Abstract
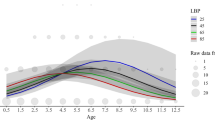
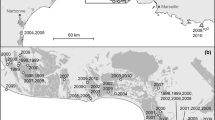
Breeding dispersal, defined as the net movement between successive breeding sites, remains a poorly understood and seldom reported phenomenon in mammals, despite its importance for population dynamics and genetics. In large herbivores, females may be more mobile during the breeding season, undertaking short-term trips (excursions) outside their normal home range. If fertilisation occurs, leading to gene flow of the male genome, this behaviour could be considered a form of breeding dispersal from a genetic point of view. Here, we investigated ranging behaviour of 235 adult roe deer using intensive GPS monitoring in six populations across Europe within the EURODEER initiative. We show that excursions are common from June to August among females, with 41.8 % (vs. 18.1 % of males) making at least one excursion. Most individuals performed only one excursion per season and departure dates for females were concentrated in time, centred on the rutting period, suggesting a link with reproduction. The distance females travelled during excursions was significantly greater than the site-specific average diameter of a male home range, while travel speed decreased once they progressed beyond this diameter, indicating search behaviour or interaction with other male(s) outside the resident male’s territory. Because adults are normally highly sedentary, the potential for mating with relatives is substantial; hence, we conclude that rut excursions could be an alternative tactic enabling females to avoid mating with a closely related male. To understand better the ultimate drivers at play, it will be crucial to explore the genetic causes and consequences of this behaviour.





Similar content being viewed by others
References
Bates D, Maechler M, Bolker B (2011) lme4: Linear mixed-effects models using S4 classes R package, Version 0.999375-39
Berteaux D, Boutin S (2000) Breeding dispersal in female North American red squirrels. Ecology 81:1311–1326. doi:10.1890/0012-9658(2000)081[1311:BDIFNA]2.0.CO;2
Bocci A, Aiello V, Lovari S (2013) Excursion behaviour of female roe deer may depend on density. Behav Processes 97:18–20. doi:10.1016/j.beproc.2013.03.004
Bonnot N, Gaillard JM, Coulon A et al (2010) No difference between the sexes in fine-scale spatial genetic structure of roe deer. PLoS One 5:e14436. doi:10.1371/journal.pone.0014436
Börger L, Franconi N, De Michele G et al (2006) Effects of sampling regime on the mean and variance of home range size estimates. J Anim Ecol 75:1393–1405. doi:10.1111/j.1365-2656.2006.01164.x
Bramley PS (1970) Territoriality and reproductive behavior of roe deer. J Reprod Fertil Suppl 11:43–70
Burnham KP, Anderson DR (1998) Model selection and inference. Springer, New York
Byers JA, Moodie JD, Hall N (1994) Pronghorn females choose vigorous mates. Anim Behav 47:33–43. doi:10.1006/anbe.1994.1005
Calabuig G, Ortego J, Cordero PJ, Aparicio JM (2008) Causes, consequences and mechanisms of breeding dispersal in the colonial lesser kestrel, Falco naumanni. Anim Behav 76:1989–1996. doi:10.1016/j.anbehav.2008.08.019
Calenge C (2006) The package "adehabitat" for R software: a tool for the analysis of space and habitat use by animal. Ecol Model 197:516–519. doi:10.1016/j.ecolmodel.2006.03.017
Clobert J, Danchin E, Dhondt AA, Nichos JD (2001) Dispersal. Oxford University Press, New York
Clutton-Brock TH (1991) The evolution of parental care. Princeton University Press, Princeton
Cohas A, Bonenfant C, Kempenaers B, Allaine D (2009) Age-specific effect of heterozygosity on survival in alpine marmots, Marmota marmota. Mol Ecol 18:1491–1503. doi:10.1111/j.1365-294X.2009.04116.x
Coulon A, Cosson JF, Angibault JM et al (2004) Landscape connectivity influences gene flow in a roe deer population inhabiting a fragmented landscape: an individual-based approach. Mol Ecol 13:2841–2850. doi:10.1111/j.1365-294X.2004.02253.x
Da Silva A, Gaillard JM, Yoccoz NG et al (2009) Heterozygosity-fitness correlations revealed by neutral and candidate gene markers in roe deer from a long-term study. Evolution 63:403–417. doi:10.1111/j.1558-5646.2008.00542.x
Danchin E, Cam E (2002) Can non-breeding be a cost of breeding dispersal? Behav Ecol Sociobiol 51:153–163. doi:10.1007/s00265-001-0423-5
Daniels SJ, Walters JR (2000) Between-year breeding dispersal in Red-cockaded Woodpeckers: multiple causes and estimated cost. Ecology 81:2473–2484
Debeffe L, Morellet N, Cargnelutti B et al (2012) Condition-dependent natal dispersal in a large herbivore: heavier animals show a greater propensity to disperse and travel further. J Anim Ecol 81:1327–1337. doi:10.1111/j.1365-2656.2012.02006.x
Fieberg J, Delgiudice G (2008) Exploring migration data using interval-censored time-to-event models. J Wildl Manag 72:1211–1219. doi:10.2193/2007-403
Fisher DO, Lara MC (1999) Effects of body size and home range on access to mates and paternity in male bridled nailtail wallabies. Anim Behav 58:121–130. doi:10.1006/anbe.1999.1119
Gaillard JM, Sempere AJ, Boutin JM et al (1992) Effects of age and body-weight on the proportion of females breeding in a population of roe deer (Capreolus-capreolus). Can J Zool-Rev Can Zool 70:1541–1545. doi:10.1139/z92-212
Garcia-Navas V, Jose Sanz J (2011) Females call the shots: breeding dispersal and divorce in blue tits. Behav Ecol 22:932–939. doi:10.1093/beheco/arr067
Greenwood PJ (1980) Mating systems, philopatry and dispersal in birds and mammals. Anim Behav 28:1140–1162. doi:10.1016/S0003-3472(80)80103-5
Greenwood PJ, Harvey PH (1982) The natal and breeding dispersal of birds. Annu Rev Ecol Syst 13:1–21. doi:10.1146/annurev.es.13.110182.000245
Hamilton WD (1990) Mate choice near or far. Am Zool 30:341–352. doi:10.1093/icb/30.2.341
Harrington DP, Fleming TR (1982) A class of rank test procedures for censored survival-data. Biometrika 69:553–566. doi:10.1093/biomet/69.3.553
Hewison AJM, Vincent JP, Reby D (1998) Social organisation of European roe deer. The European Roe Deer: the biology of success. Scandinavian University Press, oslo
Hoffman JI, Forcada J, Trathan PN, Amos W (2007) Female fur seals show active choice for males that are heterozygous and unrelated. Nature 445:912–914. doi:10.1038/nature05558
Hoffmann B, Barth D, Karg H (1978) Progesterone and estrogen-levels in peripheral plasma of the pregnant and non-pregnant roe deer (Capreolus-capreolus). Biol Reprod 19:931–935. doi:10.1095/biolreprod19.5.931
Kolodzinski JJ, Tannenbaum LV, Muller LI et al (2010) Excursive behaviors by female white-tailed deer during estrus at two mid-atlantic sites. Am Midl Nat 163:366–373. doi:10.1674/0003-0031-163.2.366
Kurt F, Hartl G, Volk F (1993) Breeding strategies and genetic-variation in European roe deer Capreolus capreolus populations. Acta Theriol (Warsz) 38:187–194
Labisky RF, Fritzen DE (1998) Spatial mobility of breeding female white-tailed deer in a low-density population. J Wildl Manag 62:1329–1334. doi:10.2307/3801998
Lamberti P, Mauri L, Merli E et al (2006) Use of space and habitat selection by roe deer Capreolus capreolus in a Mediterranean coastal area: how does woods landscape affect home range? J Ethol 24:181–188
Liberg O, Johansson A, Andersen R, Linnell JDC (1998) Mating system, mating tactics and the function of male territory in roe deer. In: Andersen R, Duncan P, Linnell J (eds) The European Roe Deer: the biology of success. Scandinavian University Press, Oslo, pp 221–256
Lovari S, Bartolommei P, Meschi F, Pezzo F (2008) Going out to mate: excursion behaviour of female roe deer. Ethology 114:886–896. doi:10.1111/j.1439-0310.2008.01549.x
Lukas D, Clutton-Brock TH (2011) Group structure, kinship, inbreeding risk and habitual female dispersal in plural-breeding mammals. J Evol Biol 24:2624–2630. doi:10.1111/j.1420-9101.2011.02385.x
Marjamaeki PH, Contasti AL, Coulson TN, McLoughlin PD (2013) Local density and group size interacts with age and sex to determine direction and rate of social dispersal in a polygynous mammal. Ecol Evol 3:3073–3082. doi:10.1002/ece3.694
Morellet N, Van Moorter B, Cargnelutti B et al (2011) Landscape composition influences roe deer habitat selection at both home range and landscape scales. Landsc Ecol 26:999–1010. doi:10.1007/s10980-011-9624-0
Morton NE, Crow JF, Muller HJ (1956) An estimate of the mutational damage in man from data on consanguineous marriages. Proc Natl Acad Sci USA 42:855–863. doi:10.1073/pnas.42.11.855
Naguib M, Altenkamp R, Griessmann B (2001) Nightingales in space: song and extra-territorial forays of radio tagged song birds. J Ornithol 142:306–312. doi:10.1046/j.1439-0361.2001.01005.x
Nicol SC, Vanpe C, Sprent J et al (2011) Spatial ecology of a ubiquitous Australian anteater, the short-beaked echidna (Tachyglossus aculeatus). J Mammal 92:101–110. doi:10.1644/09-MAMM-A-398.1
Paradis E, Baillie SR, Sutherland WJ, Gregory RD (1998) Patterns of natal and breeding dispersal in birds. J Anim Ecol 67:518–536. doi:10.1046/j.1365-2656.1998.00215.x
Pedersen MC, Dunn PO, Whittingham LA (2006) Extraterritorial forays are related to a male ornamental trait in the common yellowthroat. Anim Behav 72:479–486. doi:10.1016/j.anbehav.2006.02.010
Pusey A, Wolf M (1996) Inbreeding avoidance in animals. Trends Ecol Evol 11:201–206. doi:10.1016/0169-5347(96)10028-8
R Development Core Team (2010) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Ratcliffe PR, Mayle B (1992) Roe deer biology and management. For Commision Bull 105:1–28
Richard E, Morellet N, Cargnelutti B et al (2008) Ranging behaviour and excursions of female roe deer during the rut. Behav Processes 79:28–35. doi:10.1016/j.beproc.2008.04.008
Ronce O (2007) How does it feel to be like a rolling stone? Ten questions about dispersal evolution. Annu Rev Ecol Evol Syst 38:231–253. doi:10.1146/annurev.ecolsys.38.091206.095611
Sempéré AJ, Mauget R, Mauget C (1998) Reproductive physiology of roe deer. In: Andersen R, Duncan P, Linnell J (eds) The European Roe deer: the biology of success. Scandinavian University Press, Oslo, pp 161–188
Sillero-Zubiri C, Gottelli D, Macdonald DW (1996) Male philopatry, extra-pack copulations and inbreeding avoidance in Ethiopian wolves (Canis simensis). Behav Ecol Sociobiol 38:331–340. doi:10.1007/s002650050249
Soulsbury CD, Iossa G, Baker PJ et al (2011) Behavioral and spatial analysis of extraterritorial movements in red foxes (Vulpes vulpes). J Mammal 92:190–199. doi:10.1644/09-MAMM-A-187.1
Stopher KV, Nussey DH, Clutton-Brock TH et al (2011) The red deer rut revisited: female excursions but no evidence females move to mate with preferred males. Behav Ecol 22:808–818. doi:10.1093/beheco/arr052
Therneau T, Lumley T (2010) survival: Survival analysis, including penalised likelihood R package, Version 2.36-2
Urbano F, Cagnacci F (2014) Spatial database for GPS widlife tracking data. A practical guide to creating a data management system with PostgreSQL/PostGIS and R. Springer, Berlin
Vanpé C, Gaillard JM, Kjellander P et al (2007) Antler size provides an honest signal of male phenotypic quality in roe deer. Am Nat 169:481–493. doi:10.1086/512046
Vanpé C, Kjellander P, Galan M et al (2008) Mating system, sexual dimorphism, and the opportunity for sexual selection in a territorial ungulate. Behav Ecol 19:309–316. doi:10.1093/beheco/arm132
Vanpé C, Gaillard JM, Morellet N et al (2009a) Age-specific variation in male breeding success of a territorial ungulate species, the European roe deer. J Mammal 90:661–665. doi:10.1644/08-MAMM-A-137R.1
Vanpé C, Kjellander P, Gaillard JM et al (2009b) Multiple paternity occurs with low frequency in the territorial roe deer, Capreolus capreolus. Biol J Linn Soc 97:128–139. doi:10.1111/j.1095-8312.2009.01196.x
Vanpé C, Morellet N, Kjellander P et al (2009c) Access to mates in a territorial ungulate is determined by the size of a male’s territory, but not by its habitat quality. J Anim Ecol 78:42–51. doi:10.1111/j.1365-2656.2008.01467.x
Vanpé C, Gaillard JM, Kjellander P et al (2010) Assessing the intensity of sexual selection on male body mass and antler length in roe deer Capreolus capreolus: is bigger better in a weakly dimorphic species? Oikos 119:1484–1492. doi:10.1111/j.1600-0706.2010.18312.x
Wahlstrom LK (1994) The significance of male male-aggression for yearling dispersal in roe reer (Capreolus-capreolus). Behav Ecol Sociobiol 35:409–412. doi:10.1007/BF00165843
Winters JB, Waser PM (2003) Gene dispersal and outbreeding in a philopatric mammal. Mol Ecol 12:2251–2259. doi:10.1046/j.1365-294X.2003.01896.x
Wood SN (2011) Fast stable restricted maximum likelihood and marginal likelihood estimation of semiparametric generalized linear models. J R Stat Soc Ser B-Stat Methodol 73:3–36
Worton BJ (1989) Kernel methods for estimating the utilization distribution in home-range studies. Ecology 70:164–168. doi:10.2307/1938423
Zeh JA, Zeh DW (1996) The evolution of polyandry I: Intragenomic conflict and genetic incompatibility. Proc R Soc Lond B Biol Sci 263:1711–1717. doi:10.1098/rspb.1996.0250
Acknowledgments
This paper was conceived and written within the EURODEER collaborative project (paper 3 of the EURODEER series; www.eurodeer.org). Co-authors are grateful to all members for their support for the initiative. We also thank two anonymous referees and the handling editor for constructive comments on an earlier version of this paper. The EURODEER spatial database is hosted by the Edmund Mach Foundation. GPS data collection of the Edmund Mach Foundation was supported by the Autonomous Province of Trento under Grant N. 3479 to F. Cagnacci (BECOCERWI–Behavioural Ecology of Cervids in Relation to Wildlife Infections). F. Cagnacci thanks the Wildlife and Forest Service of the Autonomous Province of Trento and the Hunting Association of Trento Province (A.C.T.) for support and help during captures. Financial support for GPS data collection in the Bavarian Forest was provided by the EU-programme INTERREG IV (EFRE Ziel 3) and the Bavarian Forest National Park Administration. The Swedish study was supported by grants from the private foundation of “Marie Claire Cronstedts Minne”, The Swedish Environmental Protection Agency and The Swedish Association for Hunting and Wildlife Management. M. Hewison and N. Morellet would like to thank the local hunting associations, the Fédération Départementale des Chasseurs de la Haute Garonne, as well as numerous co-workers and volunteers for their assistance and, in particular, B. Cargnelutti, J.M. Angibault, B. Lourtet, D. Picot and J. Merlet. The France South study was partly funded by the INDHET ANR grant (ANR-12-BSV7-0023-02). C. Vanpé and L. Debeffe were supported by the PATCH RPDOC ANR project (ANR-12-PDOC-0017-01) attributed to CV from the French National Research Agency (PATCH project). The Norwegian study was financed by the Research Council of Norway, the Directorate for Nature Management, and the county governor’s office in Buskerud county. The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Göran C. Ericsson.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Debeffe, L., Focardi, S., Bonenfant, C. et al. A one night stand? Reproductive excursions of female roe deer as a breeding dispersal tactic. Oecologia 176, 431–443 (2014). https://doi.org/10.1007/s00442-014-3021-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-014-3021-8