Abstract
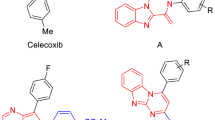
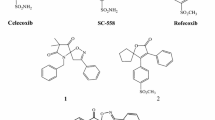
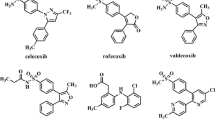
A new group of 2,4,5-triarylimidazole derivatives, possessing a methyl sulfonyl pharmacophore, were synthesized and their biological activities were evaluated for cyclooxygenase-2 (COX-2) inhibitory activity. In vitro COX-1/COX-2 structure–activity relationships were determined by varying the substituents at the para position of C-2 phenyl ring. Among the 2,4,5-triarylimidazoles, 2-(4-hydroxy phenyl)-4-(4-methylsulfonylphenyl)-5-phenyl-1H imidazole (11f) was identified as a selective COX-2 inhibitor (COX-2 IC50 = 0.15 μM; selectivity index = 75) that was less potent than the reference drug celecoxib (COX-2 IC50 = 0.06 μM; SI = 405). A molecular modeling study where 11f was docked in the binding site of COX-2 showed that the methylsulfonyl pharmacophore group is oriented in the vicinity of the COX-2 secondary pocket (Arg513, Phe518, Gly519, and Val523). The structure–activity data acquired indicate that COX-1/COX-2 inhibition is sensitive to the nature of the C-2 phenyl substituents.



Similar content being viewed by others
References
Dannhardt G, Kiefer W (2001) Cyclooxygenase inhibitors-current status and future prospects. Eur J Med Chem 36:109–126
Dogné JM, Supuran CT, Pratico D (2005) Cardiovascular effects of the coxibs. J Med Chem 48:2251–2257
Eberhart CE, Dubois RN (1995) Eicosanoids and the gastrointestinal tract. Gastroenterology 109:285–301
Fu JY, Masferrer JL, Seibert K, Raz A, Needleman P (1990) The induction and suppression of prostaglandin H2 synthase (cyclooxygenase) in human monocytes. J Biol Chem 265:16737–16740
Goodsell DS, Morris GM, Olson AJ (1996) Automated docking of flexible ligands: applications of AutoDock. J Mol Recognit 9:1–5
Ho L, Qin W, Stetka BS, Pasinetti GM (2006) Is there a future for cyclo-oxygenase in Alzheimer’s disease? CNS Drugs 20:85–98
Kanaoka S, Takai T, Yoshida K (2007) Cyclooxygenase-2 and tumor biology. Adv Clin Chem 43:59–78
Katori M, Majima M (2000) Cyclooxygenase-2: its rich diversity of roles and possible application of its selective inhibitors. Inflamm Res 49:367–392
Kawamori T, Rao CV, Seibert K, Reddy BS (1998) Chemopreventive activity of celecoxib, a specific cyclooxygenase-2 inhibitor, against colon carcinogenesis. Cancer Res 58:406–412
Kurumbail RG, Stevens AM, Gierse JK, McDonald JJ, Stegeman RA, Pak JY, Gildehaus D, Miyashiro JM, Penning TD, Seibert K, Isakson PC, Stallings WC (1996) Structural basis for selective inhibition of cyclooxygenase-2 by anti-inflammatory agents. Nature 384:644–648
Liao Z, Mason KA, Milas L (2007) Cyclo-oxygenase-2 and its inhibition in cancer: is there a role? Drug 67:821–845
Penning TD, Tally JJ, Bertenshaw SR, Carter JS, Collins PW, Docter S, Graneto MJ, Lee LF, Malecha JW, Miyashiro JM, Rogers R, Rogier DJ, Yu SS, Anderson GD, Burton EG, Cogburn JN, Gregory SA, Koboldt CM, Perkins WE, Seibert K, Veenhuizen AW, Zhang YY, Isakson PC (1997) Synthesis and biological evaluation of the 1,5-diarylpyrazole class of cyclooxygenase-2 inhibitors: identification of 4-[5-(4-methylphenyl)-3-(trifluoromethyl)-1H pyrazol-1-yl] benzenesulfonamide (SC-b58635, celecoxib). J Med Chem 40:1347–1365
Prasit P, Wang Z, Brideau C, Chan CC, Charlson S, Cromlish W, Ethier D, Evans JF, Ford-Hutchinson AW, Gauthier JY, Gordon R, Guay J, Gresser M, Kargman S, Kennedy B, Leblanc Y, Leger S, Mancini JO, Neil GP, Quellet M, Percival MD, Perrier H, Riendeau D, Rodger I, Tagari P, Therien M, Vikers P, Wong E, Xu L, Young RN, Zamboni R, Boyce S, Rupniak N, Forrest M, Visco D, Patrick D (1999) The discovery of rofecoxib, [MK 966, Vioxx, 4-(4′-methylsulfonylphenyl)-3-phenyl-2(5H)-furanone], an orally active cyclooxygenase-2-inhibitor. Bioorg Med Chem Lett 9:1773–1778
Riendeau D, Percival MD, Brideau C, Dube CS, Ethier D, Falgueyret JP, Friesen RW, Gordon R, Greig G, Guay J, Girard Y, Prasit P, Zamboni R, Rodger IW, Gresser M, Ford-Hutchinson A, Young RN, Chan CC (2002) Etoricoxib (MK-0663): preclinical profile and comparison with other agents that selectively inhibit cyclooxygenase-2. J Pharmacol Exp Ther 296:558–566
Singh SK, Saibaba V, Ravikumar V, Rao CS, Akhila V, Rao YK (2004) Synthesis and biological evaluation of 2,3-diarylpyrazines and quinoxalines as selective COX-2 inhibitors. Bioorg Med Chem 12:1881–1893
Smith WL, DeWitt DL (1996) Prostaglandin endoperoxide H synthases-1 and -2. Adv Immunol 62:167–215
Talley JJ, Bertenshaw SR, Brown DL, Carter JS, Graneto MJ, Kellogg MS, Koboldt CM, Yuan J, Zhang YY, Seibert K (2000a) N-[[(5-Methyl-3-phenyl isoxazol-4-yl)-phenyl]sulfonyl]propanamide, sodium salt, parecoxib sodium: a potent and selective inhibitor of COX-2 for parenteral administration. J Med Chem 43:1661–1663
Talley JJ, Brown DL, Carter JS, Graneto MJ, Koboldt CM, Masferrer JL, Perkins WE, Rogers RS, Shaffer AF, Zhang YY, Zweifel BS, Seibert K (2000b) 4-[5-Methyl-3-phenylisoxazol-4-yl]-benzenesulfonamide, valdecoxib: a potent and selective inhibitor of COX-2. J Med Chem 43:775–777
Vane JR (2000) The fight against rheumatism: from willow bark to COX-1 sparing drugs. J Physiol Pharmacol 51:573–586
Wolkenberg SE, Wisnoski DD, Leister WH, Wang Y, Zhao Z, Lindsley CW (2004) Efficient synthesis of imidazoles from aldehydes and 1,2-diketones using microwave irradiation. Org Lett 6:1453–1456
Zarghi A, Najafnia L, Daraie B, Dadrass OG, Hedayati M (2007) Synthesis of 2,3-diaryl-1,3-thiazolidine-4-one derivatives as selective cyclooxygenase (COX-2) inhibitors. Bioorg Med Chem Lett 17:5634–5637
Zarghi A, Zebardast T, Daraie B, Hedayati M (2009) Design and synthesis of new 1,3-benzthiazinan-4-one derivatives as selective cyclooxygenase (COX-2) inhibitors. Bioorg Med Chem 17:5369–5373
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zarghi, A., Arfaei, S. & Ghodsi, R. Design and synthesis of new 2,4,5-triarylimidazole derivatives as selective cyclooxygenase (COX-2) inhibitors. Med Chem Res 21, 1803–1810 (2012). https://doi.org/10.1007/s00044-011-9710-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-011-9710-5