Abstract
Background
We used [F-18] FDG microPET imaging as part of a longitudinal study to investigate changes in the brain.
Methods
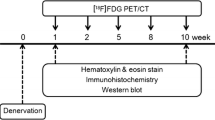
Glucose metabolism during the development of neuropathic pain after tibial and sural nerve transection (TST) model rats. MicroPET images were obtained 1 week before operation and then weekly for 8 weeks post-operation.
Results
The behavioral test was performed immediately after the every FDG administration. After TST modeling, neuropathic pain rats showed increased mechanical sensitivity of the injured hind paw. The withdrawal response to mechanical pain stimulation by von Frey filaments was observed within the first week (3.8 ± 0.73), and it rapidly increased in the third week (7.13 ± 0.82). This response reached a peak in the fourth week after surgery (9.0 ± 0.53), which persisted until the eighth week. In microPET scan imaging, cerebellum, which initially started from the ansiform lobule, was activated gradually to all part from the third week in all image acquisitions through the eighth week.
Conclusions
The longitudinal microPET scan study of brains from neuropathic pain rat models showed sequential cerebellar activity that was in accordance with results from behavioral test responses, thus supporting a role for the cerebellum in the development of neuropathic pain.




Similar content being viewed by others
References
Apkarian AV, Bushnell MC, Treede RD, Zubieta JK (2005) Human brain mechanisms of pain perception and regulation in health and disease. Eur J Pain 9:463–484
Baron R (2006) Mechanisms of disease: neuropathic pain–a clinical perspective. Nat Clin Pract Neurol 2:95–106
Becerra L, Morris S, Bazes S, Gostic R, Sherman S, Gostic J, Pendse G, Moulton E, Scrivani S, Keith D, Chizh B, Borsook D (2006) Trigeminal neuropathic pain alters responses in CNS circuits to mechanical (brush) and thermal (cold and heat) stimuli. J Neurosci 26:10646–10657
Bentley DE, Derbyshire SW, Youell PD, Jones AK (2003) Caudal cingulate cortex involvement in pain processing: an inter-individual laser evoked potential source localisation study using realistic head models. Pain 102:265–271
Bingel U, Quante M, Knab R, Bromm B, Weiller C, Buchel C (2002) Subcortical structures involved in pain processing: evidence from single-trial fMRI. Pain 99:313–321
Bingel U, Tracey I (2008) Imaging CNS modulation of pain in humans. Physiology (Bethesda) 23:371–380
Borsook D, Moulton EA, Tully S, Schmahmann JD, Becerra L (2008) Human cerebellar responses to brush and heat stimuli in healthy and neuropathic pain subjects. Cerebellum 7:252–272
Bouhassira D, Attal N, Fermanian J, Alchaar H, Gautron M, Masquelier E, Rostaing S, Lanteri-Minet M, Collin E, Grisart J, Boureau F (2004) Development and validation of the Neuropathic Pain Symptom Inventory. Pain 108:248–257
Craig AD (2003) Pain mechanisms: labeled lines versus convergence in central processing. Annu Rev Neurosci 26:1–30
Davis KD (2000) The neural circuitry of pain as explored with functional MRI. Neurol Res 22:313–317
Dimitrova A, Kolb FP, Elles HG, Maschke M, Forsting M, Diener HC, Timmann D (2003) Cerebellar responses evoked by nociceptive leg withdrawal reflex as revealed by event-related FMRI. J Neurophysiol 90:1877–1886
Dowdall T, Robinson I, Meert TF (2005) Comparison of five different rat models of peripheral nerve injury. Pharmacol Biochem Behav 80:93–108
Dum RP, Levinthal DJ, Strick PL (2009) The spinothalamic system targets motor and sensory areas in the cerebral cortex of monkeys. J Neurosci 29:14223–14235
Ekerot CF, Garwicz M, Schouenborg J (1991) The postsynaptic dorsal column pathway mediates cutaneous nociceptive information to cerebellar climbing fibres in the cat. J Physiol 441:275–284
Finnerup NB, Otto M, McQuay HJ, Jensen TS, Sindrup SH (2005) Algorithm for neuropathic pain treatment: an evidence based proposal. Pain 118:289–305
Grodd W, Hulsmann E, Ackermann H (2005) Functional MRI localizing in the cerebellum. Neurosurg Clin N Am 16:77–99, v
Iadarola MJ, Max MB, Berman KF, Byas-Smith MG, Coghill RC, Gracely RH, Bennett GJ (1995) Unilateral decrease in thalamic activity observed with positron emission tomography in patients with chronic neuropathic pain. Pain 63:55–64
Ingvar M (1999) Pain and functional imaging. Philos Trans R Soc Lond B Biol Sci 354:1347–1358
Jie W, Pei-Xi C (1992) Discharge response of cerebellar Purkinje cells to stimulation of C-fiber in cat saphenous nerve. Brain Res 581:269–272
Kelly RM, Strick PL (2003) Cerebellar loops with motor cortex and prefrontal cortex of a nonhuman primate. J Neurosci 23:8432–8444
Keltner JR, Furst A, Fan C, Redfern R, Inglis B, Fields HL (2006) Isolating the modulatory effect of expectation on pain transmission: a functional magnetic resonance imaging study. J Neurosci 26:4437–4443
Lee BH, Won R, Baik EJ, Lee SH, Moon CH (2000) An animal model of neuropathic pain employing injury to the sciatic nerve branches. Neuroreport 11:657–661
Lenz FA, Rios M, Zirh A, Chau D, Krauss G, Lesser RP (1998) Painful stimuli evoke potentials recorded over the human anterior cingulate gyrus. J Neurophysiol 79:2231–2234
Maschke M, Erichsen M, Drepper J, Jentzen W, Muller SP, Kolb FP, Diener HC, Timmann D (2002) Limb flexion reflex-related areas in human cerebellum. Neuroreport 13:2325–2330
Mason P (2005) Ventromedial medulla: pain modulation and beyond. J Comp Neurol 493:2–8
Meyer ME, Cottrell GA, Van Hartesveldt C (1993) Intracerebral haloperidol potentiates the dorsal immobility response in the rat. Pharmacol Biochem Behav 44:157–160
Moisset X, Bouhassira D (2007) Brain imaging of neuropathic pain. Neuroimage 37(Suppl 1):S80–S88
Moulton EA, Schmahmann JD, Becerra L, Borsook D (2010) The cerebellum and pain: passive integrator or active participator? Brain Res Rev 65:14–27
Ohyama T, Nores WL, Murphy M, Mauk MD (2003) What the cerebellum computes. Trends Neurosci 26:222–227
Petrovic P, Ingvar M, Stone-Elander S, Petersson KM, Hansson P (1999) A PET activation study of dynamic mechanical allodynia in patients with mononeuropathy. Pain 83:459–470
Peyron R, Garcia-Larrea L, Gregoire MC, Convers P, Lavenne F, Veyre L, Froment JC, Mauguiere F, Michel D, Laurent B (1998) Allodynia after lateral-medullary (Wallenberg) infarct. A PET study. Brain 121(Pt 2):345–356
Peyron R, Laurent B, Garcia-Larrea L (2000) Functional imaging of brain responses to pain. A review and meta-analysis (2000). Neurophysiol Clin 30:263–288
Peyron R, Schneider F, Faillenot I, Convers P, Barral FG, Garcia-Larrea L, Laurent B (2004) An fMRI study of cortical representation of mechanical allodynia in patients with neuropathic pain. Neurology 63:1838–1846
Restuccia D, Della Marca G, Valeriani M, Leggio MG, Molinari M (2007) Cerebellar damage impairs detection of somatosensory input changes. A somatosensory mismatch-negativity study. Brain 130:276–287
Romero A, Rojas S, Cabanero D, Gispert JD, Herance JR, Campillo A, Puig MM (2011) A (1)(8)F-fluorodeoxyglucose MicroPET imaging study to assess changes in brain glucose metabolism in a rat model of surgery-induced latent pain sensitization. Anesthesiology 115:1072–1083
Saab CY, Willis WD (2001) Nociceptive visceral stimulation modulates the activity of cerebellar Purkinje cells. Exp Brain Res 140:122–126
Schmidt BL, Tambeli CH, Barletta J, Luo L, Green P, Levine JD, Gear RW (2002) Altered nucleus accumbens circuitry mediates pain-induced antinociception in morphine-tolerant rats. J Neurosci 22:6773–6780
Schmidt BL, Tambeli CH, Gear RW, Levine JD (2001) Nicotine withdrawal hyperalgesia and opioid-mediated analgesia depend on nicotine receptors in nucleus accumbens. Neuroscience 106:129–136
Schmidt BL, Tambeli CH, Levine JD, Gear RW (2002) mu/delta cooperativity and opposing kappa-opioid effects in nucleus accumbens-mediated antinociception in the rat. Eur J Neurosci 15:861–868
Schweinhardt P, Fransson P, Olson L, Spenger C, Andersson JL (2003) A template for spatial normalisation of MR images of the rat brain. J Neurosci Methods 129:105–113
Schweinhardt P, Glynn C, Brooks J, McQuay H, Jack T, Chessell I, Bountra C, Tracey I (2006) An fMRI study of cerebral processing of brush-evoked allodynia in neuropathic pain patients. Neuroimage 32:256–265
Seminowicz DA, Laferriere AL, Millecamps M, Yu JS, Coderre TJ, Bushnell MC (2009) MRI structural brain changes associated with sensory and emotional function in a rat model of long-term neuropathic pain. Neuroimage 47:1007–1014
Stoodley CJ, Schmahmann JD (2010) Evidence for topographic organization in the cerebellum of motor control versus cognitive and affective processing. Cortex 46:831–844
Tambeli CH, Fischer L, Monaliza SL, Menescal-de-Oliveira L, Parada CA (2012) The functional role of ascending nociceptive control in defensive behavior. Brain Res 1464:24–29
Tesche CD, Karhu JJ (2000) Anticipatory cerebellar responses during somatosensory omission in man. Hum Brain Mapp 9:119–142
Tracey I (2005) Nociceptive processing in the human brain. Curr Opin Neurobiol 15:478–487
Witting N, Kupers RC, Svensson P, Jensen TS (2006) A PET activation study of brush-evoked allodynia in patients with nerve injury pain. Pain 120:145–154
Woolf CJ, Mannion RJ (1999) Neuropathic pain: aetiology, symptoms, mechanisms, and management. Lancet 353:1959–1964
Zimmermann M (1983) Ethical guidelines for investigations of experimental pain in conscious animals. Pain 16:109–110
Acknowledgments
This study was financially supported by a grant from the Industrial Source Technology Development Program (no.10033812) of the Ministry of Knowledge Economy (MKE).
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Additional information
Jinhyung Kim and Jaewoo Shin contributed equally to this work.
Rights and permissions
About this article
Cite this article
Kim, J., Shin, J., Oh, JH. et al. Longitudinal FDG microPET imaging of neuropathic pain: does cerebellar activity correlate with neuropathic pain development in a rat model?. Acta Neurochir 157, 1051–1057 (2015). https://doi.org/10.1007/s00701-015-2415-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00701-015-2415-7