Abstract
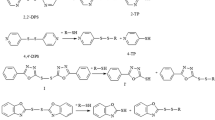
Since its introduction in 1959, Ellman's reagent (5,5′-dithio-bis(2-nitrobenzoic acid)) has been the favorite reagent for spectrophotometric measurement of protein sulfhydryls. Meanwhile however, evidence has accumulated that many protein sulfhydryls give an incomplete reaction with Ellman's reagent, even during prolonged assay times. In the present study, the kinetic problem was solved by including cystamine as a "mediator" between the protein sulfhydryl and Ellman's reagent, as previously applied in an enzymatic thiol assay [9]. As an alternative, 4,4′-dithiodipyridine (DTDP) was used in place of Ellman's reagent. Due to its small size, amphiphilic nature, and lack of charge, DTDP quickly reacts with poorly accessible protein sulfhydryls, without any catalysis by cystamine. The DTDP method and the Ellman/cystamine method were both optimized for maximal sensitivity, minimal sample consumption (detection limit 0.2 nmol mL–1, determination limit 0.6 nmol mL–1), and minimal assay time (5 min). In validation experiments, both methods gave identical results and the measured sulfhydryls/protein matched the expected values. Electronic supplementary material to this paper can be obtained by using the Springer Link server located at http://dx.doi.org/10.1007/s00216-002-1347-2.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Electronic Publication
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Riener, C.K., Kada, G. & Gruber, H.J. Quick measurement of protein sulfhydryls with Ellman's reagent and with 4,4′-dithiodipyridine. Anal Bioanal Chem 373, 266–276 (2002). https://doi.org/10.1007/s00216-002-1347-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-002-1347-2