Abstract
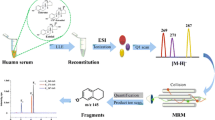
A selective and sensitive liquid chromatography-tandem mass spectrometry method for the determination of very low levonorgestrel (d-(−)-norgestrel) serum levels such as those found in patients using levonorgestrel-releasing intrauterine devices (IUDs) was developed. To achieve the sub-nanomolar sensitivity needed to measure such serum levels, a diethyl ether extraction sample preparation protocol was applied prior to the online solid-phase extraction-liquid chromatography-tandem mass spectrometry (SPE-LC-MS/MS) assay. Analyte quantification from the selected reaction monitoring experiments relied on the use of sixfold deuterated norgestrel as internal standard. The final method was linear up to 1.50 ng/ml with a lower limit of quantification (LLOQ) of 0.05 ng/ml. It was found to be precise and accurate with imprecision <8% and bias <6% assessed at three control levels. Total analyte recovery measured in patient pools at three concentration levels was found to exceed 92%. Matrix interferences were excluded by post-column analyte infusion experiments. As a proof of concept, a set of IUD patient serum samples was screened for their levonorgestrel content. A total of 97.5% (n = 94) of the samples did show serum levels exceeding the LLOQ, proving the applicability of the assay in relevant clinical cohorts. This method must not be used for diagnostic or therapeutic purposes, since it did not undergo formal performance evaluation in the sense of the in vitro diagnostic directive (98/79/EG) of the European community.




Similar content being viewed by others
References
Sidhu J, Job S, Singh S, Philipson R (2006) The pharmacokinetic and pharmacodynamic consequences of the co-administration of lamotrigine and a combined oral contraceptive in healthy female subjects. Br J Clin Pharmacol 61:191–199
Patsalos PN, Perucca E (2003) Clinically important drug interactions in epilepsy: interactions between antiepileptic drugs and other drugs. Lancet Neurol 2:473–481
Sabers A, Ohman I, Christensen J, Tomson T (2003) Oral contraceptives reduce lamotrigine plasma levels. Neurology 61:570–571
Concin H, Bösch H, Hintermüller P, Hohlweg T, Mursch-Edlmayr G, Pinnisch B, Schmidl-Amann S, Schulz-Greinwald G, Unterlerchner D, Wagner T, Mattle V, Wildt L, Fiala C (2009) Use of the levonorgestrel-releasing intrauterine system: an Austrian perspective. Curr Opin Obstet Gynecol 21:1–9
Watson TG, Stewart BJ (1988) A sensitive direct radioimmunoassay for assessing D-norgestrel levels in human plasma. Ann Clin Biochem 25:280–287
Berzas JJ, Rodriguez J, Castaneda G (1997) Simultaneous determination of ethinylestradiol and levonorgestrel in oral contraceptives by derivative spectrophotometry. Analyst 122:41–44
Nevado JJB, Flores JR, Penalvo GC (1997) Simultaneous spectrophotometric determination of ethinylestradiol and levonorgestrel by partial least squares and principal component regression multivariate calibration. Anal Chim Acta 340:257–265
Kook K, Gabelnick H, Duncan G (2002) Pharmacokinetics of levonorgestrel 0.75 mg tablets. Contraception 66:73–76
Wu Z, Zang C, Yang C, Zhang X, Wu E (2000) Simultaneous quantitative determination of norgestrel and progesterone in human serum by high-performance liquid chromatography-tandem mass spectrometry with atmospheric pressure chemical ionization. Analyst 125:2201–2205
Wang Q, Wu Z, Wang Y, Luo G, Wu E, Gao X, Li G (2001) Determination of levonorgestrel in human serum by liquid chromatographic-electrospray tandem mass spectrometry. Anal Lett 34:103–112
Theron HB, Coetzee C, Sutherland FCW, Wiesner JL, Swart KJ (2004) Selective and sensitive liquid chromatography-tandem mass spectrometry method for the determination of levonorgestrel in human plasma. J Chromatogr B 813:311–336
Licea-Perez H, Wang S, Bowen CL, Yang E (2007) A semi-automated 96-well plate method for the simultaneous determination of oral contraceptives in human plasma using ultra performance liquid chromatography coupled with tandem mass spectrometry. J Chromatogr B 852:69–76
Zhao LZ, Zhong GP, Bi HC, Ding L, Deng Y, Guan S, Chen X, Huang ZY, Huang M (2008) Determination of levonorgestrel in human plasma by liquid chromatography-tandem mass spectrometry method: application to a bioequivalence study of two formulations in healthy volunteers. Biomed Chromatogr 22:519–526
Liu F, Xu Y, Liu A, Xu F, Hu W, Guo Q (2008) LC-Tandem-MS Validation for quantitative analysis of levonorgestrel in human plasma. Chromatographia 68:707–712
Lòpez de Alda MJ, Barcelo D (2001) Review of analytical methods for the determination of estrogenes and progesterones in wastewaters. Fresenius J Anal Chem 371:437–447
Díaz-Cruz MS, Lopez de Alda MJ, Lopez R, Barcelo D (2003) Determination of estrogenes and progesterones by mass spectrometric techniques (GC/MS, LC/MC and LC/MS/MS). J Mass Spectrom 38:917–923
Kuch HM, Ballschmiter K (2000) Determination of endogenous and exogenous estrogens in effluents from sewage treatment plants at the ng/L-level. Fresenius J Anal Chem 366:392–395
Matejicek D, Kuban V (2007) High performance liquid chromatography/ion-trap mass spectrometry for separation and simultaneous determination of ethynylestradiol, gestodene, levonorgestrel, cyproterone acetate and desogestrel. Anal Chim Acta 588:304–315
Seger C, Tentschert K, Stöggl W, Griesmacher A, Ramsay SL (2009) A rapid HPLC-MS/MS method for the simultaneous quantification of cyclosporine A, tacrolimus, sirolimus and everolimus in human blood samples. Nat Protoc 4:526–534
U.S. Department of Health and Human Services, Food and Drug Administration, Center for Drug Evaluation and Research (CDER), Center for Veterinary Medicine (CVM). http://www.fda.gov/downloads/Drugs/GuidanceComplianceRegulatoryInformation/Guidances/UCM070107.pdf. Accessed online 10th January 2011
Matuszewski BK, Constanzer ML, Chavez-Eng CM (2003) Strategies for the assessment of matrix effect in quantitative bioanalytical methods based on HPLC-MS/MS. Anal Chem 75:3019–3030
Official Journal of the European Communities L 331, 7.12.1998, p. 1–37. http://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=CELEX:31998L0079:EN:NOT. Accessed online 10th January 2011
Delatour T, Mottier P, Gremaud E (2007) Limits of suspicion, recognition and confirmation as concepts that account for the confirmation transitions at the detection limit for quantification by liquid chromatography–tandem mass spectrometry. J Chromatogr B 1169:103–110
Acknowledgment
The authors thank Gernot Franz and Hartwig Pfisterer for excellent technical assistance in the mass spectrometry laboratory.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in the special issue Analytical Sciences in Austria with Guest Editors G. Allmaier, W. Buchberger, and K. Francesconi.
Rights and permissions
About this article
Cite this article
Moser, C., Gschließer, A., Mattle, V. et al. An ultra-sensitive online SPE-LC-MS/MS method for the quantification of levonorgestrel released from intrauterine devices. Anal Bioanal Chem 400, 2655–2662 (2011). https://doi.org/10.1007/s00216-011-4790-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-011-4790-0