Abstract
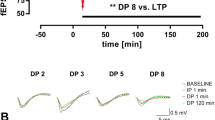
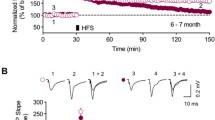
Various animal models of Alzheimer disease (AD) are characterized by deficits in spatial memory that are causally related to altered synaptic function and impairment of long-term potentiation (LTP) in the hippocampus. In Tg2576 AD mice, we compared LTP in 2 major hippocampal pathways, Schaffer collateral (SC) and mossy fiber (MF) pathways. Whereas LTP was completely abolished in the SC pathway of Tg2576 mice, we found no decrease in LTP induced by stimulation of the MF pathway. In fact, we found that in the MF pathway, LTP was slightly, but significantly, enhanced compared with that in the MF pathway of WT littermates. This pathway-specific impairment of LTP is not attributable to alterations in transmitter release, as indicated by an unaltered paired-pulse ratio. These results suggest that the spatial memory deficits normally seen in AD models arise primarily from LTP impairment at the SC pathway.
Similar content being viewed by others
References
Almeida, C.G., Tampellini, D., Takahashi, R.H., Greengard, P., Lin, M.T., Snyder, E.M., and Gouras, G.K. (2005). Beta-amyloid accumulation in APP mutant neurons reduces PSD-95 and GluR1 in synapses. Neurobiol. Dis. 20, 187–198.
Barber, R.C. (2010). Biomarkers for early detection of Alzheimer disease. J. Am. Osteopath. Assoc. 110, S10–15.
Bukalo, O., and Dityatev, A. (2006). Analysis of neural cell functions in gene knockout mice: electrophysiological investigation of synaptic plasticity in acute hippocampal slices. Methods Enzymol. 417, 52–66.
Chapman, P.F., White, G.L., Jones, M.W., Cooper-Blacketer, D., Marshall, V.J., Irizarry, M., Younkin, L., Good, M.A., Bliss, T.V., Hyman, B.T., et al. (1999). Impaired synaptic plasticity and learning in aged amyloid precursor protein transgenic mice. Nat. Neurosci. 2, 271–276.
D’Amelio, M., Cavallucci, V., Middei, S., Marchetti, C., Pacioni, S., Ferri, A., Diamantini, A., De Zio, D., Carrara, P., Battistini, L., et al. (2011). Caspase-3 triggers early synaptic dysfunction in a mouse model of Alzheimer’s disease. Nat. Neurosci. 14, 69–76.
Doussau, F., Humeau, Y., Benfenati, F., and Poulain, B. (2010). A novel form of presynaptic plasticity based on the fast reactivation of release sites switched off during low-frequency depression. J. Neurosci. 30, 16679–16691.
Duyckaerts, C., Delatour, B., and Potier, M.C. (2009). Classification and basic pathology of Alzheimer disease. Acta Neuropathol. 118, 5–36.
Gilbert, P.E., and Brushfield, A.M. (2009). The role of the CA3 hippocampal subregion in spatial memory: a process oriented behavioral assessment. Prog Neuropsychopharmacol. Biol. Psychiatry 33, 774–781.
Hsiao, K., Chapman, P., Nilsen, S., Eckman, C., Harigaya, Y., Younkin, S., Yang, F., and Cole, G. (1996). Correlative memory deficits, Abeta elevation, and amyloid plaques in transgenic mice. Science 274, 99–102.
Jacobsen, J.S., Wu, C.C., Redwine, J.M., Comery, T.A., Arias, R., Bowlby, M., Martone, R., Morrison, J.H., Pangalos, M.N., Reinhart, P.H., et al. (2006). Early-onset behavioral and synaptic deficits in a mouse model of Alzheimer’s disease. Proc. Natl. Acad. Sci. USA 103, 5161–5166.
Jin, K., Galvan, V., Xie, L., Mao, X.O., Gorostiza, O.F., Bredesen, D.E., and Greenberg, D.A. (2004) Enhanced neurogenesis in Alzheimer’s disease transgenic (PDGF-APPSw, Ind) mice. Proc. Natl. Acad. Sci. USA 101, 13363–13367.
Kawarabayashi, T., Younkin, L.H., Saido, T.C., Shoji, M., Ashe, K.H., and Younkin, S.G. (2001). Age-dependent changes in brain, CSF, and plasma amyloid (beta) protein in the Tg2576 transgenic mouse model of Alzheimer’s disease. J. Neurosci. 21, 372–381.
Langston, R.F., Stevenson, C.H., Wilson, C.L., Saunders, I., and Wood, E.R. (2010). The role of hippocampal subregions in memory for stimulus associations. Behav. Brain Res. 215, 275–291.
Lledo, P.M., Alonso, M., and Grubb, M.S. (2006). Adult neurogenesis and functional plasticity in neuronal circuits. Nat. Rev. Neurosci. 7, 179–193.
Moreno, H., Yu, E., Pigino, G., Hernandez, A.I., Kim, N., Moreira, J. E., Sugimori, M., and Llinas, R.R. (2009). Synaptic transmission block by presynaptic injection of oligomeric amyloid beta. Proc. Natl. Acad. Sci. USA 106, 5901–5906.
Morrison, J.H., and Hof, P.R. (1997). Life and death of neurons in the aging brain. Science 278, 412–419.
Nagy, V., Bozdagi, O., Matynia, A., Balcerzyk, M., Okulski, P., Dzwonek, J., Costa, R.M., Silva, A.J., Kaczmarek, L., and Huntley, G.W. (2006). Matrix metalloproteinase-9 is required for hippocampal late-phase long-term potentiation and memory. J. Neurosci. 26, 1923–1934.
Ondrejcak, T., Klyubin, I., Hu, N.W., Barry, A.E., Cullen, W.K., and Rowan, M.J. (2010). Alzheimer’s disease amyloid beta-protein and synaptic function. Neuromol. Med. 12, 13–26.
Ribaut-Barassin, C., Dupont, J.L., Haeberle, A.M., Bombarde, G., Huber, G., Moussaoui, S., Mariani, J., and Bailly, Y. (2003). Alzheimer’s disease proteins in cerebellar and hippocampal synapses during postnatal development and aging of the rat. Neuroscience 120, 405–423.
Rowan, M.J., Klyubin, I., Cullen, W.K., and Anwyl, R. (2003). Synaptic plasticity in animal models of early Alzheimer’s disease. Philos. Trans. R Soc. Lond. B Biol. Sci. 358, 821–828.
Ruiz, A., Campanac, E., Scott, R.S., Rusakov, D.A., and Kullmann, D.M. (2010). Presynaptic GABAA receptors enhance transmission and LTP induction at hippocampal mossy fiber synapses. Nat. Neurosci. 13, 431–438.
Saura, C.A., Choi, S.Y., Beglopoulos, V., Malkani, S., Zhang, D., Shankaranarayana Rao, B.S., Chattarji, S., Kelleher, R.J., 3rd, Kandel, E.R., Duff, K., et al. (2004). Loss of presenilin function causes impairments of memory and synaptic plasticity followed by age-dependent neurodegeneration. Neuron 42, 23–36.
Snyder, E.M., Nong, Y., Almeida, C.G., Paul, S., Moran, T., Choi, E.Y., Nairn, A.C., Salter, M.W., Lombroso, P.J., Gouras, G.K., et al. (2005). Regulation of NMDA receptor trafficking by amyloid-beta. Nat. Neurosci. 8, 1051–1058.
Sutherland, R.J., Whishaw, I.Q., and Kolb, B. (1983). A behavioural analysis of spatial localization following electrolytic, kainate-or colchicine-induced damage to the hippocampal formation in the rat. Behav. Brain Res. 7, 133–153.
Wang, H., Song, L., Lee, A., Laird, F., Wong, P.C., and Lee, H.K. (2010). Mossy fiber long-term potentiation deficits in BACE1 knock-outs can be rescued by activation of alpha7 nicotinic acetylcholine receptors. J. Neurosci. 30, 13808–13813.
Witton, J., Brown, J.T., Jones, M.W., and Randall, A.D. (2010). Altered synaptic plasticity in the mossy fibre pathway of transgenic mice expressing mutant amyloid precursor protein. Mol. Brain 3, 32.
Wojtowicz, T., and Mozrzymas, J.W. (2010). Late phase of long-term potentiation in the mossy fiber-CA3 hippocampal pathway is critically dependent on metalloproteinases activity. Hippocampus 20, 917–921.
Xavier, G.F., and Costa, V.C. (2009). Dentate gyrus and spatial behaviour. Prog. Neuropsychopharmacol. Biol. Psychiatry 33, 762–773.
Yamin, G. (2009). NMDA receptor-dependent signaling pathways that underlie amyloid beta-protein disruption of LTP in the hippocampus. J. Neurosci. Res. 87, 1729–1736.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Jung, J.H., An, K., Kwon, O.B. et al. Pathway-specific alteration of synaptic plasticity in Tg2576 mice. Mol Cells 32, 197–201 (2011). https://doi.org/10.1007/s10059-011-0077-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10059-011-0077-8