Abstract
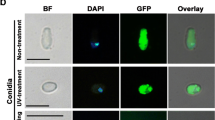
Heterotrimeric G-proteins play key roles in the transduction of extracellular signals to various downstream effectors in eukaryotes. In our previous study, a T-DNA insertional mutant A1-412, in which the promoter of a putative Gγ subunit gene MGG1 was disrupted, was impaired in asexual/sexual sporulation, appressorium formation, and pathogenicity in Magnaporthe oryzae. Here the roles of MGG1 in regulating fungal development and plant infection were further investigated and verified using a gene deletion strategy. Targeted gene deletion mutants of MGG1 exhibited similar phenotypes to those of A1-412. The Δmgg1 mutants were unable to differentiate appressorium on hydrophobic surfaces and nonpathogenic to susceptible hosts. The defects of the Δmgg1 mutants in appressorium formation were partially restored by adding exogenous cAMP or IBMX (a phosphodiesterase inhibitor), although the induced appressoria were still nonfunctional. Expressing Mgg1-GFP fusion protein in an Δmgg1 mutant could complement all phenotypes of the mutant, and bright GFP fluorescence was observed at the periphery of fungal cells, indicating that Mgg1 mainly localizes to plasma membrane. Quantitative RT-PCR analysis revealed that deletion of MGG1 resulted in a significant reduction in mRNA levels of the genes encoding Gα (MagA, MagB, and MagC), Gβ (Mgb1), and adenylate cyclase (Mac1). Moreover, intracellular cAMP accumulation was significantly reduced in Δmgg1 mutants compared to that in the wild-type strain. Taken together, our results suggested that Gγ subunit Mgg1 might act upstream of cAMP signaling pathway and play critical roles in regulation of conidiation, appressorium formation, mating, and plant infection in M. oryzae.






Similar content being viewed by others
References
Choi W, Dean RA (1997) The adenylate cyclase gene MAC1 of Magnaporthe grisea controls appressorium formation and other aspects of growth and development. Plant Cell 9:1973–1983
De Jong JC, McCormack BJ, Smirnoff N, Talbot NJ (1997) Glycerol generates turgor in rice blast. Nature 389:244–245
Dean RA, Talbot NJ, Ebbole DJ, Farman ML, Mitchell TK, Orbach MJ, Thon M, Kulkarni R, Xu JR, Pan H, Read ND, Lee YH, Carbone I, Brown D, Oh YY, Donofrio N, Jeong JS, Soanes DM, Djonovic S, Kolomiets E, Rehmeyer C, Li W, Harding M, Kim S, Lebrun MH, Bohnert H, Coughlan S, Butler J, Calvo S, Ma LJ, Nicol R, Purcell S, Nusbaum C, Galagan JE, Birren BW (2005) The genome sequence of the rice blast fungus Magnaporthe grisea. Nature 434:980–986
DeZwaan TM, Carroll AM, Valent B, Sweigard JA (1999) Magnaporthe grisea Pth11p is a novel plasma membrane protein that mediates appressorium differentiation in response to inductive substrate cues. Plant Cell 11:2013–2030
Ebbole DJ (2007) Magnaporthe as a model for understanding host-pathogen interactions. Annu Rev Phytopathol 45:437–456
Gummer JP, Trengove RD, Oliver RP, Solomon PS (2012) A comparative analysis of the heterotrimeric G-protein Gα, Gβ and Gγ subunits in the wheat pathogen Stagonospora nodorum. BMC Microbiol 12:131
Hirschman JE, Jenness DD (1999) Dual lipid modification of the yeast Gγ subunit Ste18p determines membrane localization of Gβγ. Mol Cell Biol 19:7705–7711
Howard RJ, Valent B (1996) Breaking and entering-host penetration by the fungal rice blast pathogen Magnaporthe grisea. Annu Rev Microbiol 50:491–512
Howard RJ, Ferrari MA, Roach DH, Money NP (1991) Penetration of hard substrates by a fungus employing enormous turgor pressures. Proc Natl Acad Sci USA 88:11281–11284
Krystofova S, Borkovich KA (2005) The heterotrimeric G-protein subunits GNG-1 and GNB-1 form a Gβγ dimer required for normal female fertility, asexual development, and Gα protein levels in Neurospora crassa. Eukaryot Cell 4:365–378
Landry S, Hoffman CS (2001) The git5 Gbeta and git11 Ggamma form an atypical Gbetagamma dimer acting in the fission yeast glucose/cAMP pathway. Genetics 157:1159–1168
Lee YH, Dean RA (1993) cAMP regulates infection structure formation in the plant pathogenic fungus Magnaporthe grisea. Plant Cell 5:693–700
Lee YH, Dean RA (1994) Hydrophobicity of contact surface induces appressorium formation of Magnaporthe grisea. FEMS Microbiol Lett 115:71–76
Li L, Wright SJ, Krystofova S, Park G, Borkovich KA (2007) Heterotrimeric G protein signaling in filamentous fungi. Annu Rev Microbiol 61:423–452
Li Y, Yan X, Wang H, Liang S, Ma WB, Fang MY, Talbot NJ, Wang ZY (2010) MoRic8 is a novel component of G-protein signaling during plant infection by the rice blast fungus Magnaporthe oryzae. Mol Plant Microbe Interact 23:317–331
Li L, He L, Lai Y, Shao Y, Chen F (2014) Cloning and functional analysis of the Gβ gene Mgb1 and the Gγ gene Mgg1 in Monascus ruber. J Microbiol 52:35–43
Liang S, Wang ZY, Liu PJ, Li DB (2006) A Gγ subunit promoter T-DNA insertion mutant A1-412 of Magnaporthe grisea is defective in appressorium formation, penetration and pathogenicity. Chin Sci Bull 51:2214–2218
Liu S, Dean RA (1997) G protein alpha subunit genes control growth, development, and pathogenicity of Magnaporthe grisea. Mol Plant Microbe Interact 10:1075–1086
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitive PCR and the 2−ΔΔCT method. Methods 25:402–408
Mitchell TK, Dean RA (1995) The cAMP-dependent protein kinase catalytic subunit is required for appressorium formation and pathogenesis by the rice blast pathogen Magnaporthe grisea. Plant Cell 7:1869–1878
Neves SR, Ram PT, Iyengar R (2002) G protein pathways. Science 296:1636–1639
Nishimura M, Park G, Xu JR (2003) The G-beta subunit MGB1 is involved in regulating multiple steps of infection-related morphogenesis in Magnaporthe grisea. Mol Microbiol 50:231–243
Park G, Xue C, Zhao X, Kim Y, Orbach M, Xu JR (2006) Multiple upstream signals converge on the adaptor protein Mst50 in Magnaporthe grisea. Plant Cell 18:2822–2835
Seo JA, Han KH, Yu JH (2005) Multiple roles of a heterotrimeric G protein γ-subunit in governing growth and development of Aspergillus nidulans. Genetics 171:81–89
Shin KS, Kwon NJ, Yu JH (2009) Gβγ-mediated growth and developmental control in Aspergillus fumigatus. Curr Genet 55:631–641
Skamnioti P, Gurr SJ (2007) Magnaporthe grisea cutinase2 mediates appressorium differentiation and host penetration and is required for full virulence. Plant Cell 19:2674–2689
Talbot NJ (2003) On the trail of a cereal killer: exploring the biology of Magnaporthe grisea. Annu Rev Microbiol 57:177–202
Talbot NJ, Ebbole DJ, Hamer JE (1993) Identification and characterization of MPG1, a gene involved in pathogenicity from the rice blast fungus Magnaporthe grisea. Plant Cell 5:1575–1590
Wilson RA, Talbot NJ (2009) Under pressure: investigating the biology of plant infection by Magnaporthe oryzae. Nat Rev Microbiol 7:185–195
Xiao JZ, Watanabe T, Kamakura T, Ohshima A, Yamaguchi I (1994) Studies on cellular differientiation of Magnaporthe grisea. Physicochemical aspects of substratum surfaces in relation of appressorium formation. Physiol Mol Plant Pathol 44:227–236
Xu JR, Hamer JE (1996) MAP kinase and cAMP signaling regulate infection structure formation and pathogenic growth in the rice blast fungus Magnaporthe grisea. Genes Dev 10:2696–2706
Xu JR, Urban M, Sweigard JA, Hamer JE (1997) The CPKA gene of Magnaporthe grisea is essential for appressorial penetration. Mol Plant Microbe Interact 10:187–194
Yang Q, Poole SI, Borkovich KA (2002) A G-protein β subunit required for sexual and vegetative development and maintenance of normal Gα protein levels in Neurospora crassa. Eukaryot Cell 1:378–390
Acknowledgments
This work was supported by National Key Basic Research and Development Program of China (2012CB114002), by Program for Changjiang Scholars and Innovative Research Team in University (IRT0943), and the Natural Science Foundation of China (Grant No. 30670073 and 31370172) to ZYW.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by M. Kupiec.
Rights and permissions
About this article
Cite this article
Li, Y., Que, Y., Liu, Y. et al. The putative Gγ subunit gene MGG1 is required for conidiation, appressorium formation, mating and pathogenicity in Magnaporthe oryzae . Curr Genet 61, 641–651 (2015). https://doi.org/10.1007/s00294-015-0490-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00294-015-0490-1