic group, against the literature where 60–90 % had subarachnoid haemorrhage. We think that further research should support this finding.
Furthermore, the description of radiologic findings as size, feeding artery aneurysms, high-flow/fistulous nidus and venous drainage, will help many neurovascular radiologist and surgeons to take the best therapeutic option either surgical, endovascular or combined in each case. However, only the AVM size (<3 cm) was associated with a statistical higher risk for haemorrhage in their series.
In the present study, there is no clear relationship between clinical-radiological findings and the treatment adopted, probably because of the wide variability of both, so we think that a classification and therapeutic protocol to posterior fossa AVMs needs further to be clarified, as the authors concluded.
Finally, we would like to remark, once again, the relevant role played by a multidisciplinary team for this pathology, and we congratulate the authors for the article and also their results.
Karl Schaller, Geneva, Switzerland
This is a comprehensive retrospective report by a group of experienced neurovascular surgeons and interventional neuroradiologists who are reporting on a large series (53 patients) who had been diagnosed for posterior fossa AVM. Three quarters of all patients were admitted following AVM rupture. Twenty percent of patients were not treated, and the vast majority underwent a combination of endovascular treatment and microsurgical removal, or embolization, or microsurgery alone, respectively. Two patients had radiosurgery. Among those 11 patients who were not treated, in four of them the AVM was judged untreatable.
In my opinion, this is an important clinical series of this distinct group of cranial AVMs—those located in the posterior fossa. I had wished the authors had highlighted a little more the potential relevance of intranidal aneurysms, which might be amenable to selective treatment in otherwise intractably complex AVMS. It is, however, always difficult to look into all these subtle angiographical details, especially when analysing a series of such a long study period.
Some of the conclusions do not come as surprise, notably that the initial neurological condition is predictive for outcome. It is clearly not advised to be more conservative in one’s attitude when it comes to the clinical judgement of posterior fossa AVMs: Especially in view of the ARUBA trial, neurologists who are seeing AVM-patients more and more frequently and even the fewer and fewer fully trained neurovascular surgeons may develop a more conservative attitude towards intracranial AVMs. With all the respect for the group who has contributed to the ARUBA trial, it is too early to derive to the conclusion that conservative therapy and surveillance would be less harmful than treatment in all cranial AVMs. There are by far too many substantial methodological flaws in the ARUBA study, and the observation period was far from being long enough to observe AVM ruptures further down the timeline.
With the present study regarding posterior fossa AVS the authors have demonstrated in my opinion, that due to their particular localization, these AVMs pose an important and maybe so far underrated threat to the patients. This is illustrated for example by the fact that three quarters of the patient presented with AVM rupture, which is a far higher rate than the nowadays presumed 30–50 % of supra-tentorial AVMs. In addition, posterior fossa haemorrhages frequently require extremely rapid surgical removal. I would thus distinguish posterior fossa AVMs from those localized supra-tentorally for very practical and logical reasons, and I would be very hesitant to apply a conservative attitude based on easy grounds such as the ARUBA study. As the authors have clearly pointed out: It is the initial clinical state, which is the most important determinant for long-term outcome. Should thus unruptured posterior fossa AVMS come to the attention of a dedicated neurovascular team, then elaborate high-resolution and selective angiography will become instrumental in decision-making, as additional venous anomalies, such as stenosis, or intranidal aneurysms may render certain AVMs of a particular risk for rupture. Then the indication for surgery should be made more deliberately than it seems presently to be the case within the somewhat unsettled neurovascular community following the insufficient ARUBA study.
The authors are to be commended for having highlighted with a diligently worked up series of posterior fossa AVMs, that such AVMs are a treatable disease, ideally when they come to our attention unruptured, and the surgeons, together with their closest partner from interventional radiology, should lead the discussion and not just following it, and we should guide the patients rather than leaving them in the hands of neurologists and epidemiologists who are at safe distance when it comes to taking concrete responsibilities in case of AVM rupture of a previously diagnosed unruptured one for which an observational attitude was taken.
Rights and permissions
About this article
Cite this article
Stein, KP., Wanke, I., Schlamann, M. et al. Posterior fossa arterio-venous malformations: current multimodal treatment strategies and results. Neurosurg Rev 37, 619–628 (2014). https://doi.org/10.1007/s10143-014-0551-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10143-014-0551-9