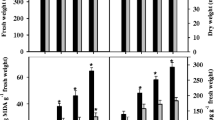
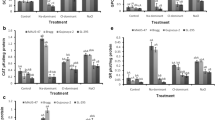
Abstract
Despite the fact that when subjected to salinity stress most plants accumulate high concentrations of sodium (Na+) and chloride (Cl−) ions in their tissues, major research has however been focused on the toxic effects of Na+. Consequently, Cl− toxicity mechanisms in annual plants, particularly in inducing oxidative stress, are poorly understood. Here, the extent to which Na+ and/or Cl− ions contribute in inducing oxidative stress and regulating the adaptive antioxidant defense is shown in two Indica rice genotypes differing in their salt tolerance. Equimolar (100 mM) concentrations of Na+, Cl−, and NaCl (EC ≈ 10 dS m−1) generated free-radical (O2 •−, •OH) and non-radical (H2O2) forms of reactive oxygen species (ROS) and triggered cell death in leaves of 21-day-old hydroponically grown rice seedlings as evident by spectrophotometric quantifications and histochemical visualizations. The magnitude of ROS-mediated oxidative damage was higher in sensitive cultivar, whereas NaCl proved to be most toxic among the treatments. Salt treatments significantly increased activities of antioxidant enzymes and their isozymes including superoxide dismutase, catalase, peroxidase, ascorbate peroxidase, and glutathione reductase. Na+ and Cl− ions showed additive effects under NaCl in activating the antioxidant enzyme machinery, and responses were more pronounced in tolerant cultivar. The expression levels of SodCc2, CatA, and OsPRX1 genes were largely consistent with the activities of their corresponding enzymes. Salt treatments caused an imbalance in non-enzymatic antioxidants ascorbic acid, α-tocopherol, and polyphenols, with greater impacts under NaCl than Na+ and Cl− separately. Results revealed that though Cl− was relatively less toxic than its counter-cation, its effects cannot be totally ignored. Both the cultivars responded in the same manner, but the tolerant cultivar maintained lower Na+/K+ and ROS levels coupled with better antioxidant defense under all three salt treatments.











Similar content being viewed by others
Abbreviations
- APX:
-
Ascorbate peroxidase
- CAT:
-
Catalase
- DAB:
-
Diaminobenzidine
- EC:
-
Electrical conductivity
- GAPDH :
-
Rice glyceraldehyde 3-phosphate dehydrogenase
- GR:
-
Glutathione reductase
- GSH:
-
Reduced glutathione
- GSSG:
-
Oxidized glutathione
- NBT:
-
Nitro blue tetrazolium
- POX:
-
Peroxidase
- ROS:
-
Reactive oxygen species
- RT-PCR:
-
Reverse transcriptase polymerase chain reaction
- SOD:
-
Superoxide dismutase
References
Achary VMM, Patnaik AR, Panda BB (2012) Oxidative biomarkers in leaf tissue of barley seedlings in response to aluminum stress. Ecotoxicol Environ Saf 75:16–26. doi:10.1016/j.ecoenv.2011.08.015
Aebi H (1984) Catalase in vitro. Meth Enzymol 105:121–126. doi:10.1016/S0076-6879(84)05016-3
Anderson JV, Hess JL, Chevone BI (1990) Purification, characterization, and immunological properties for two isoforms of glutathione reductase from eastern white pine needles. Plant Physiol 94:1402–1409. doi:10.1104/pp.94.3.1402
Anderson MD, Prasad TK, Stewart CR (1995) Changes in isozyme profiles of catalase, peroxidase, and glutathione reductase during acclimation to chilling in mesocotyls of maize seedlings. Plant Physiol 109:1247–1257. doi:10.1104/pp.109.4.1247
Anil VS, Krishnamurthy P, Kuruvilla S, Sucharitha K, Thomas G, Mathew MK (2005) Regulation of the uptake and distribution of Na+ in shoots of rice (Oryza sativa) variety Pokkali: role of Ca2+ in salt tolerance response. Physiol Plant 124:451–464. doi:10.1111/j.1399-3054.2005.00529.x
Anjum NA, Ahmad I, Mohmood I, Pacheco M, Duarte AC, Pereira E, Umar S, Ahmad A, Khan NA, Iqbal M, Prasad NMV (2012) Modulation of glutathione and its related enzymes in plants’ responses to toxic metals and metalloids—a review. Environ Exp Bot 75:307–324. doi:10.1016/j.envexpbot.2011.07.002
Beauchamp C, Fridovich I (1971) Superoxide dismutase: improved assays and an assay applicable to acrylamide gels. Anal Biochem 44:276–287. doi:10.1016/0003-2697(71)90370-8
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein–dye binding. Anal Biochem 72:248–254. doi:10.1016/0003-2697(76)90527-3
Cameron GR, Milton RF, Allan JW (1943) Toxicity of tannic acid: an experimental investigation. Lancet 242:179–186. doi:10.1016/S0140-6736(00)87163-9
Castillo FI, Penel I, Greppin H (1984) Peroxidase release induced by ozone in Sedum album leaves. Plant Physiol 74:846–851. doi:10.1104/pp.74.4.846
Chaitanya KSK, Naithani SC (1994) Role of superoxide, lipid peroxidation and superoxide dismutase in membrane perturbation during loss of viability in seeds of Shorea robusta Gaertn.f. New Phytol 126:623–627. doi:10.1111/j.1469-8137.1994.tb02957.x
Chapman HD, Pratt PF (1961) Method for analysis of soil, plants and waters. University of California, Berkeley, CA
Chawla S, Jain S, Jain V (2013) Salinity induced oxidative stress and antioxidant system in salt-tolerant and salt-sensitive cultivars of rice (Oryza sativa L.). J Plant Biochem Biotechnol 22:27–34. doi:10.1007/s13562-012-0107-4
Dang YP, Dalal RC, Mayer DG, McDonald M, Routley R, Schwenke GD, Buck SR, Daniells IG, Singh DK, Manning W, Ferguson N (2008) High subsoil chloride concentrations reduce soil water extraction and crop yield on Vertisols in north-eastern Australia. Aust J Agr Res 59:321–330. doi:10.1071/AR07192
Dhindsa RA, Plumb-Dhindsa P, Thorpe TA (1981) Leaf senescence: correlated with increased permeability and lipid peroxidation and decreased levels of superoxide dismutase and catalase. J Exp Bot 126:93–101. doi:10.1093/jxb/32.1.93
El-Shabrawi H, Kumar B, Kaul T, Reddy MK, Singla-Pareek SL, Sopory SK (2010) Redox homeostasis, antioxidant defense and methylglyoxal detoxification as markers for salt tolerance in Pokkali rice. Protoplasma 245:85–96. doi:10.1007/s00709-010-0144-6
Gill SS, Tuteja N (2010) Reactive oxygen species and antioxidant machinery in abiotic stress tolerance in crop plants. Plant Physiol Biochem 48:909–930. doi:10.1016/j.plaphy.2010.08.016
Halliwell B, Gutteridge JMC, Aruma O (1987) The deoxyribose method: a simple ‘test tube’ assay for determination of rate constant for reaction of hydroxyl radicals. Anal Biochem 165:215–219. doi:10.1016/0003-2697(87)90222-3
Huang Z, Zhao L, Chen D, Liang M, Liu Z, Hongbo S, Xiaohua L (2013) Salt stress encourages proline accumulation by regulating proline biosynthesis and degradation in Jerusalem artichoke plantlets. PLoS ONE 8(4):e62085. doi:10.1371/journal.pone.0062085
Huseynova IM, Sultanova NF, Aliyev JA (2014) Histochemical visualization of ROS and antioxidant response to viral infections of vegetable crops grown in Azerbaijan. Plant Physiol Biochem 81:26–35. doi:10.1016/j.plaphy.2014.03.002
Ismail A, Takeda S, Nick PR (2014) Life and death under salt stress: same players, different timing? J Exp Bot. doi:10.1093/jxb/eru159
Kamyab-Talesh F, Mousavi SF, Asadi R, Rezaei M, Khaledian MR (2014) Evaluation of some rice cultivars response to salinity stress using resistance indices. Arch Agron Soil Sci 60:1303–1314. doi:10.1080/03650340.2014.891730
Khan MA, Shirazi MU, Khan MA, Mujtaba SM, Islam E, Mumtaz S (2009) Role of proline, K/Na ratio and chlorophyll content in salt tolerance of wheat (Triticum aestivum L.). Pak J Bot 41:633–638
Kumar V, Khare T (2014) Individual and additive effects of Na+ and Cl− ions on rice under salinity stress. Arch Agron Soil Sci. doi:10.1080/03650340.2014.936400
Kumar V, Shriram V, Nikam TD, Jawali N, Shitole MG (2009) Antioxidant enzyme activities and protein profiling under salt stress in indica rice genotypes differing in salt tolerance. Arch Agron Soil Sci 55:379–394. doi:10.1080/03650340802595543
Lin CC, Kao CH (2001) Relative importance of Na+, Cl−, and abscisic acid in NaCl induced inhibition of root growth of rice seedlings. Plant Soil 237:165–171. doi:10.1023/A:1013321813454
Loreto F, Velikova V (2001) Isoprene produced by leaves protects the photosynthetic apparatus against ozone damage, quenches ozone products, and reduces lipid peroxidation of cellular membranes. Plant Physiol 127:1781–1787. doi:10.1104/pp.010497
Luo Q, Yu B, Liu Y (2005) Differential sensitivity to chloride and sodium ions in seedlings of Glycine max and G. soja under NaCl stress. J Plant Physiol 162:1003–1012. doi:10.1016/j.jplph.2004.11.008
Mahanty S, Kaul T, Pandey P, Reddy RA, Mallikarjuna G, Reddy CS et al (2012) Biochemical and molecular analyses of copper–zinc superoxide dismutase from a C4 plant Pennisetum glaucum reveals an adaptive role in response to oxidative stress. Gene 505:309–317. doi:10.1016/j.gene.2012.06.001
Malick CP, Singh MB (1980) Plant enzymology and histo-enzymology. Kalyani, New Delhi
Menezes-Benavente L, Teixeira FK, Alvim Kamei CL, Margis-Pinheiro M (2004) Salt stress induces altered expression of genes encoding antioxidant enzymes in seedlings of a Brazilian indica rice (Oryza sativa L.). Plant Sci 166:323–331. doi:10.1016/j.plantsci.2003.10.001
Mittler R (2002) Oxidative stress, antioxidants and stress tolerance. Trends Plant Sci 7:405–410. doi:10.1016/S1360-1385(02)02312-9
Mittler R, Zilinskas BA (1993) Detection of ascorbate peroxidase activity in native gels by inhibition of the ascorbate-dependent reduction of nitroblue tetrazolium. Anal Biochem 212:540–546. doi:10.1006/abio.1993.1366
Moron MS, Depierre JW, Mannervik B (1979) Levels of glutathione, glutathione reductase and glutathione S-transferase activities in rat lung and liver. Biochim et Biophys Acta 582:67–78. doi:10.1016/0304-4165(79)90289-7
Moya JL, Gomez-Cadenas A, Primo-Millo E, Talon M (2003) Chloride absorption in salt-sensitive Carrizo citrange and salt-tolerant Cleopatra mandarin citrus rootstocks in linked to water use. J Exp Bot 54:825–833. doi:10.1093/jxb/erg064
Mukharjee SP, Choudhari MA (1983) Implication of water stress-induced changes in the levels of endogenous ascorbic acid and hydrogen peroxide in Vigna seedlings. Physiol Plant 58:166–170. doi:10.1111/j.1399-3054.1983.tb04162.x
Munns R, Tester S (2008) Mechanism of salinity tolerance. Annu Rev Plant Biol 59:651–681. doi:10.1146/annurev.arplant.59.032607.092911
Munns R, James RA, Xu B, Athman A, Conn SJ, Jordans C et al (2012) Wheat grain yield on saline soils is improved by an ancestral Na+ transporter gene. Nat Biotechnol 30:360–364. doi:10.1038/nbt.2120
Nakano Y, Asada K (1981) Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in spinach chloroplasts. Plant Cell Physiol 22:867–880
Qureshi MI, Abdin MZ, Ahmad J, Iqbal M (2013) Effect of long-term salinity on cellular antioxidants, compatible solute and fatty acid profile of Sweet Annie (Artemisia annua L.). Phytochemistry 95:215–223. doi:10.1016/j.phytochem.2013.06.026
Romeo-Puertas MC, Rodriguez-serrano M, Corpas FJ, Gomez M, del-Rio LA, Sandalio LM (2004) Cadmium-induced subcellular accumulation of O2 •− and H2O2 in pea leaves. Plant Cell Environ 27:1122–1134. doi:10.1111/j.1365-3040.2004.01217.x
Rosenberg HR (1992) Chemistry and physiology of vitamins. Inter Science, New York, pp 452–453
Sairam RK, Srivastava GC, Agarwal S, Meena RC (2005) Differences in antioxidant activity in response to salinity stress in tolerant and susceptible wheat genotypes. Biol Plant 49:85–91. doi:10.1007/s10535-005-5091-2
Smith IK, Vierheller TL, Thorne CA (1988) Assay of glutathione reductase in crude tissue homogenate using 5,5′-dithiobis (2-nitrobenzoic acid). Anal Biochem 175:408–413. doi:10.1016/0003-2697(88)90564-7
Tavakkoli E, Rengasamy P, McDonald GK (2010) High concentrations of Na+ and Cl− ions in soil solution have simultaneous detrimental effects on growth of faba bean under salinity stress. J Exp Bot 61:4449–4459. doi:10.1093/jxb/erq251
Tavakkoli E, Fatehi F, Coventry S, Rengasamy P, McDonald GK (2011) Additive effects of Na+ and Cl− ions on barley growth under salinity stress. J Exp Bot 62:2189–2203. doi:10.1093/jxb/erq422
Teakle NL, Tyerman SD (2010) Mechanisms of Cl− transport contributing to salt tolerance. Plant Cell Environ 33:566–589. doi:10.1111/j.1365-3040.2009.02060.x
Tilman D, Balzer C, Hill J, Belfort BL (2011) Global food demand and the sustainable intensification of agriculture. Proc Natl Acad Sci USA 108:20260–20264. doi:10.1073/pnas.1116437108
Trivedi D, Gill SS, Yadav S, Tuteja N (2013) Genome-wide analysis of glutathione reductase (GR) genes from rice and Arabidopsis. Plant Signal Behav 8:e2301–e2307. doi:10.4161/psb.23021
Turan S, Tripathy BC (2013) Salt and genotype impact on antioxidative enzymes and lipid peroxidation in two rice cultivars during de-etiolation. Protoplasma 250:209–222. doi:10.1007/s00709-012-0395-5
Turkan I, Demiral T (2009) Recent development in understanding salinity tolerance. Environ Exp Bot 67:2–9. doi:10.1016/j.envexpbot.2009.05.008
Vranova E, Inze D, Van Breusegem F (2002) Signal transduction during oxidative stress. J Exp Bot 53:1227–1236. doi:10.1093/jexbot/53.372.1227
White PJ, Broadley MR (2001) Chloride in soils and its uptake and movement within the plant: a review. Ann Bot 88:967–988. doi:10.1006/anbo.2001.1540
Woodbury W, Spencer AK, Stahmann MA (1971) An improved procedure using ferricyanide for detecting catalase isozymes. Anal Biochem 44:301–305. doi:10.1016/0003-2697(71)90375-7
Xu G, Magen H, Tarchitzky J, Kafkafi U (2000) Advances in chloride nutrition of plants. Adv Agron 68:97–150. doi:10.1016/S0065-2113(08)60844-5
Yao S, Chen S, Xu D, Lan H (2010) Plant growth and responses of antioxidants of Chenopodium album to long-term NaCl and KCl stress. Plant Growth Regul 60:115–125. doi:10.1007/s10725-009-9426-4
Yildiztugay E, Ozfidan-Konakci C, Kucukoduk M (2014) Modulation of osmotic adjustment and antioxidant status in salt-stressed leaves of Thermopsis turcica. Acta Physiol Plant 36:125–138. doi:10.1007/s11738-013-1393-8
Zhang XK, Zhou QH, Cao JH, Yu BJ (2011) Differential Cl− salt tolerance and NaCl-induced alternations of tissue and cellular ion fluxes in Glycine max, Glycine soja and their hybrid seedlings. J Agron Crop Sci 197:329–339. doi:10.1111/j.1439-037x.2011.00467.x
Acknowledgments
Financial support from the Science and Engineering Research Board (SERB), Government of India (grant number SR/FT/LS-93/2011) for carrying out this work is gratefully acknowledged. The authors also like to acknowledge the use of facilities created under DST-FIST and DBT Star College Schemes implemented at Modern College, Ganeshkhind, Pune, and thank the college authorities for permitting to utilize these facilities.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling Editor: Peter Nick
Rights and permissions
About this article
Cite this article
Khare, T., Kumar, V. & Kishor, P.B.K. Na+ and Cl− ions show additive effects under NaCl stress on induction of oxidative stress and the responsive antioxidative defense in rice. Protoplasma 252, 1149–1165 (2015). https://doi.org/10.1007/s00709-014-0749-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00709-014-0749-2