Abstract
Rationale
The h5-HT7 receptor is subject to inactivation by risperidone and 9-OH-risperidone, apparently through a pseudo-irreversible complex formed between these drugs and the receptor. Although risperidone and 9-OH-risperidone (“inactivating antagonists”) completely inactivate the receptor, only 50% of the receptors form a pseudo-irreversible complex with these drugs.
Objectives
This study aims to more fully determine the mechanism(s) responsible for the novel effects of risperidone and 9-OH-risperidone and to determine if the inactivation can be reversed (reactivation).
Methods
The ability of non-inactivating drugs (competitive antagonists) to dissociate wash-resistant [3H]risperidone binding from h5-HT7 receptors was investigated. Also, the ability of non-inactivating drugs to reactivate inactivated h5-HT7 receptors was investigated, using cAMP accumulation as a functional endpoint.
Results
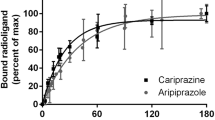
The competitive (non-inactivating) antagonists clozapine and mesulergine released the wash-resistant [3H]risperidone binding to the h5-HT7 receptor. The competitive antagonists clozapine, SB269970, mianserin, cyproheptadine, mesulergine, and ICI169369 reactivated the risperidone-inactivated h5-HT7 receptors in a concentration-dependent manner. The potencies for reactivation closely match the affinities of these drugs for the h5-HT7 receptor (r 2 = 0.95), indicating that the reactivating antagonists are binding to and producing their effects through the orthosteric binding site of the h5-HT7 receptor. Bioluminescence resonance energy transfer analyses indicate that the h5-HT7 receptor forms homodimers.
Conclusions
The ability of the non-inactivating drugs to bind h5-HT7 orthosteric sites and reverse the wash-resistant effects of risperidone or 9-OH-risperidone, also bound to h5-HT7 orthosteric sites, is evidence for protomer–protomer interactions between h5-HT7 homodimers. This is the first demonstration of a non-mutated G-protein-coupled receptor homodimer engaging in protomer–protomer interactions in an intact cell preparation.










Similar content being viewed by others
References
Angers S, Salahpour A, Joly E, Hilairet S, Chelsky D, Dennis M, Bouvier M (2000) Detection of beta 2-adrenergic receptor dimerization in living cells using bioluminescence resonance energy transfer (BRET). Proc Natl Acad Sci USA 97:3684–3689
Ayoub MA, Pfleger KD (2010) Recent advances in bioluminescence resonance energy transfer technologies to study GPCR heteromerization. Curr Opin Pharmacol 10:44–52
Bard JA, Zgombick J, Adham N, Vaysse P, Branchek TA, Weinshank RL (1993) Cloning of a novel human serotonin receptor (5-HT7) positively linked to adenylate cyclase. J Biol Chem 268:23422–23426
Blein S, Hawrot E, Barlow P (2000) The metabotropic GABA receptor: molecular insights and their functional consequences. Cell Mol Life Sci 57:635–650
Bokoch MP, Zou Y, Rasmussen SG, Liu CW, Nygaard R, Rosenbaum DM, Fung JJ, Choi HJ, Thian FS, Kobilka TS, Puglisi JD, Weis WI, Pardo L, Prosser RS, Mueller L, Kobilka BK (2010) Ligand-specific regulation of the extracellular surface of a G-protein-coupled receptor. Nature 463:108–112
Bouvier M (2001) Oligomerization of G-protein-coupled transmitter receptors. Nat Rev Neurosci 2:274–286
Chabre M, Cone R, Saibil H (2003) Biophysics: is rhodopsin dimeric in native retinal rods? Nature 426:30–31
Cheng Y, Prusoff WH (1973) Relationship between the inhibition constant (K1) and the concentration of inhibitor which causes 50 per cent inhibition (I50) of an enzymatic reaction. Biochem Pharmacol 22:3099–3108
Conn PJ, Christopoulos A, Lindsley CW (2009) Allosteric modulators of GPCRs: a novel approach for the treatment of CNS disorders. Nat Rev Drug Discov 8:41–54
Devi LA (2001) Heterodimerization of G-protein-coupled receptors: pharmacology, signaling and trafficking. Trends Pharmacol Sci 22:532–537
Ferre S, Baler R, Bouvier M, Caron MG, Devi LA, Durroux T, Fuxe K, George SR, Javitch JA, Lohse MJ, Mackie K, Milligan G, Pfleger KD, Pin JP, Volkow ND, Waldhoer M, Woods AS, Franco R (2009) Building a new conceptual framework for receptor heteromers. Nat Chem Biol 5:131–134
Fotiadis D, Jastrzebska B, Philippsen A, Muller DJ, Palczewski K, Engel A (2006) Structure of the rhodopsin dimer: a working model for G-protein-coupled receptors. Curr Opin Struct Biol 16:252–259
Gommeren W, Janssen PFM, Schotte A, Leysen JE (1997) Detection of 5HT7 receptors using 3H-risperidone: a brain regional distribution. Psychotic Disorders and Antipsychotics S4:120
Han Y, Moreira IS, Urizar E, Weinstein H, Javitch JA (2009) Allosteric communication between protomers of dopamine class A GPCR dimers modulates activation. Nat Chem Biol 5:688–695
Herrick-Davis K, Grinde E, Harrigan TJ, Mazurkiewicz JE (2005) Inhibition of serotonin 5-hydroxytryptamine2c receptor function through heterodimerization: receptor dimers bind two molecules of ligand and one G-protein. J Biol Chem 280:40144–40151
Herrick-Davis K, Weaver BA, Grinde E, Mazurkiewicz JE (2006) Serotonin 5-HT2C receptor homodimer biogenesis in the endoplasmic reticulum: real-time visualization with confocal fluorescence resonance energy transfer. J Biol Chem 281:27109–27116
Knight JA, Smith C, Toohey N, Klein MT, Teitler M (2009) Pharmacological analysis of the novel, rapid, and potent inactivation of the human 5-hydroxytryptamine7 receptor by risperidone, 9-OH-risperidone, and other inactivating antagonists. Mol Pharmacol 75:374–380
Lovenberg TW, Baron BM, De Lecea L, Miller JD, Prosser RA, Rea MA, Foye PE, Racke M, Slone AL, Siegel BW (1993) A novel adenylyl cyclase-activating serotonin receptor (5-HT7) implicated in the regulation of mammalian circadian rhythms. Neuron 11:449–458
Milligan G, Smith NJ (2007) Allosteric modulation of heterodimeric G-protein-coupled receptors. Trends Pharmacol Sci 28:615–620
Minneman KP (2007) Heterodimerization and surface localization of G protein coupled receptors. Biochem Pharmacol 73:1043–1050
Pfleger KD, Eidne KA (2005) Monitoring the formation of dynamic G-protein-coupled receptor–protein complexes in living cells. Biochem J 385:625–637
Pin JP, Neubig R, Bouvier M, Devi L, Filizola M, Javitch JA, Lohse MJ, Milligan G, Palczewski K, Parmentier M, Spedding M (2007) International union of basic and clinical pharmacology. LXVII. Recommendations for the recognition and nomenclature of G protein-coupled receptor heteromultimers. Pharmacol Rev 59:5–13
Purohit A, Smith C, Herrick-Davis K, Teitler M (2005) Stable expression of constitutively activated mutant h5HT6 and h5HT7 serotonin receptors: inverse agonist activity of antipsychotic drugs. Psychopharmacology (Berl) 179:461–469
Shen Y, Monsma FJ Jr, Metcalf MA, Jose PA, Hamblin MW, Sibley DR (1993) Molecular cloning and expression of a 5-hydroxytryptamine7 serotonin receptor subtype. J Biol Chem 268:18200–18204
Smith C, Rahman T, Toohey N, Mazurkiewicz J, Herrick-Davis K, Teitler M (2006) Risperidone irreversibly binds to and inactivates the h5-HT7 serotonin receptor. Mol Pharmacol 70:1264–1270
Toohey N, Klein MT, Knight J, Smith C, Teitler M (2009) Human 5-HT7 receptor-induced inactivation of forskolin-stimulated adenylate cyclase by risperidone, 9-OH-risperidone and other "inactivating antagonists". Mol Pharmacol 76:552–559
Author information
Authors and Affiliations
Corresponding author
Additional information
This work was supported by the United States Public Health Service [Grant MH56650].
Rights and permissions
About this article
Cite this article
Teitler, M., Toohey, N., Knight, J.A. et al. Clozapine and other competitive antagonists reactivate risperidone-inactivated h5-HT7 receptors: radioligand binding and functional evidence for GPCR homodimer protomer interactions. Psychopharmacology 212, 687–697 (2010). https://doi.org/10.1007/s00213-010-2001-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-010-2001-x