Abstract
Background
The degree of hepatic fibrosis in biliary atresia (BA) correlates with the prognosis of the disease and thus, early diagnosis of liver fibrosis is clinically important. Liver biopsy is the gold standard for the evaluation of liver fibrosis, but it is an invasive procedure requiring sedation in children. Therefore, it is desirable to identify a noninvasive method for diagnosis and follow-up of hepatic fibrosis.
Objective
The purpose of this study is to evaluate the possibility of quantifying liver fibrosis in infants by T2 relaxation time measurements.
Materials and methods
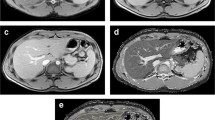
The institutional review board approved this prospective study and parental informed consent was obtained. During MR cholangiopancreatography using a 1.5-T MR scanner in infants with neonatal cholestasis, T2 relaxation time of the liver was calculated with the mean signal intensities measured on images obtained using spin-echo sequences (TR/TE, 2,000/20, 40, 60, 80, 100, 120, 140, 160 ms). A normal control study was performed during spinal MRI in infants with anorectal malformation and normal liver enzyme profiles. A liver biopsy was obtained in the children with cholestasis. The correlation between histopathological fibrosis stage and T2 relaxation time was evaluated by Kendall’s Tau-b test.
Results
Twenty-five infants (male: female, 12:13; age range 0–11 months, mean 3.2 months), 14 with neonatal cholestasis (9 BA and 5 non-BA) and 11 normal controls were included in this study. Relaxation times (mean ± standard deviation [SD]) for the liver were 57.8 ms ± 8.8 in the normal control group (n = 11) and 56.8 ms ± 9.6 in the BA group (n = 9) without statistically significant differences (P = 0.811). T2 relaxation times were not significantly different between the low stage (≤F1) and high stage (≥F2) fibrosis (mean 57.8 vs 56.8; P = 0.934).
Conclusion
T2 relaxation of a normal infant liver at 1.5-T had a mean value of 57.8 ms, which is comparable with adult data (46–57 ms). However, T2 relaxation time was not different in patients with BA and did not correlate with stage of fibrosis.



Similar content being viewed by others
References
Friedman SL (2008) Hepatic fibrosis—overview. Toxicology 254:120–129
Wallace K, Burt AD, Wright MC (2008) Liver fibrosis. Biochem J 411:1–18
Lefkowitch JH (1998) Biliary atresia. Mayo Clin Proc 73:90–95
Hartley JL, Davenport M, Kelly DA (2009) Biliary atresia. Lancet 374:1704–1713
Haafiz AB (2010) Liver fibrosis in biliary atresia. Expert Rev Gastroenterol Hepatol 4:335–343
Oh SH, Kim KM, Kim DY et al (2010) Long-term outcomes of pediatric living donor liver transplantation at a single institution. Pediatr Transplant Jul [Epub ahead of print]
Faria SC, Ganesan K, Mwangi I et al (2009) MR imaging of liver fibrosis: current state of the art. Radiographics 29:1615–1635
Afdhal NH, Nunes D (2004) Evaluation of liver fibrosis: a concise review. Am J Gastroenterol 99:1160–1174
Kreft B, Dombrowski F, Block W et al (1999) Evaluation of different models of experimentally induced liver cirrhosis for MRI research with correlation to histopathologic findings. Invest Radiol 34:360–366
Aube C, Moal F, Oberti F et al (2007) Diagnosis and measurement of liver fibrosis by MRI in bile duct ligated rats. Dig Dis Sci 52:2601–2609
Stark DD, Bass NM, Moss AA et al (1983) Nuclear magnetic resonance imaging of experimentally induced liver disease. Radiology 148:743–751
Stark DD, Goldberg HI, Moss AA et al (1984) Chronic liver disease: evaluation by magnetic resonance. Radiology 150:149–151
Ito K, Mitchell DG, Siegelman ES (2002) Cirrhosis: MR imaging features. Magn Reson Imaging Clin N Am 10:75–92, vi
The French METAVIR Cooperative Study Group (1994) Intraobserver and interobserver variations in liver biopsy interpretation in patients with chronic hepatitis C. Hepatology 20:15–20
Balistreri WF, Grand R, Hoofnagle JH et al (1996) Biliary atresia: current concepts and research directions. Summary of a symposium. Hepatology 23:1682–1692
Pariente D, Franchi-Abella S (2010) Paediatric chronic liver diseases: how to investigate and follow up? Role of imaging in the diagnosis of fibrosis. Pediatr Radiol 40:906–919
Menten R, Leonard A, Clapuyt P et al (2010) Transient elastography in patients with cystic fibrosis. Pediatr Radiol 40:1231–1235
Abe Y, Yamashita Y, Tang Y et al (2000) Calculation of T2 relaxation time from ultrafast single shot sequences for differentiation of liver tumors: comparison of echo-planar, HASTE, and spin-echo sequences. Radiat Med 18:7–14
Cieszanowski A, Szeszkowski W, Golebiowski M et al (2002) Discrimination of benign from malignant hepatic lesions based on their T2-relaxation times calculated from moderately T2-weighted turbo SE sequence. Eur Radiol 12:2273–2279
de Bazelaire CM, Duhamel GD, Rofsky NM et al (2004) MR imaging relaxation times of abdominal and pelvic tissues measured in vivo at 3.0 T: preliminary results. Radiology 230:652–659
Aisen AM, Doi K, Swanson SD (1994) Detection of liver fibrosis with magnetic cross-relaxation. Magn Reson Med 31:551–556
Guibaud L, Lachaud A, Touraine R et al (1998) MR cholangiography in neonates and infants: feasibility and preliminary applications. AJR 170:27–31
Avni FE, Segers V, De Maertelaer V et al (2002) The evaluation by magnetic resonance imaging of hepatic periportal fibrosis in infants with neonatal cholestasis: preliminary report. J Pediatr Surg 37:1128–1133
McCarville MB, Hillenbrand CM, Loeffler RB et al (2010) Comparison of whole liver and small region-of-interest measurements of MRI liver R2* in children with iron overload. Pediatr Radiol 40:1360–1367
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lee, MJ., Kim, MJ., Yoon, CS. et al. Evaluation of liver fibrosis with T2 relaxation time in infants with cholestasis: comparison with normal controls. Pediatr Radiol 41, 350–354 (2011). https://doi.org/10.1007/s00247-010-1874-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00247-010-1874-5