Abstract
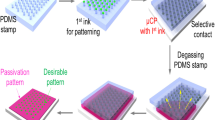
We report on a straightforward method for creating micropatterns of multiple biomolecules. The anti-fouling agent 2-methacryloyloxyethylphosphorylcholine (MPC) polymer and a photolabile linker (PL) were covalently linked to an amino-terminated silane surface. Patterns were generated by selective removal of the MPC polymer via UV irradiation. Multiple micropatterns of fluorescein isothiocyanate (FITC)-labeled bovine serum albumin (BSA) and rhodamine-labeled goat fragment antigen-binding fragments (FAB) were deposited on a same glass substrate. We also employed micropatterning of multiple biomolecules in that Texas red-labeled BSA and FITC-labeled rabbit anti-mouse IgG were placed inside a microchannel.

This technique was based on combining 2-methacryloyloxyethyl phosphorylcholine (MPC) polymer, which is known to non-biofouling compound, photocleavable linker (PL) that was modified to localize cells and to connect between MPC polymer and amino-terminated silanized surface. Using ultraviolet (UV) light illumination, MPC polymer can be selectively eliminated by photochemical reaction in order which resulted in micropatterning of multiple biomolecules.





Similar content being viewed by others
References
Kusnezow W, Hoheisel JD (2003) Solid supports for microarray immunoassays. J Mol Recognit 16:165–176
MacBeath G, Schreiber SL (2000) Printing proteins as microarrays for high-throughput function determination. Science 289:1760–1763
Haab BB, Dunham MJ, Brown PO (2001) Protein microarray for highly parallel detection and quantitation of specific proteins and antibodies in complex solutions. Genome Biol 2:1–13
Campbell RE (2009) Fluorescent-protein based biosensors: modulation of energy transfer as a design principle. Anal Chem 81:5972–5979
Chalmeau J, Salomé L, Thibault C, Severac C, Vieu C (2007) Elaboration of micro-domains of supported bilayer membranes using micro-contact printing. Microelectron Eng 84:1754–1757
Iversen L, Cherouati N, Berthing NT, Stamou D, Martinez KL (2008) Templated protein assembly on micro-contact-printed surface patterns. Use of the SNAP-tag protein functionality. Langmuir 24:6375–6381
Mooney JF, Hunt AJ, Mcintosh JR, Liberko CA, Walba DM, Rogers CT (1996) Patterning of functional antibodies and other proteins by photolithography of silane monolayers. Proc Natl Acad Sci 93:12287–12291
Flounders AW, Brandon DL, Bates AH (1997) Patterning of immobilized antibody layers via photolithography and oxygen plasma exposure. Biosens Bioelectron 12:447–456
Lim JH, Ginger DS, Lee KB, Heo J, Nam JM, Mirkin CA (2003) Direct-write dip-pen nanolithography of proteins on modified silicon oxide surfaces. Angew Chem Int Ed 42:2309–2312
Valiokas R, Vaitekonis A, Klenkar G, Trinkunas G, Liedberg B (2006) Selective recruitment of membrane protein complexes onto gold substrates patterned by dip-pen nanolithography. Langmuir 22:3456–3460
Gu J, Yam CM, Li S, Cai C (2004) Nanometric protein arrays on protein-resistant monolayers on silicon surfaces. J Am Chem Soc 126:8098–8099
Maury P, Escalante M, Péter M, Reinhoudt DN, Subramaniam V, Huskens J (2007) Creating nanopatterns of His-Tagged proteins on surfaces by nanoimprint lithography using specific NiNTA-Histidine interactions. Small 9:1584–1592
Escalante M, Zhao Y, Ludden MJW, Vermeij R, Olsen JD, Berenschot E, Hunter CN, Huskens J, Subramaniam V, Otto C (2008) Nanometer arrays of functional light harvesting antenna complexes by nanoimprint lithography and host−guest interactions. J Am Chem Soc 130:8892–8893
Rundqvist J, Mendoza B, Werbin JL, Heinz WF, Lemmon C, Romer L, Haviland DB, Hoh JH (2007) High fidelity functional patterns of an extracellular matrix protein by electron beam-based inactivation. J Am Chem Soc 129:59–67
Robin S, Gandhi AA, Gregor M, Laffir FR, Pecenik T, Pecenik A, Soulimane T, Tofail SAM (2011) Charge specific protein placement at submicrometer and nanometer scale by direct modification of surface potential by electron beam. Langmuir 27:14968–14974
Shin DS, Lee KN, Jang KH, Kim JK, Jung WJ, Kim YK, Lee YS (2003) Protein patterning by maskless photolithography on hydrophilic polymer-grafted surface. Biosens Bioelectron 19:485–494
Jonkheijm P, Weinrich D, Köhn M, Engelkamp H, Christianen PCM, Kuhlmann J, Maan JC, Nüsse D, Schroeder H, Wacker R, Breinbauer R, Niemeyer CM, Waldmann H (2008) Photochemical surface patterning by the thiol-ene reaction. Angew Chem Int Ed 47:4421–4424
Khademhosseini A, Suh KY, Jon S, Eng G, Yeh J, Chen GJ, Langer R (2004) A soft lithographic approach to fabricate patterned microfluidic channels. Anal Chem 76:3675–3681
Tsukahara T, Hibara A, Ikeda Y, Kitamori T (2007) NMR study of water molecules confined in extended nanospaces. Angew Chem Int Ed 46:1180–1183
Akiyama Y, Yoshitake A, Morishima K, Kogi A, Kikutani Y, Tokeshi M, Kitamori T (2004) Rapid bonding of pyrex glass microchips. Electrophoresis 28:994–1001
Balakirev MY, Porte S, Vernaz-Griz M, Berger M, Arié JP, Fouqué B, Chatelain F (2005) Photochemical patterning of biological molecules inside a glass capillary. Anal Chem 77:5474–5479
Kaji H, Hashimoto M, Nishizawa M (2006) On-demand patterning of protein matrixes inside a microfluidic device. Anal Chem 78:5469–5473
Jang K, Sato K, Mawatari K, Konno T, Ishihara K, Kitamori T (2009) Surface modification by 2-methacryloyloxyethyl phosphorylcholine coupled to a photolabile linker for cell micro patterning. Biomaterials 30:1413–1420
Jang K, Sato K, Tanaka Y, Xu Y, Sato M, Nakajima T, Mawatari K, Konno T, Ishihara K, Kitamori T (2010) An efficient surface modification using 2-methacryloyloxyehtyl phosphorylcholine to control cell attachment via photochemical reaction in a microchannel. Lab Chip 10:1937–1945
Holmes CP (1997) Model studies for new o-nitrobenzyl photolabile linkers: substituent effects on the rates of photochemical cleavage. J Org Chem 62:2370–2380
Kloxin AM, Kasko AM, Salinas CN, Anseth KS (2009) Photodegradable hydrogels for dynamic tuning of physical and chemical properties. Science 324:59–63
Ishihara K, Ueda T, Nakabayashi N (1990) Preparation of phospholipid polymers and their properties as polymer hydrogel membranes. Polym J 22:355–360
Xu Y, Takai M, Ishihara K (2009) Protein adsorption and cell adhesion on cationic, neutral, and anionic 2-methacryloyloxyethyl phosphorylcholine copolymer surfaces. Biomaterials 30:4930–4938
Sigal GB, Mrksich M, Whitesides GM (1998) Effect of surface wettability on the adsorption of proteins and detergents. J Am Chem Soc 120:3464–3473
Roach P, Farrar D, Perry CC (2005) Interpretation of protein adsorption: surface-induced conformational changes. J Am Chem Soc 127:8168–8173
Acknowledgments
This study was supported by a Core Research of Evolutional Science & Technology (CREST) from Japan Science and Technology Agency (JST) and by the Global Center of Excellence for Mechanical Systems Innovation (GMSI) from The University of Tokyo Global COE program.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jang, K., Xu, Y., Sato, K. et al. Micropatterning of biomolecules on a glass substrate in fused silica microchannels by using photolabile linker-based surface activation. Microchim Acta 179, 49–55 (2012). https://doi.org/10.1007/s00604-012-0856-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00604-012-0856-8