Abstract
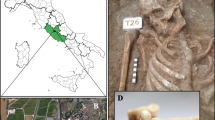
Neanderthals disappeared sometime between 30,000 and 24,000 years ago. Until recently, Neanderthals were understood to have been predominantly meat-eaters; however, a growing body of evidence suggests their diet also included plants. We present the results of a study, in which sequential thermal desorption-gas chromatography-mass spectrometry (TD-GC-MS) and pyrolysis-gas chromatography-mass spectrometry (Py-GC-MS) were combined with morphological analysis of plant microfossils, to identify material entrapped in dental calculus from five Neanderthal individuals from the north Spanish site of El Sidrón. Our results provide the first molecular evidence for inhalation of wood-fire smoke and bitumen or oil shale and ingestion of a range of cooked plant foods. We also offer the first evidence for the use of medicinal plants by a Neanderthal individual. The varied use of plants that we have identified suggests that the Neanderthal occupants of El Sidrón had a sophisticated knowledge of their natural surroundings which included the ability to select and use certain plants.


Similar content being viewed by others
References
Barton RNE (2000) Mousterian hearths and shellfish: late Neanderthal activities in Gibraltar. In: Stringer CB, Barton RNE, Finlayson JC (eds) Neanderthals on the edge: 150th anniversary conference of the Forbes' Quarry discovery, Gibraltar. Oxbow Books, Oxford, pp 211–220
Bergström J (1999) Tobacco smoking and supragingival dental calculus. J Clin Periodontol 26:541–547
Bergström J (2005) Tobacco smoking and subgingival dental calculus. J Clin Periodontol 32:81–88
Blumenschine RJ, Peters CR, Masao FT, Clarke RJ, Deino AL, Hay RL, Swisher CC, Stanistreet IG, Ashley GM, McHenry LJ, Sikes NE, van der Merwe NJ, Tactikos JC, Cushing AE, Deocampo D, Njau JK, Ebert JI (2003) Late Pliocene homo and hominid land use from Western Olduvai Gorge, Tanzania. Science 299:1217–1221
Bocherens H (2009) Neanderthal dietary habits: review of the isotopic evidence. In: Hublin JJ and Richards MP (eds.) The Evolution of Hominin Diets: Integrating Approaches to the Study of Palaeolithic Subsistence. Springer, http://www.springer.com/social+sciences/archaeology+%26+anthropology/book/978-1-4020-9698-3, pp 241–250
Boeda E, Bonilauri S, Connan J, Jarvie D, Mercier N, Tobey M, Valladas H, Al-Sakhel H, Muhesen S (2008) Middle Palaeolithic bitumen use at Umm el Tiel around 70,000 BP. Antiquity 82:853–861
Buckley SA, Stott AW, Evershed RP (1999) Studies of organic residues from ancient Egyptian mummies using high temperature-gas chromatography-mass spectrometry and sequential thermal desorption-gas chromatography-mass spectrometry and pyrolysis-gas chromatography-mass spectrometry. Analyst 124:443–452
Burke A (2000) Hunting in the Middle Palaeolithic. Int J Osteoarchaeol 10:281–285
Charlier P, Huynh-Charlier I, Munoz O, Billard M, Brun L, Grandmaison GLD (2010) The microscopic (optical and SEM) examination of dental calculus deposits (DCD). Potential interest in forensic anthropology of a bio-archaeological method. Legal Medicine 12:163–171
Collins MJ, Copeland L (2011) Ancient starch: cooked or just old? Proc Natl Acad Sci USA 108(22):E145
Collison R (1968) Starch retrogradation. In: Radley JA (ed) Starch and its derivatives. Chapman and Hall Ltd, London, pp 168–193
Connan J (1999) Use and trade of bitumen in antiquity and prehistory: molecular archaeology reveals secrets of past civilisations. Phil Trans Royal Soc B (Biological Sciences) 354:33–50
Copeland L, Blazek J, Salman H, Tang CM (2009) Form and functionality of starch. Food Hydrocolloid 23:1527–1534
Cousins D, Huffman MA (2002) Medicinal properties in the diet of gorillas: an ethnopharmacological evaluation. African Study Monographs 23:65–89
de Torres T, Ortiz JE, Grün R, Eggins S, Valladas H, Mercier N, Tisnérat-Laborde N, Julià R, Soler V, Martínez E, Sánchez-Moral S, Cañaveras JC, Lario J, Lalueza-Fox C, Badal E, Rosas A, Santamaría D, de la Rasilla M, Fortea J (2010) Dating of the hominid (Homo neanderthalensis) remains accumulation from El Sidrón Cave (Piloña, Asturias, North Spain): an example of multi-methodological approach to the dating of Upper Pleistocene sites. Archaeometry 52:680–705
Eglinton G, Hamilton RJ, Raphael RA, Gonzalez AG (1962) Hydrocarbon constituents of the wax coatings of plant leaves: a taxonomic survey. Nature 193:739–742
El Zaatari S, Grine FE, Ungar PS, Hublin JJ (2011) Ecogeographic variation in Neandertal dietary habits: evidence from occlusal molar microwear texture analysis. J Hum Evol 61:411–424
Estalrrich A, Rosas A, García-Vargas S, García-Tabernero A, Santamaría D, de la Rasilla M (2011) Brief communication: subvertical grooves on interproximal wear facets from the El Sidrón (Asturias, Spain) Neanderthal dental sample. Am J Phys Anthropol 144:154–161
Finlayson C, Pacheco FG, Rodríguez-Vidal J, Fa DA, Gutierrez López JM, Pérez AS, Finlayson G, Allue E, Baena Preysler J, Cáceres I, Carrión JS, Fernández Jalvo Y, Gleed-Owen CP, Jimenez Espejo FJ, López P, López Sáez JA, Riquelme Cantal JA, Sánchez Marco A, Giles Guzman FK, Fuentes N, Valarino CV, Villalpando A, Stringer CB, Martinez Ruiz F, Sakamoto T (2006) Late survival of Neanderthals at the southernmost extreme of Europe. Nature 443:850–853
Hardy BL (2010) Climatic variability and plant food distribution in Pleistocene Europe: implications for Neanderthal diet and subsistence. Quat Sci Rev 29:662–679
Hardy K, Blakeney T, Copeland L, Kirkham J, Wrangham R, Collins M (2009) Starch granules, dental calculus and new perspectives on ancient diet. J Archaeol Sci 36:248–255
Hardy K, van de Locht R, Wilson J, Tugay O (2013) Starch granules and complex carbohydrates at Çatalhöyük. In: Hodder I (ed) Humans and landscapes of Çatalhöyük: reports from the 2000-2008 seasons. Los Angeles: Cotsen; Ankara: British Institute of Archaeology at Ankara
Henry AG, Brooks AS, Piperno DR (2011) Microfossils in calculus demonstrate consumption of plants and cooked foods in Neanderthal diets (Shanidar III, Iraq; Spy I and II, Belgium). Proc Natl Acad Sci USA 108:486–491
Hillson S (2001) Recording dental caries in archaeological human remains. Int J Osteoarchaeol 11:249–289
Huffman MA (1997) Current evidence for self-medication in primates: a multidisciplinary perspective. Yearb Phys Anthropol 40:171–200
Huffman MA (2003) Animal self-medication and ethno-medicine: exploration and exploitation of the medicinal properties of plants. Proc Nutr Soc 62:371–381
Huffman MA, Vitazkova SK (2007) Primates, plants, and parasites: the evolution of animal self-medication and ethnomedicine. In Elisabetsky E, Etkin NL (eds.) Ethnopharmacology, e-book http://www.eolss.net, Eolss Publishers, Oxford
Jones M (2009) Moving North: Archaeobotanical Evidence for Plant Diet in Middle and Upper Paleolithic Europe. In Hublin JJ and Richards MP (eds.) The Evolution of Hominin Diets: Integrating Approaches to the Study of Palaeolithic Subsistence. Springer, http://www.springer.com/social+sciences/archaeology+%26+anthropology/book/978-1-4020-9698-3, pp 171–180
Kaplan L, Smith MB, Sneddon LA (1992) Cereal grain phytoliths of southwest Asia and Europe. In Rapp, G Jr, Mulholland SC (eds) Phytolith systematics—emerging issues. Advances in Archaeological Museum Science Vol.1 Plenum Press, New York, pp149–174
Krief S, Hladik CM, Haxaire C (2005) Ethnomedicinal and bioactive properties of plants ingested by wild chimpanzees in Uganda. J Ethnopharmacol 101:1–15
Kruge MK, Suárez-Ruiz NI (1991) Organic geochemistry and petrography of Spanish oil shales. Fuel 70:1298–1302
Laden G, Wrangham R (2005) The rise of the hominids as an adaptive shift in fallback foods: plant underground storage organs (USOs) and australopith origins. J Hum Evol 49:482–498. doi:10.1016/j.jhevol.2005.05.007
Lalueza C, Pérez-Pérez A (1993) The diet of the Neanderthal child Gibralter 2 (Devil's Tower) through the study of the vestibular striation pattern. J Hum Evol 24:29–41
Lalueza-Fox C, Gigli E, de la Rasilla M, Fortea J, Rosas A (2009) Bitter taste perception in Neanderthals through the analysis of the TAS2R38 gene. Biol Letters 5:809–811
Lalueza-Fox C, Rosas A, Estalrrich A, Gigli E, Campos PF, GarcíaTabernero A, García-Vargas S, Sánchez-Quinto F, Ramírez O, Civit S, Bastir M, Huguet R, Santamaría D, Gilbert MPT, Willerslev E, de la Rasilla M (2011) Genetic evidence for patrilocal mating behaviour among Neandertal groups. Proc Natl Acad Sci 108:250–253
Laseter JL, Weete J, Weber DJ (1968) Alkanes, fatty acid methyl esters, and free fatty acids in surface wax of Ustilago maydis. Phytochemistry 7:1177–1181
Lev E, Kislev M, Bar Yosef O (2005) Mousterian vegetal food in Kebara Cave, Mt. Carmel. J Arch Sci 32:475–484
Lieverse AR (1999) Diet and the aetiology of dental calculus. Int J Osteoarchaeol 9:219–232
Lisonbee LD, Villalba JJ, Provenza FD, Hall JO (2003) Tannins and self-medication: implications for sustainable parasite control in herbivores. P Nutr Soc 62:361–370. doi:10.1079/PNS2003243
Madella M, Jones MK, Goldberg P, Goren Y, Hovers E (2002) The exploitation of plant resources by Neanderthals in Amud Cave (Israel): the evidence from Phytolith studies. J Arch Sci 29:703–719
Maudinas B, Villoutreix J (1977) Fatty acid methyl esters in photosynthetic bacteria. Phytochemistry 16:1299–1300
McCobb LME, Briggs DEG, Evershed RP, Hall AR, Hall RA (2001) Preservation of fossil seeds from a 10th Century AD Cess Pit at Coppergate, York. J Arch Sci 28:929–940
Miller G (2011) Sweet here, salty there: evidence for a taste map in the Mammalian Brain. Science 333(6047):1213
O’Connell JF, Hawkes K, Blurton Jones NG (1999) Grandmothering and the evolution of Homo erectus. J Hum Evol 36:461–485
Pap I, Tillier AM, Arensburg B, Weiner SM (1995) First scanning electron microscope analysis of dental calculus from European Neanderthals: Subalyuk, (Middle Palaeolithic, Hungary). Preliminary report. B Soc Ant Paris 7:69–72
Pérez-Pérez A, Espurz V, Bermúdez de Castro JM, de Lumley MA, Turbón D (2003) Non-occlusal dental microwear variability in a sample of Middle and Late Pleistocene human populations from Europe and the Near East. J Hum Evol 44:497–513
Perry GH, Dominy NJ, Claw KG, Lee AS, Fiegler H, Redon R, Werner J, Villanea FA, Mountain JL, Misra R, Carter NP, Lee C, Stone AC (2007) Diet and the evolution of human amylase gene copy number variation. Nat Genet 39:1256–1260
Piperno DR, Dillehay TD (2008) Starch grains on human teeth reveal early broad crop diet in northern Peru. Proc Natl Acad Sci USA 105:19622–19627
Ré-Poppi N, Santiago-Silva MR (2002) Identification of polycyclic aromatic hydrocarbons and methoxylated phenols in wood smoke emitted during production of charcoal. Chromatographia 55:475–481
Richards MP, Trinkaus E (2009) Isotopic evidence for the diets of European Neanderthals and early modern humans. Proc Natl Acad Sci 106:16034–16039
Rodriguez E, Wrangham RW (1993) Zoopharmacognosy: the use of medicinal plants by animals. In: Downum KR, Romeo JT, Stafford H (eds) Recent advances in phytochemistry, vol. 27: phytochemical potential of tropic plants, vol 27. Plenum, New York, pp 89–105
Rogge WF, Hildemann LM, Mazurek MA, Cass GR, Simoneit BRT (1998) Sources of fine organic aerosol 9. Pine, oak, and synthetic log combustion in residential fireplaces. Environ Sci Technol 32:13–22
Rosas A, Martínez-Maza C, Bastir M, García-Tabernero A, Lalueza-Fox C, Huguet R, Ortiz JE, Julià R, Soler V, de Torres T, Martínez E, Cañaveras JC, Sánchez-Moral S, Cueva S, Lario J, Santamaría D, de la Rasilla M, Fortea J (2006) Paleobiology and comparative morphology of a late Neandertal sample from El Sidrón, Asturias, Spain. Proc Natl Acad Sci 103:19266–19271
Rosas A, Estalrrich A, García-Tabernero A, Basti M, García-Vargas S, Sánchez-Meseguer A, Huguet R, Lalueza-Fox C, Peña-Melián A, Kranioti E, Santamaría D, Rasilla de la M, Fortea J. (2012) Les Néandertaliens d’El Sidrón (Asturies, Espagne). Actualisation d’un nouvel échantillon. L’Anthropologie 116:57–76
Sandgathe DM, Hayden B (2003) Did Neanderthals eat inner bark? Antiquity 77:709–718
Scott GR, Poulson SR (2012) Stable carbon and nitrogen isotopes of human dental calculus: a potentially new non-destructive proxy for paleodietary analysis. J Archaeol Sci 39:1338–1393
Seng TH, Tahir NM, Abas MR (2007) Aliphatic and PAHs emissions from open burning of selected tropical woods. Malaysian J Anal Sci 11:36–41
Senter SD, Horvat RJ, Forbus WR (1983) Comparative GLC-MS analysis of phenolic acids of selected tree nuts. J Food Sci 48:798–799
Shorland FB (1962) The comparative aspects of fatty acid occurrence and distribution. In: Florkin M, Mason HS (eds) Comparative biochemistry Vol 3. Academic, New York, pp 1–102
Singer MS, Mace KC, Bernays EA (2009) Self-medication as adaptive plasticity: increased ingestion of plant toxins by parasitized caterpillars. PLoS One 4(3):e4796
Vandermeersch B, Arensburg B, Tillier AM, Rak Y, Weiner S, Spiers M, Aspillaga . (1994). Middle Palaeolithic dental bacteria from Kebara, Israël. C R Acad Sci Paris 319:727–731
Varlet V, Knockaert C, Prost C, Serot T (2006) Comparison of odor-active volatile compounds of fresh and smoked salmon. J Agric Food Chem 54:3391–3401
Williams PFV, Douglas AG (1986) Organic Geochemistry of British Kimmeridge clay 2. Acyclic isoprenoid alkanes in Kimmeridge shale oils. Fuel 65:1728–1734
Wilson J, Hardy K, Allen R, Copeland L, Wrangham R, Collins M (2010) Automated classification of starch granules using supervised pattern recognition of morphological properties. J Archaeol Sci 37:594–604
Wood RE, Higham TFG, de Torres T, Tisnerát-Laborde N, Vallardas H, Ortiz JE, Lalueza-Foz C, Sánchez-Moral S, Cañaveras JC, Rosas A, Santamaría D, de la Rasilla M (2012) A new date for the Neanderthals from El Sidrón Cave (Asturias, Northern Spain). Archaeometry. doi:10.1111/j.1475-4754.2012.00671.x
Wrangham RW, Holland Jones J, Laden G, Pilbeam D, Conklin-Brittain NL (1999) The raw and the stolen. Cooking and the ecology of human origins. Curr Anthropol 40:567–594
Acknowledgments
SEM was conducted at the Imaging and Cytometry Unit, University of York. Stephen Buckley was funded by grant WT074315, TD/Py-GC-MS was conducted in the Department of Civil Engineering and Geosciences Newcastle University; Paul Donohoe and Ian Harrison are thanked for technical assistance. Field work was supported by Consejería de Cultura del Principado de Asturias, and some technical aspects by Ministerio de Ciencia e Innovación, Spain, grant CGL2006-02131. Thanks to Megazyme International for providing the starch assay kit and to William Milliken, Royal Botanic Gardens, Kew, for his confirmation that yarrow and camomile are most likely to have been ingested for medicinal purposes as they have little nutritional value. We thank the anonymous reviewers for their suggestions and we extend our thanks to Dr Joann Fletcher for her many suggested literary improvements.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by: Sven Thatje
Rights and permissions
About this article
Cite this article
Hardy, K., Buckley, S., Collins, M.J. et al. Neanderthal medics? Evidence for food, cooking, and medicinal plants entrapped in dental calculus. Naturwissenschaften 99, 617–626 (2012). https://doi.org/10.1007/s00114-012-0942-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00114-012-0942-0