Abstract
Background
The outcome of stage III gastric cancer patients treated by D2 dissection followed by adjuvant chemotherapy with S-1 remains unsatisfactory. Moreover, some patients with a preoperative diagnosis of stage II/III turn out to be stage IV after surgical exploration, and a standard postoperative treatment for this population has not been established.
Methods
A feasibility study of postoperative S-1/cisplatin (CDDP) was performed with patients who underwent gastrectomy for what turned out to be a stage IV gastric cancer. The primary endpoint of the trial was the relative dose intensity during five courses of S-1/CDDP. Several criteria to skip, postpone, or reduce the dose had been predetermined.
Results
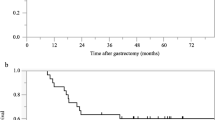
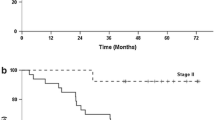
Between 2007 and 2009, 31 patients were accrued, including 19 patients who were positive for peritoneal washing cytology, 6 with peritoneal seeding, 5 with metastasis to the paraaortic nodes, and 4 with other distant metastases. Only 7 patients completed five cycles as planned (median, two cycles). The median relative dose intensities of S-1 and CDDP were 37% and 40%, respectively. Causes of treatment failure were failure to fulfill criteria for starting a new course within 5 weeks of the last administration of S-1 in 7, patient refusal in 6, disease recurrence/progression in 4, need to reduce dose by two levels in 4, and two successive skips of CDDP in 3 patients. The median progression-free survival time of all patients was 363 days.
Conclusions
Although promising in the neoadjuvant and advanced/metastatic setting, S-1/CDDP is too toxic as a postgastrectomy treatment for Japanese patients.
Article PDF
Similar content being viewed by others
References
Pisani P, Parkin D, Bray F, Ferlay J. Estimates of the worldwide mortality from 25 cancers in 1990. Int J Cancer 1999;83:18–29.
Macdonald J, Smalley S, Benedetti J, Hundahl SA, Estes NC, Stemmermann GN, et al. Chemoradiotherapy after surgery compared with surgery alone for adenocarcinoma of the stomach or gastroesophageal junction. N Engl J Med 2001;345:725–730.
Cunningham D, Allum W, Stenning S, Thompson JN, Van de Velde CJ, Nicolson M, et al. Perioperative chemotherapy versus surgery alone for resectable gastroesophageal cancer. N Engl J Med 2006;355:11–20.
Sakuramoto S, Sasako M, Yamaguchi T, Kinoshita T, Fujii M, Nashimoto A, et al. Adjuvant chemotherapy for gastric cancer with S-1, an oral fluoropyrimidine. N Engl J Med 2007;357:1810–1820.
Koizumi W, Kurihara M, Nakano S, Hasegawa K. Phase II study of S-1, a novel oral derivative of 5-fluorouracil, in advanced gastric cancer. For the S-1 Cooperative Gastric Cancer Study Group. Oncology 2000;58:191–197.
Sakata Y, Ohtsu A, Horikoshi N, Sugimachi K, Mitachi Y, Taguchi T. Late phase II study of novel oral fluoropyrimidine anticancer drug S-1 (1 M tegafur-0.4 M gimestat-1 M otastat potassium) in advanced gastric cancer patients. Eur J Cancer 1998;34:1715–1720.
Koizumi W, Tanabe S, Saigenji K, Ohtsu A, Boku N, Nagashima F, et al. Phase I/II study of S-1 combined with cisplatin in patients with advanced gastric cancer. Br J Cancer 2003;89:2207–2212.
Koizumi W, Narahara H, Hara T, Takagane A, Akiya T, Takagi M, et al. S-1 plus cisplatin versus S-1 alone for first-line treatment of advanced gastric cancer (SPIRITS trial): a phase III trial. Lancet Oncol 2008;9:215–221.
Kodera Y. Disseminated cancer cells in the peritoneal cavity: what can we do when we detect them? Gastric Cancer 2008;11:192–193.
Nakagawa S, Nashimoto A, Yabusaki H. Role of staging laparoscopy with peritoneal lavage cytology in the treatment of locally advanced gastric cancer. Gastric Cancer 2007;10:29–34.
Aiko T, Sasako M. The new Japanese classification of gastric carcinoma: points to be revised. Gastric Cancer 1998;1:25–30.
Maruyama K, Kaminishi M, Hayashi K, Isobe Y, Honda I, Katai H, et al. Gastric cancer treated in 1991 in Japan: data analysis of nationwide registry. Gastric Cancer 2006;9:51–66.
Fujitani K, Yang H, Kurokawa Y, Park DJ, Tsujinaka T, Park BJ, et al. Randomized controlled trial comparing gastrectomy plus chemotherapy with chemotherapy alone in advanced gastric cancer with a single non-curable factor: Japan Clinical Oncology Group Study JCOG 0705 and Korea Gastric Cancer Association Study KGCA01. Jpn J Clin Oncol 2008;38:504–506.
Yamanaka T, Matsumoto S, Teramukai S, Ishiwata R, Nagai Y, Fukushima M. Analysis of risk factors for severe adverse effects of oral 5-fluorouracil S-1 in patients with advanced gastric cancer. Gastric Cancer 2007;10:129–134.
Kinoshita T, Nashimoto A, Yamamura Y, Okamura T, Sasako M, Sakamoto J, et al. Feasibility study of adjuvant chemotherapy with S-1 (TS-1; tegafur, gimeracil, oteracil potassium) for gastric cancer. Gastric Cancer 2004;7:104–109.
Kodera Y, Ito S, Mochizuki Y, Kondo K, Koshikawa K, Suzuki N, et al. A phase II study of radical surgery followed by postoperative chemotherapy with S-1 for gastric carcinoma with free cancer cells in the peritoneal cavity (CCOG0301 study). Eur J Surg Oncol 2009;35:1158–1163.
Uedo N, Narahara H, Ishihara R, Takiuchi H, Goto M, Fjitani K, et al. Phase II study of a combination of irinotecan and S-1 in patients with advanced gastric cancer (OGSG0002). Oncology 2007;73:65–71.
Narahara H, Fujitani K, Takiuchi H, Sugimoto N, Inoue K, Uedo N, et al. Phase II study of a combination of S-1 and paclitaxel in patients with unresectable or metastatic gastric cancer. Oncology 2008;74:37–41.
Yoshida K, Ninomiya M, Takakura N, Hirabayashi N, Takiyama W, Sato Y, et al. Phase II study of docetaxel and S-1 combination therapy for advanced or recurrent gastric cancer. Clin Cancer Res 2006;12:3402–3407.
Cunningham D, Starling N, Rao S, Iveson T, Nicolson M, Coxon F, et al. Capecitabine and oxaliplatin for advanced esophagogastric cancer. N Engl J Med 2008;358:36–46.
Koizumi W, Takiuchi H, Yamada Y, Boku N, Fuse N, Muro K, et al. Phase II study of oxaliplatin plus S-1 as first-line treatment for advanced gastric cancer (G-SOX study). Ann Oncol 2010;21:1001–1005.
Chipponi J, Huguier M, Pezet D, Basso N, Hay JM, Quandalle P, et al. Randomized trial of adjuvant chemotherapy after curative resection for gastric cancer. Am J Surg 2004;187:440–445.
Bouché O, Ychou M, Burtin P, Bedenne L, Ducreux M, Lebreton G, et al. Adjuvant chemotherapy with 5-fluorouracil and cisplatin compared with surgery alone for gastric cancer: 7-year results of the FFCD randomized phase III trial (8801). Ann Oncol 2005;16:1488–1497.
Kochi M, Fujii M, Kanamori N, Kaiga T, Takahashi T, Kobayashi M, et al. Neoadjuvant chemotherapy with S-1 and CDDP in advanced gastric cancer. J Cancer Res Clin Oncol 2006;132:781–785.
Satoh S, Hasegawa S, Ozaki N, Okabe H, Watanabe G, Nagayama S, et al. Retrospective analysis of 45 consecutive patients with advanced gastric cancer treated with neoadjuvant chemotherapy using an S-1/CDDP combination. Gastric Cancer 2006;9:129–135.
Author information
Authors and Affiliations
Consortia
Additional information
for the Chubu Clinical Cancer Group
Rights and permissions
About this article
Cite this article
Kodera, Y., Ishiyama, A., Yoshikawa, T. et al. A feasibility study of postoperative chemotherapy with S-1 and cisplatin (CDDP) for gastric carcinoma (CCOG0703). Gastric Cancer 13, 197–203 (2010). https://doi.org/10.1007/s10120-010-0559-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10120-010-0559-y