Abstract
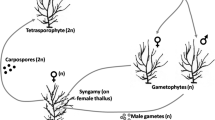
Many organisms survive stressful conditions through a tolerant life history stage. The life history known as the alternation of generations is typical of temperate kelps, producing diploid macroscopic stages, and both haploid and diploid microscopic stages, with the haploid stages thought to be stress tolerant. The survival of microscopic stages of the giant kelp Macrocystis pyrifera during El Niño has been suggested, yet has never been tested. This mechanism could be critical for population persistence, particularly at the southern limit of the range in the Northern Hemisphere, which is greatly impacted by El Niño conditions. The purpose of this study was to determine if microscopic stages of giant kelp could survive and recover from El Niño-type conditions and whether those from a population near its southern limit were more tolerant than a population at the center of its range. Microscopic stages were exposed to a laboratory simulation of potential El Niño conditions (high temperature, with and without light and nitrate) for 8 weeks and then allowed to recover at optimal conditions (low temperature and high nitrate) for 8 weeks, while controls were left at optimal conditions the entire 16 weeks test period. Haploid developmental stages from both populations survived and recovered from stressful conditions with no population level effect, suggesting haploid stress-tolerance may be widespread. The more advanced the developmental stage, and the presence of nitrate, resulted in significantly greater recovery for haploids. Yet, none of these stages were able to go on to produce sporophytes, whereas all controls did. There was a large population-level effect for diploids, however, with only microscopic diploid stages (embryonic sporophytes) from the southern-limit population recovering from El Niño simulated stress, suggesting ecotypic adaptation for microscopic sporophytes. Diploid recovery was significantly greater with light. We propose that the diploid stage is the most likely to survive and recover after El Niño conditions, as it would avoid obligate egg and sperm encounters after the stress period. The survivorship of the microscopic diploid in a seed bank analogue may be how the isolated southern-limit populations are able to recover after mass disappearance during El Niño.



Similar content being viewed by others
References
Bernstein H, Byerly H, Hopf F, Michod R (1985) Genetic damage, mutation, and the evolution of sex. Science 229:1277–1281
Bolton J, Luning K (1982) Optimal growth and maximal survival temperatures of Atlantic Laminaria species in culture. Mar Biol 87:131–135
Chapman ARO (1984) Reproduction, recruitment, and mortality of two species of Laminaria in southwest Nova Scotia. J Exp Mar Biol Ecol 78:99–109
Clarke A (2003). Costs and consequences of evolutionary temperature adaptation. Trends Ecol Evol 18:573–581
Clayton M (1987) Isogamy and a fucalean type of life history in the antarctic brown algae, Ascoseira mirabilis. Botanica Marina 30:447–54
Clayton M (1990) The adaptive significance of life history characters in selected orders of marine brown macroalgae. Aust J Ecol 15:439–452
Cohen CS, Strathman R (1996) Embryos at the edge of tolerance: effects of environmental and structure of egg masses on supply of oxygen to embryos. Biol Bull 190:8–15
Crow JF, Kimura M (1978) Evolution in sexual and asexual populations. Am Nat 909:439–450
Dawson Y (1951) A further study of upwelling and associated vegetation along Pacific Baja California, Mexico. J Mar Res 10:39–58
Dawson Y (1952) Circulation within Bahía Vizcaino, Baja California, and its effects on marine vegetation. Am J Bot 39:425–432
Dayton P (1973) Dispersion and persistence of the annual intertidal alga Postelsia palmaeformis. Ecology 54:433–438
Dayton P (1985) Ecology of kelp communities. Annu Rev Ecol Syst 16:215–45
Dayton P, Tegner M (1984) Catastrophic storms, El Niño, and patch stability in a southern California kelp community. Science 224:283–85
Dean T, Jacobsen FR (1984) Growth of juvenile Macrocystis pyrifera (Laminariales) in relation to environmental factors. Mar Biol 83:301–11
Devinny J, Volse L (1978) Effects of sediments on the development of Macrocystis pyrifera gametophytes. Mar Biol 48:343–348
Deysher L, Dean T (1986a). In situ recruitment of sporophytes of the giant kelp, Macrocystis pyrifera: effects of physical factors. J Exp Mar Biol Ecol 103:41–63
Deysher L, Dean T (1986b) Interactive effect of light and temperature on sporophyte production in the giant kelp, Macrocystis pyrifera. Mar Biol 93:17–20
Durazo R, Baumgartner T (2002) Evolution of oceanographic conditions off Baja California: 1997–1999. Prog Oceanogr 54:7–31
Edwards M (2004) Estimating scale-dependency in disturbance impacts: El Niños and giant kelp forests in the northeast Pacific. Oecologia 138(3):436–447
Espinoza J, Chapman ARO (1983) Ecotypic differentiation of Laminaria longicruris in relation to seawater nitrate concentration. Mar Biol 74:213–218
Fields P, Graham J, Rosenblat H, Somero G (1993) Effects of expected global climate change on marine faunas. Trends Ecol Evol 8:361–3671
Foster M, Schiel D (1985) The ecology of giant kelp forests in California: a community profile. U.S. Fish and wildlife service, Biological Report 85(7.2)
Gerard V (1984) Physiological effects of El Niño on giant kelp in southern California. Mar Biol Letts 5:317–322
Gerard V (1988) Ecotypic differentiation in light related traits of the kelp Laminaria saccharina. Mar Biol 97:25–36
Gerard V (1990) Ecotypic differentiation in the kelp Laminaria saccharina:phase-specific adaptation in a complex life cycle. Mar Biol 107:519–28
Gerard V (1997) The role of nitrogen nutrition in high-temperature tolerance of the kelp Laminaria saccharina (Chromophyta). J Phycol 33:800–810
Gerard V, Dubois K (1988) Temperature ecotypes near the southern boundary of the kelp Laminaria saccharina. Mar Biol 97:575–580
Hernández-Carmona G, Rodríguez-Montesinos Y, Casas-Valdez M, Vilchis M, Sanchez-Rodríguez I (1991) Evaluation of the beds of Macrocystis pyrifera in the Baja California Peninsula, Mexico III. Summer 1986 and seasonal variation. Ciencias Marinas 17:121–145
Hoffman A, Santelices B (1991) Banks of algal microscopic forms: hypotheses on their functioning and comparisons with seed banks. Mar Ecol Prog Ser 79:185–194
Hummel H, Amiard-Triquet C, Bachelet G, Desprez M, Marchand J, Sylvand B, Amiard J, Rybarczyk R, Boogards H, Sinke J, de Wolf L (1996) Sensitivity to stress of the estuarine bivalve Macoma balthica from areas between the Netherlands and its southern limits. J Sea Res 35:315–321
Huyer A, Smith RL (1985) The signature of El Niño off Oregon in 1982–83. J Geophys Res 90:7133–7142
Jackson GA (1977) Nutrients and production of giant kelp, Macrocystis pyrifera, off southern California. Limnol Oceanogr 22:979–995
John D (1994) Alternation of generation in algae: its complexity, maintenance and evolution. Biol Rev 69:275–91
Kahru M, Mitchell G (2000) Influence of the 1997–98 El Niño on the surface chlorophyll in the California current. J Geophys Lett 26:2937–2940
Kinlan B, Graham M, Sala E, Dayton P (2003) Arrested development of giant kelp (Macrocystis pyrifera, Phaeophyceae) embryonic sporophytes: a mechanism for delayed recruitment in perennial kelps. J Phycol 39:47–57
Kopczak C, Zimmerman R, Kremer J (1991) Variation in nitrogen physiology and growth among geographically isolated populations of the giant kelp Macrocystis pyrifera (Phaeophyta). J Phycol 27:149–158
Ladah L (2003) The shoaling of nutrient-enriched subsurface waters as a mechanism to sustain primary productivity off Central Baja California during El Niño winters. J Mar Systems 42:145–152
Ladah L, Zertuche-Gonzalez JA (2004) Giant kelp (Macrocystis pyrifera) survival in deep water (25–40 m) during El Niño of 1997–1998 in Baja California, Mexico. Botanica Marina 47:367–372
Ladah L, Zertuche-Gonzalez JA, Hernandez-Carmona G (1999) Rapid recovery giant kelp (Macrocystis pyrifera, Phaeophyceae) recruitment near its southern limit in Baja California after mass disappearance during ENSO 1997–1998. J Phycol 35:1106–1112
Lee JA, Brinkuis BH (1986) Reproductive phenology of Laminaria saccharina (L.) Lamour (Phaeophyta) at the southern limit of its distribution in the northwestern Atlantic Ocean. J Phycol 22:276–285
Lewis WM (1985) Nutrient scarcity as an evolutionary cause of haploidy. Am Nat 125:692–701
Lubchenco J, Cubit J (1980) Heteromorphic life histories of certain marine algae as adaptations to variations in herbivory. Ecology 64:1116–23
Lynn R, Simpson J (1987) The California current system: the seasonal variability of its physical characteristics. J Geophys Res 92:12947–966
Martinez E (1999) Latitudinal differences in thermal tolerance among microscopic sporophytes of the kelp Lessonia nigrescens (Phaeophyta: Laminariales). Pac Sci 53(1):74–81
Martinez E, Santelices B (1998) Selective mortality on haploid and diploid microscopic stages of Lessonia nigrescens Bory (Phaeophyta, Laminariales). J Exp Mar Biol Ecol 229:219–239
Maynard-Smith J (1978) The evolution of sex. Cambridge University Press, Cambridge, pp 222
McGowan J, Cayan D, Dorman L (1998) Climate–ocean variability and ecosystem response in the Northeast Pacific. Science 281:210–217
Miller A, Schneider N (2000) Interdecadal climate regime dynamics in the North Pacific Ocean: theories, observations and ecosystem impacts. Prog Oceanogr 47:355–379
North W (1994) Review of Macrocystis biology. In: Akatsuka I (ed) Biology of economic algae. Academic Publishing, Netherlands, pp 447–527
North W, Zimmerman R (1984) Influences of macronutrients and water temperatures on summertime survival of Macrocystis canopies. Hydrobiología 116/117:419–24
Ott F (1965) Synthetic media and techniques for the xenic culture of marine algae and flagellates. VA J Sci (N.S.)
Palacios-Hernandez E, Argote-Espinosa M, Amador-Buenrostro A, Mancilla-Peraza M (1996) Simulación de la circulación barotropica inducida por viento en Bahía Sebastián Vizcaíno, B.C. Atmosfera 9:171–188
Perrot V, Richerd S, Valero M (1991) Transition from haploidy to diploidy. Nature 351:315–317
Portner H (2002) Climate variations and the physiological basis of temperature dependant biogeography: systemic to molecular hierarchy of thermal tolerance in animals. Comp Biochem Physiol A Mol Integr Physiol 133:303–321
Provasoli L (1968) Media and prospects for the cultivation of marine algae. In: Watanabe A, A. Hattori (eds) Cultures and collections of algae. Proc US–Japan Conf. Hakone. Sept 1966. Japanese Society of Plant Physiology 63–75
Reed D, Neushul M, Ebeling A (1991) Role of density on gametophyte growth and reproduction in the kelps Macrocystis pyrifera and Pterygophora californica. J Phycol 27:361–366
Reed D, Anderson T, Ebeling A, Anghera M (1997) The role of reproductive synchrony in the colonization potential of kelp. Ecology 78(8):2443–2457
Sagarin R, Barry J, Gilman S, Baxter C (1999) Climate-related change in an intertidal community over short and long time scales. Ecol Monogr 69:465–490
Santelices B (1990) Patterns of reproduction, dispersal, and recruitment in seaweeds. Oceanogr Mar Biol Annu Rev 28:177–276
Santelices B, Hoffman A, Aedo D, Bobadilla M, Otaiza R (1995) A bank of microscopic forms on disturbed boulders and stones in tide pools. Mar Ecol Prog Ser 129:215–228
Scagel R, Bandoni R, Maze J, Rouse R, Scholfield G, Stein J (1982) Nonvascular plants. An evolutionary survey. Wadsworth publishing Company, Belmont
Seymour R, Tegner M, Dayton P, Parnell P (1989) Storm wave induced mortality of giant kelp, Macrocystis pyrifera, in southern California. Estuar Coast Shelf Sci 28:277–292
Tegner M, Dayton P (1987) El Niño effects on southern California kelp communities. Adv Ecol Res 17:243–279
Tegner M, Dayton P (1991) Sea urchins, El Niño’s, and long term stability of southern California kelp forest communities. Mar Ecol Prog Ser 77:49–63
Tegner M, Dayton P, Edwards P, Riser K (1996) Is there evidence for long-term climatic change in Southern California kelp forests? Calcofi Reports 37:111–126
tom Dieck I (1993) Temperature tolerance and survival in darkness of kelp gametophytes (Laminariales, Phaeophyta): ecological and biogeographic implications. Mar Ecol Prog Ser 100:253–264
Trowbridge CD (1994) Life at the edge: population dynamics and salinity tolerance of a high intertidal, pool-dwelling ascoglossan opisthobranch on New Zealand rocky shores. J Exp Mar Biol Ecol 182:65–84
Vermeij GJ (1980) Biogeography and adaptation––patterns of marine life. Harvard University Press, Cambridge, p 332
Zimmerman R, Kremer J (1984) Episodic nutrient supply to a kelp forest ecosystem in Southern California. J Mar Res 42:591–604
Zimmerman R, Robertson DL (1985) Effects of El Niño on local hydrography and growth of giant kelp Macrocystis pyrifera, at Santa Catalina Island, California. Limnol Oceanogr 30:1298–1302
Acknowledgements
The companies of Productos del Pacifico and Abulones Cultivados, the Fishing Cooperative in Bahia Tortugas, and the Fishing Cooperative Estaban Cantu assisted with field research logistics and permits. Diving would not have been possible without assistance from J. Guzman and R. Bermudez. Marco Aurelio provided laboratory assistance. The authors acknowledge funding and support from the UABC (4078-22) and SIMACCONACYT (970106007) grants to J.A.Z., and UC MEXUSCONACYT awards, IAI-EPCOR (CRN-062), AMELIS-CONACYT (J37689/V), and SEP-CONACYT (J50046) research grants to L.B.L. Special thanks to Dr. C. Barilloti, Dr. A. Cabello, and P. Chagoya.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by P. W. Sammarco.
Rights and permissions
About this article
Cite this article
Ladah, L.B., Zertuche-González, J.A. Survival of microscopic stages of a perennial kelp (Macrocystis pyrifera) from the center and the southern extreme of its range in the Northern Hemisphere after exposure to simulated El Niño stress. Mar Biol 152, 677–686 (2007). https://doi.org/10.1007/s00227-007-0723-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00227-007-0723-z