Abstract
Rationale
Individuals with fetal alcohol spectrum disorder (FASD) are at increased risk for substance use disorders (SUD). In typically developing individuals, susceptibility to SUD is associated with alterations in dopamine and hypothalamic-pituitary-adrenal (HPA) systems, and their interactions. Prenatal alcohol exposure (PAE) alters dopamine and HPA systems, yet effects of PAE on dopamine-HPA interactions are unknown. Amphetamine-stress cross-sensitization paradigms were utilized to investigate sensitivity of dopamine and stress (HPA) systems, and their interactions following PAE.
Methods
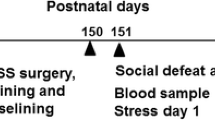
Adult Sprague-Dawley offspring from PAE, pair-fed, and ad libitum-fed control groups were assigned to amphetamine-(1–2 mg/kg) or saline-treated conditions, with injections every other day for 15 days. Fourteen days later, all animals received an amphetamine challenge (1 mg/kg) and 5 days later, hormones were measured under basal or acute stress conditions. Amphetamine sensitization (augmented locomotion, days 1–29) and cross-sensitization with acute restraint stress (increased stress hormones, day 34) were assessed.
Results
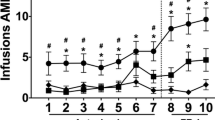
PAE rats exhibited a lower threshold for amphetamine sensitization compared to controls, suggesting enhanced sensitivity of dopaminergic systems to stimulant-induced changes. Cross-sensitization between amphetamine (dopamine) and stress (HPA hormone) systems was evident in PAE, but not in control rats. PAE males exhibited increased dopamine receptor expression (medial prefrontal cortex (mPFC)) compared to controls.
Conclusions
PAE alters induction and expression of sensitization/cross-sensitization, as reflected in locomotor, neural, and endocrine changes, in a manner consistent with increased sensitivity of dopamine and stress systems. These results provide insight into possible mechanisms that could underlie increased prevalence of SUD, as well as the impact of widely prescribed stimulant medications among adolescents with FASD.



Similar content being viewed by others
References
Ahmed SH, Cador M (2006) Dissociation of psychomotor sensitization from compulsive cocaine consumption. Neuropsychopharmacology 31:563–571
Alati R, Clavarino A, Najman JM, O’Callaghan M, Bor W, Mamun AA, Williams GM (2008) The developmental origin of adolescent alcohol use: findings from the Mater University study of pregnancy and its outcomes. Drug Alcohol Depend 98:136–143
Antelman SM, Eichler AJ, Black CA, Kocan D (1980) Interchangeability of stress and amphetamine in sensitization. Science 207:329–331
Badiani A, Robinson TE (2004) Drug-induced neurobehavioral plasticity: the role of environmental context. Behav Pharmacol 15:327–339
Baer JS, Sampson PD, Barr HM, Connor PD, Streissguth AP (2003) A 21-year longitudinal analysis of the effects of prenatal alcohol exposure on young adult drinking. Arch Gen Psychiatry 60:377–385
Barbier E, Houchia H, Warnaulta V, Pierrefichea O, Daousta M, Naassila M (2009) Effects of prenatal and postnatal maternal ethanol on offspring response to alcohol and psychostimulants in long evans rats. Neuroscience 161:427–440
Barr AM, Hofmann CE, Weinberg J, Phillips AG (2002) Exposure to repeated, intermittent d-amphetamine induces sensitization of HPA axis to a subsequent stressor. Neuropsychopharmacology 26:286–294
Becker JB, Molenda H, Hummer DL (2001) Gender differences in the behavioral responses to cocaine and amphetamine. Implications for mechanisms mediating gender differences in drug abuse. Ann N Y Acad Sci 937:172–187
Berman RF, Hannigan JH (2000) Effects of prenatal alcohol exposure on the hippocampus: spatial behavior, electrophysiology, and neuroanatomy. Hippocampus 10:94–110
Blanchard BA, Steindorf S, Wang S, LeFevre R, Mankes RF, Glick SD (1993) Prenatal ethanol exposure alters ethanol-induced dopamine release in nucleus accumbens and striatum in male and female rats. Alcohol Clin Exp Res 17:974–981
Browman KE, Badiani A, Robinson TE (1998) Modulatory effect of environmental stimuli on the susceptibility to amphetamine sensitization: a dose-effect study in rats. J Pharmacol Exp Ther 287:1007–1014
Cabib S, Puglisi-Allegra S (2012) The mesoaccumbens dopamine in coping with stress. Neurosci Biobehav Rev 36:79–89
Capper-Loup C, Canales JJ, Kadaba N, Graybiel AM (2002) Concurrent activation of dopamine D1 and D2 receptors is required to evoke neural and behavioral phenotypes of cocaine sensitization. J Neurosci 22:6218–6227
Carlson JN, Glick SD (1989) Cerebral lateralization as a source of interindividual differences in behavior. Experientia 45:788–798
Carlson JN, Fitzgerald LW, Keller RW Jr, Glick SD (1993) Lateralized changes in prefrontal cortical dopamine activity induced by controllable and uncontrollable stress in the rat. Brain Res 630:178–187
Castner SA, Williams GV (2007) From vice to virtue: insights from sensitization in the nonhuman primate. Prog Neuropsychopharmacol Biol Psychiatry 31:1572–1592
Chen W, Maier S, West J (1997) Prenatal alcohol treatment attenuated postnatal cocaine-induced elevation of dopamine concentration in nucleus accumbens: a preliminary study. Neurotoxicol Teratol 19:39–46
Chotro MG, Arias C, Laviola G (2007) Increased ethanol intake after prenatal ethanol exposure: studies with animals. Neurosci Biobehav Rev 31:181–191
Deroche V, Piazza PV, Maccari S, Le Moal M, Simon H (1992) Repeated corticosterone administration sensitizes the locomotor response to amphetamine. Brain Res 584:309–313
Doig J, McLennan JD, Gibbard WB (2008) Medication effects on symptoms of attention-deficit/hyperactivity disorder in children with fetal alcohol spectrum disorder. J Child Adolesc Psychopharmacol 18:365–371
Gallo PV, Weinberg J (1981) Corticosterone rhythmicity in the rat: interactive effects of dietary restriction and schedule of feeding. J Nutr 111:208–218
Haley DW, Handmaker NS, Lowe J (2006) Infant stress reactivity and prenatal alcohol exposure. Alcohol Clin Exp Res 30:2055–2064
Hannigan JH, Pilati ML (1991) The effects of chronic postweaning amphetamine on rats exposed to alcohol in utero: weight gain and behavior. Neurotoxicol Teratol 13:649–656
Haseltine FP (2000) Gender differences in addiction and recovery. J Womens Health Gend Based Med 9:579–583
Hellemans KG, Verma P, Yoon E, Yu WK, Young AH, Weinberg J (2010) Prenatal alcohol exposure and chronic mild stress differentially alter depressive- and anxiety-like behaviors in male and female offspring. Alcohol Clin Exp Res 34:633–645
Hooks MS, Juncos JL, Justice JB Jr, Meiergerd SM, Povlock SL, Schenk JO, Kalivas PW (1994) Individual locomotor response to novelty predicts selective alterations in D1 and D2 receptors and mRNAs. J Neurosci 14:6144–6152
Hu M, Becker JB (2003) Effects of sex and estrogen on behavioral sensitization to cocaine in rats. J Neurosci 23:693–699
Ikemoto S (2002) Ventral striatal anatomy of locomotor activity induced by cocaine, D-amphetamine, dopamine and D1/D2 agonists. Neuroscience 113:939–955
Jacobson SW, Bihun JT, Chiodo LM (1999) Effects of prenatal alcohol and cocaine exposure on infant cortisol levels. Dev Psychopathol 11:195–208
Kajimoto K, Allan A, Cunningham LA (2013) Fate analysis of adult hippocampal progenitors in a murine model of fetal alcohol spectrum disorder (FASD). PLoS One 8:e73788
Koob GF (2008) A role for brain stress systems in addiction. Neuron 59:11–34
Koob G, Kreek MJ (2007) Stress, dysregulation of drug reward pathways, and the transition to drug dependence. Am J Psychiatry 164:1149–1159
Krieger DT (1974) Food and water restriction shifts corticosterone. Temp Act Brain Amine Periodicity Endocrinol 95:1195–1201
Le Moal M (2009) Drug abuse: vulnerability and transition to addiction. Pharmacopsychiatry 42(Suppl 1):S42–S55
Lee S, Schmidt D, Tilders F, Rivier C (2000) Increased activity of the hypothalamic-pituitary-adrenal axis of rats exposed to alcohol in utero: role of altered pituitary and hypothalamic function. Mol Cell Neurosci 16:515–528
Livy DJ, Miller EK, Maier SE, West JR (2003) Fetal alcohol exposure and temporal vulnerability: effects of binge-like alcohol exposure on the developing rat hippocampus. Neurotoxicol Teratol 25:447–458
Lovallo WR (2006) Cortisol secretion patterns in addiction and addiction risk. Int J Psychophysiol 59:195–202
MacLennan AJ, Maier SF (1983) Coping and the stress-induced potentiation of stimulant stereotypy in the rat. Science 219:1091–1093
McLachlan K, Rasmussen C, Pei J, Reynolds J, Weinberg J (2013) Diurnal cortisol patterns in children with FASD. Alcohol Clin Exp Res 133A: Poster.
Nielsen DM, Crosley KJ, Keller RW Jr, Glick SD, Carlson JN (1999) Rotation, locomotor activity and individual differences in voluntary ethanol consumption. Brain Res 823:80–87
O’Connor MJ, Paley B (2009) Psychiatric conditions associated with prenatal alcohol exposure. Dev Disabil Res Rev 15:225–234
Pacak K, Palkovits M (2001) Stressor specificity of central neuroendocrine responses: implications for stress-related disorders. Endocr Rev 22:502–548
Paxinos G, Watson C (2005) The rat brain in stereotaxic coordinates. Academic Press
Piazza PV, Deminiere JM, le Moal M, Simon H (1990) Stress- and pharmacologically-induced behavioral sensitization increases vulnerability to acquisition of amphetamine self-administration. Brain Res 514:22–26
Ramsay DS, Bendersky MI, Lewis M (1996) Effect of prenatal alcohol and cigarette exposure on two- and six-month-old infants’ adrenocortical reactivity to stress. J Pediatr Psychol 21:833–840
Rasband WS (1997–2011) ImageJ. (Health, N. I. o., ed) Bethesda, Maryland, USA: http://imagej.nih.gov/ij/
Robinson TE, Becker JB (1986) Enduring changes in brain and behavior produced by chronic amphetamine administration: a review and evaluation of animal models of amphetamine psychosis. Brain Res 396:157–198
Sarnyai Z, Shaham Y, Heinrichs SC (2001) The role of corticotropin-releasing factor in drug addiction. Pharmacol Rev 53:209–243
Shen RY, Hannigan JH, Kapatos G (1999) Prenatal ethanol reduces the activity of adult midbrain dopamine neurons. Alcohol Clin Exp Res 23:1801–1807
Shen RY, Choong KC, Thompson AC (2007) Long-term reduction in ventral tegmental area dopamine neuron population activity following repeated stimulant or ethanol treatment. Biol Psychiatry 61:93–100
Shetty A, Burrows R, Phillips D (1993) Alterations in neuronal development in the substantia nigra pars compacta following in utero ethanol exposure: immunohistochemical and Golgi studies. Neuroscience 52:311–322
Sinha R (2008) Chronic stress, drug use, and vulnerability to addiction. Ann N Y Acad Sci 1141:105–130
Sliwowska JH, Barker JM, Barha CK, Lan N, Weinberg J, Galea LA (2010) Stress-induced suppression of hippocampal neurogenesis in adult male rats is altered by prenatal ethanol exposure. Stress 13:301–313
Spear LP (1996) Assessment of the effects of developmental toxicants: pharmacological and stress vulnerability of offspring. NIDA Res Monogr 164:125–145
Sweitzer MM, Donny EC, Hariri AR (2012) Imaging genetics and the neurobiological basis of individual differences in vulnerability to addiction. Drug Alcohol Depend 123(Suppl 1):S59–S71
Taylor AN, Branch BJ, Van Zuylen JE, Redei E (1988) Maternal alcohol consumption and stress responsiveness in offspring. Adv Exp Med Biol 245:311–317
Thanh NX, Jonsson E (2010) Drinking alcohol during pregnancy: evidence from Canadian Community Health Survey 2007/2008. J Popul Ther Clin Pharmacol 17:e302–e307
Uban KA, Sliwowska JH, Lieblich S, Ellis LA, Yu WK, Weinberg J, Galea LAM (2010) Prenatal alcohol exposure reduces the proportion of newly produced neurons and glia in the dentate gyrus of the hippocampus in female rats. Horm Behav 58:835–843
Uban KA, Rummel J, Floresco SB, Galea LA (2012) Estradiol modulates effort-based decision making in female rats. Neuropsychopharmacology 37:390–401
Uban KA, Comeau W, Ellis L, Galea LAM, Weinberg J (2013) Basal regulation of HPA and dopamine systems is altered differentially in males and females by prenatal alcohol exposure and chronic variable stress. PNEC 38:1953–1966
Vanderschuren LJ, Pierce RC (2010) Sensitization processes in drug addiction. Curr Top Behav Neurosci 3:179–195
Volkow ND, Wang GJ, Fowler JS, Logan J, Gatley SJ, Wong C, Hitzemann R, Pappas NR (1999) Reinforcing effects of psychostimulants in humans are associated with increases in brain dopamine and occupancy of D(2) receptors. J Pharmacol Exp Ther 291:409–415
Volkow ND, Fowler JS, Wang GJ, Swanson JM, Telang F (2007) Dopamine in drug abuse and addiction: results of imaging studies and treatment implications. Arch Neurol 64:1575–1579
Volkow ND, Wang GJ, Fowler JS, Tomasi D, Telang F (2011) Addiction: beyond dopamine reward circuitry. Proc Natl Acad Sci U S A 108:15037–15042
Wang J, Haj-Dahmane S, Shen RY (2006) Effects of prenatal ethanol exposure on the excitability of ventral tegmental area dopamine neurons in vitro. J Pharmacol Exp Ther 319:857–863
Wang YC, Wang CC, Lee CC, Huang AC (2010) Effects of single and group housing conditions and alterations in social and physical contexts on amphetamine-induced behavioral sensitization in rats. Neurosci Lett 486:34–37
Weinberg J, Sliwowska JH, Lan N, Hellemans KG (2008) Prenatal alcohol exposure: foetal programming, the hypothalamic-pituitary-adrenal axis and sex differences in outcome. J Neuroendocrinol 20:470–488
Xu C, Shen RY (2001) Amphetamine normalizes the electrical activity of dopamine neurons in the ventral tegmental area following prenatal ethanol exposure. J Pharmacol Exp Ther 297:746–752
Young EA (1998) Sex differences and the HPA axis: implications for psychiatric disease. J Gend Specif Med 1:21–27
Acknowledgments
We would like to thank Linda Ellis who developed the IHC protocol for DA-R expression, Wayne Yu for technical support with RIAs, Nikki Kitay, Farinaz Poursoltani, and Andrew Choe for their valuable assistance with data collection and brain slicing, Stephanie Lieblich for her technical support with IHC, and Dr. Douglas Allan and Luba Veverytsa at the Facility for Synaptic Imaging at the University of British Columbia for training and access to their confocal microscope for imaging of dopamine receptors. This research was funded by grants from the Canadian Foundation for Fetal Alcohol Research (CFFAR) to JW and LAMG, and NIH/NIAAA R37 AA007789 to JW. LAMG is also supported by grants from CIHR, NSERC and Alzheimer’s Society for Canada. KAU was funded by IMPART (CIHR STIHR Training Program) and grant support.
Disclosure statement
No financial support from any individual or corporate body has been received for compensation of work; therefore, the authors declare no potential conflict of interest.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Joanne Weinberg and Liisa A. M. Galea are equal senior authors.
Rights and permissions
About this article
Cite this article
Uban, K.A., Comeau, W.L., Bodnar, T. et al. Amphetamine sensitization and cross-sensitization with acute restraint stress: impact of prenatal alcohol exposure in male and female rats. Psychopharmacology 232, 1705–1716 (2015). https://doi.org/10.1007/s00213-014-3804-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-014-3804-y