Abstract
X-ray chemical element imaging has the potential to enable fundamental breakthroughs in the understanding of biological systems because chemical element interactions with organelles can be studied at the sub-cellular level. What is the distribution of trace metals in cells? Do some elements accumulate within sub-cellular organelles? What are the chemical species of the elements in these organelles? These are some of the fundamental questions that can be addressed by use of X-ray chemical element imaging with synchrotron radiation beams. For precise location of the distribution of the elements, identification of cellular organelles is required; this can be achieved, after appropriate labelling, by use of fluorescence microscopy. As will be discussed, this approach imposes some limitations on sample preparation. For example, standard immunolabelling procedures strongly modify the distribution of the elements in cells as a result of the chemical fixation and permeabilization steps. Organelle location can, however, be performed, by use of a variety of specific fluorescent dyes or fluorescent proteins, on living cells before cryogenic fixation, enabling preservation of element distribution. This article reviews the methods used for fluorescent organelle labelling and X-ray chemical element imaging and speciation of single cells. Selected cases from our work and from other research groups are presented to illustrate the potential of the combination of the two techniques.

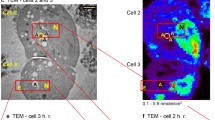
Synchrotron X-ray fluorescence distribution maps of Ca, P and S in yeast cells. Elemental distribution maps (green color scale) were combined with the image of vacuoles labeled with Arg-CMAC (red color scale). The yellow signal of superposed images shows that Ca and P are preferentially located within the vacuole.








Similar content being viewed by others
References
McRae R, Bagchi P, Sumalekshmy S, Fahrni CJ (2009) In situ imaging of metals in cells and tissues. Chem Rev 109(10):4780–4827
Ortega R, Devès G, Carmona A (2009) Bio-metals imaging and speciation in cells using proton and synchrotron radiation X-ray micro-spectroscopy. J R Soc Interface 6:S649–S658
Vogt S, Ralle M (2013) Opportunities in multidimensional trace metal imaging: taking copper-associated disease research to the next level. Anal Bioanal Chem 405:1809–1820
Carmona A, Cloetens P, Deves G, Bohic S, Ortega R (2008) Nano-imaging of trace metals by synchrotron X-ray fluorescence into dopaminergic single cells and neurite-like Processes. J Anal At Spectrom 23:1083–1088
Chen S, Deng J, Yuan Y, Flachenecker C, Mak R, Hornberger B, Jin Q, Shu D, Lai B, Maser J, Roehrig C, Paunesku T, Gleber SC, Vine DJ, Finney L, VonOsinski J, Bolbat M, Spink I, Chen Z, Steele J, Trapp D, Irwin J, Feser M, Snyder E, Brister K, Jacobsen C, Woloschak G, Vogt S (2014) The Bionanoprobe: hard X-ray fluorescence nanoprobe with cryogenic capabilities. J Synchrotron Rad 21:66–75
Matsuyama S, Shimura M, Mimura H, Fujii M, Yumoto H, Sano Y, Yabashi M, Nishino Y, Tamasaku K, Ishikawad T, Yamauchie K (2009) Trace element mapping of a single cell using a hard x-ray nanobeam focused by a Kirkpatrick-Baez mirror system. X-Ray Spectrom 38:89–94
Ortega R (2011) Direct speciation analysis of inorganic elements in single cells using X-ray absorption spectroscopy. J Anal At Spectrom 26:23–29
Ortega R, Carmona A, Llorens I, Solari PL (2012) X-ray absorption spectroscopy of biological samples. A tutorial. J Anal At Spectrom 27:2054–2065
Finney L, Mandava S, Ursos L, Zhang W, Rodi D, Vogt S, Legnini D, Maser J, Ikpatt F, Olopade OI, Glesne D (2007) X-ray fluorescence microscopy reveals large-scale relocalization and extracellular translocation of cellular copper during angiogenesis. Proc Natl Acad Sci U S A 104(7):2247–2252
Kim AM, Vogt S, O’Halloran TV, Woodruff TK (2010) Zinc availability regulates exit from meiosis in maturing mammalian oocytes. Nat Chem Biol 6(9):674–681
Twining BS, Baines SB, Fisher NS, Maser J, Vogt S, Jacobsen C, Tovar-Sanchez A, Sañudo-Wilhelmy SA (2003) Quantifying trace elements in individual aquatic protist cells with a synchrotron X-ray fluorescence microprobe. Anal Chem 75(15):3806–3816
Twining BS, Baines SB, Vogt S, de Jonge MD (2008) Exploring ocean biogeochemistry by single-cell microprobe analysis of protist elemental composition. J Eukaryot Microbiol 55(3):151–162
Leonardo T, Farhi E, Boisson AM, Vial J, Cloetens P, Bohic S, Rivasseau C (2014) Determination of elemental distribution in green micro-algae using synchrotron radiation nano X-ray fluorescence (SR-nXRF) and electron microscopy techniques–subcellular localization and quantitative imaging of silver and cobalt uptake by Coccomyxa actinabiotis. Metallomics 6(2):316–329
Roschzttardtz H, Grillet L, Isaure MP, Conéjéro G, Ortega R, Curie C, Mari S (2011) The plant cell nucleolus as a hot spot for iron. J Biol Chem 286(32):27863–27866
McRae R, Lai B, Vogt S, Fahrni C (2006) Correlative microXRF and optical immunofluorescence microscopy of adherent cells labeled with ultrasmall gold particles. J Struct Biol 155:22–29
Bacquart T, Devès G, Carmona A, Tucoulou R, Bohic S, Ortega R (2007) Subcellular speciation analysis of trace element oxidation states using synchrotron radiation micro-X-ray absorption near edge structure. Anal Chem 79(19):7353–7359
Bacquart T, Devès G, Ortega R (2010) Direct speciation analysis of arsenic in sub-cellular compartments using micro-X-ray absorption spectroscopy. Environ Res 110(5):413–416
McRae R, Lai B, Fahrni CJ (2013) Subcellular redistribution and mitotic inheritance of transition metals in proliferating mouse fibroblast cells. Metallomics 5(1):52–61
Carmona A, Roudeau S, Perrin L, Veronesi G, Ortega R (2014) Environmental manganese compounds accumulate as Mn(II) within the Golgi apparatus of dopamine cells: relationship between speciation, subcellular distribution, and cytotoxicity. Metallomics 6(4):822–832
Tivol WF, Briegel A, Jensen GJ (2008) An improved cryogen for plunge freezing. Microsc Microanal 14:375–379
Schwartz CL, Sarbash VI, Ataullakhanov FI, McIntosh JR, Nicastro D (2007) Cryo-fluorescence microscopy facilitates correlations between light and cryo-electron microscopy and reduces the rate of photobleaching. J Microsc 227:98–109
Matsuyama S, Shimura M, Fujii M, Maeshima K, Yumoto H, Mimura H, Sano Y, Yabashi M, Nishino Y, Tamasaku K, Ishizaka Y, Ishikawae T, Yamauchia K (2010) Elemental mapping of frozen-hydrated cells with cryo-scanning X-ray fluorescence microscopy. X-Ray Spectrom 39:260–266
Goldstein M, Watkins S (2008) Immunohistochemistry. Curr Protoc Mol Biol 14:14.6
Hackett MJ, McQuillan JA, El-Assaad F, Aitken JB, Levina A, Cohen DD, Siegele R, Carter EA, Grau GE, Hunt NH, Lay PA (2011) Chemical alterations to murine brain tissue induced by formalin fixation: implications for biospectroscopic imaging and mapping studies of disease pathogenesis. Analyst 136(14):2941–2952
Carmona A, Deves G, Ortega R (2008) Quantitative micro-analysis of metal ions in subcellular compartments of cultured dopaminergic cells by combination of three ion beam techniques. Anal Bioanal Chem 390(6):1585–1594
Chu WK, Mayer JW, Nicolet MA (1978) Backscattering spectrometry. Academic Press, New York
Hudder A, Nathanson L, Deutscher MP (2003) Organization of mammalian cytoplasm. Mol Cell Biol 23:9318–9326
Carmona A, Devès G, Roudeau S, Cloetens P, Bohic S, Ortega R (2010) Manganese accumulates within Golgi apparatus in dopaminergic cells as revealed by synchrotron X-Ray fluorescence nano-imaging. ACS Chem Neurosci 1(3):194–203
Yang L, McRae R, Henary MM, Patel R, Lai B, Vogt S, Fahrni CJ (2005) Imaging of the intracellular topography of copper with a fluorescent sensor and by synchrotron x-ray fluorescence microscopy. Proc Natl Acad Sci U S A 102(32):11179–11184
Life Technologies (2010) The Molecular probes® handbook-a guide to fluorescent probes and labelling technologies. 11th edn
Yousif LF, Stewart KM, Kelley SO (2009) Targeting mitochondria with organelle-specific compounds: strategies and applications. Chembiochem 10(12):1939–1950
Portugal J, Waring MJ (1988) Assignment of DNA binding sites for 4′,6-diamidine-2-phenylindole and bisbenzimide (Hoechst 33258). A comparative footprinting study. Biochim Biophys Acta 949(2):158–168
Chen AY, Yu C, Gatto B, Liu LF (1993) DNA minor groove-binding ligands: a different class of mammalian DNA topoisomerase I inhibitors. Proc Natl Acad Sci U S A 90(17):8131–8135
Dunn T, Gable K, Beeler T (1994) Regulation of cellular Ca2+ by yeast vacuoles. J Biol Chem 269(10):7273–7278
Li SC, Kane PM (2009) The yeast lysosome-like vacuole: endpoint and crossroads. Biochim Biophys Acta 1793(4):650–663
Dolman NJ, Kilgore JA, Davidson MW (2013) A review of reagents for fluorescence microscopy of cellular compartments and structures, part I: BacMam labelling and reagents for vesicular structures. Curr Protoc Cytom Chapter 12:Unit 12.30
De Giorgi F, Ahmed Z, Bastianutto C, Brini M, Jouaville LS, Marsault R, Murgia M, Pinton P, Pozzan T, Rizzuto R (1999) Targeting GFP to organelles. Methods Cell Biol 58:75–85
Life Technologies. http://www.lifetechnologies.com/fr/fr/home/life-science/cell-analysis/cell-structure/celllight-ready-to-use-fluorescent-protein-based-reagents.html#table 1 Accessed 12 Feb 2014
Paunesku T, Vogt S, Lai B, Maser J, Stojićević N, Thurn KT, Osipo C, Liu H, Legnini D, Wang Z, Lee C, Woloschak GE (2007) Intracellular distribution of TiO2–DNA oligonucleotide nanoconjugates directed to nucleolus and mitochondria indicates sequence specificity. Nano Lett 7(3):596–601
Paunesku T, Ke T, Dharmakumar R, Mascheri N, Wu A, Lai B, Vogt S, Maser J, Thurn K, Szolc-Kowalska B, Larson A, Bergan RC, Omary R, Li D, Lu ZR, Woloschak GE (2008) Gadolinium-conjugated TiO2–DNA oligonucleotide nanoconjugates show prolonged intracellular retention period and T1-weighted contrast enhancement in magnetic resonance images. Nanomedicine 4(3):201–207
Thurn KT, Paunesku T, Wu A, Brown EM, Lai B, Vogt S, Maser J, Aslam M, Dravid V, Bergan R, Woloschak GE (2009) Labeling TiO2 nanoparticles with dyes for optical fluorescence microscopy and determination of TiO2-DNA nanoconjugate stability. Small 11:1318–1325
Yuan Y, Chen S, Paunesku T, Gleber SC, Liu WC, Doty CB, Mak R, Deng J, Jin Q, Lai B, Brister K, Flachenecker C, Jacobsen C, Vogt S, Woloschak GE (2010) Epidermal growth factor receptor targeted nuclear delivery and high-resolution whole cell X-ray imaging of Fe3O4@TiO2 nanoparticles in cancer cells. ACS Nano 7(12):10502–10517
Delehanty JB, Blanco-Canosa JB, Bradburne CE, Susumu K, Stewart MH, Prasuhn DE, Dawson PE, Medintz IL (2013) Site-specific cellular delivery of quantum dots with chemoselectively-assembled modular peptides. Chem Commun (Camb) 49:7878–7880
Corezzi S, Urbanelli L, Cloetens P, Emiliani C, Helfen L, Bohic S, Elisei F, Fioretto D (2009) Synchrotron-based X-ray fluorescence imaging of human cells labeled with CdSe quantum dots. Anal Biochem 388:33–39
Faas FG, Bárcena M, Agronskaia AV, Gerritsen HC, Moscicka KB, Diebolder CA, van Driel LF, Limpens RW, Bos E, Ravelli RB, Koning RI, Koster AJ (2013) Localization of fluorescently labeled structures in frozen-hydrated samples using integrated light electron microscopy. J Struct Biol 181:283–290
Schorb M, Briggs JA (2014) Correlated cryo-fluorescence and cryo-electron microscopy with high spatial precision and improved sensitivity. Ultramicroscopy 143:24–32
McDermott G, Le GMA, Knoechel CG, Uchida M, Larabell CA (2009) Soft X-ray tomography and cryogenic light microscopy: the cool combination in cellular imaging. Trends Cell Biol 19:587–595
Hagen C, Guttmann P, Klupp B, Werner S, Rehbein S, Mettenleiter TC, Schneider G, Grünewald K (2012) Correlative VIS-fluorescence and soft X-ray cryo-microscopy/tomography of adherent cells. J Struct Biol 177:193–201
Duke EM, Razi M, Weston A, Guttmann P, Werner S, Henzler K, Schneider G, Tooze SA, Collinson LM (2014) Imaging endosomes and autophagosomes in whole mammalian cells using correlative cryo-fluorescence and cryo-soft X-ray microscopy (cryo-CLXM). Ultramicroscopy 143:77–87
Maser J, Osanna A, Wang Y, Jacobsen C, Kirz J, Spector S, Winn B, Tennant D (2000) Soft X-ray microscopy with a cryo scanning transmission X-ray microscope: I. Instrumentation, imaging and spectroscopy. J Microsc 197:68–79
Schneider G, Guttmann P, Heim S, Rehbein S, Mueller F, Nagashima K, Heymann JB, Müller WG, McNally JG (2010) Three-dimensional cellular ultrastructure resolved by X-ray microscopy. Nat Methods 7:985–987
Kosior E, Bohic S, Suhonen H, Ortega R, Devès G, Carmona A, Marchi F, Guillet JF, Cloetens P (2012) Combined use of hard X-ray phase contrast imaging and X-ray fluorescence microscopy for subcellular metal quantification. J Struct Biol 177(2):239–247
Acknowledgments
The authors thank the guest editors N. Jakubowski and P. Dittrich for their kind invitation to publish in this special issue dedicated to “Single Cell Analysis”. In their research the authors have benefited from access to beamlines ID21 and ID22 at the European Synchrotron Radiation Facility (ESRF), Grenoble, France. We are sincerely grateful to M. Salomé, J. Susini, G. Veronesi, S. Bohic, R. Tucoulou, and P. Cloetens from ESRF and to G. Devès and T. Bacquart from CENBG for providing assistance during the experiments. Some of the developments reported in this review were financially supported by the ANR (Agence Nationale de la Recherche) program PIRIBIO (ANR-09-PIRI-0029-01).
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in the topical collection Single Cell Analysis with guest editors Petra Dittrich and Norbert Jakubowski.
Rights and permissions
About this article
Cite this article
Roudeau, S., Carmona, A., Perrin, L. et al. Correlative organelle fluorescence microscopy and synchrotron X-ray chemical element imaging in single cells. Anal Bioanal Chem 406, 6979–6991 (2014). https://doi.org/10.1007/s00216-014-8004-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-014-8004-4