Abstract
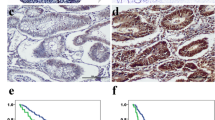
Tenascin-C (TN-C) is an extracellular matrix protein which participates in different processes like normal fetal development, wound healing, inflammation, keloids and rheumatoid arthritis. Furthermore, the immunostaining for TN-C is seen in the stroma of various malignant tumors as in glioblastoma multiforme (GBM), however, the significance of these findings is still not clear. In this study 62 GBM samples were analyzed immunohistochemically for distribution patterns of TN-C and correlated with angiogenesis and tumor cell proliferation. Tenascin-C in GBM localizes in two compartments, perivascular and intercellular space. Intercellular tenascin-C (TN-C ic) showed focal distribution in 66%, and diffuse one in 34% of cases. Perivascular tenascin-C (TN-C pv) showed strong correlation with microvascular density (MVD) and vascular endothelial growth factor (VEGF) expression. Moreover, it seems that TN-C pv enhanced the effect of VEGF. Intercellular TN-C did not correlate with MVD and VEGF expression, but showed strong correlation with proliferation index. Furthermore, tumors with diffuse TN-C ic expression had higher proliferation indices than tumors with focal TN-C expression. Our results indicate that TN-C plays a role in angiogenesis and tumor cell proliferation, but beside the intensity of expression, the distribution patterns are also important in these processes. This study also suggests that perivascular and intercellular TN-C compartments have probably different sources and different roles in GBM.
Similar content being viewed by others
References
Brooks PC, Dark RA, Cheresh DA: Requirement of vascular integrin alfa v beta 3 for angiogenesis. Science 264: 569–571, 1994
Castellani P, Siri A, Zardi L, et al: Distribution of tenascin in human gliomas is not related to ceil proliferation. J Neurol Neurosurg Psychiatry 62: 290–291, 1997
Castellan R, Caballero S, Hamdi KH, et at Effects of tenascin-C on normal and diabetic retinal endothelial cells in culture. Investigative Ophthalmol Vis Sci 43: 2758–2766, 2002
Chintala SK, Rao JS: Invasion in human glioma: role of extracellular matrix. Front Biosci 11: 324–339, 1996
Chiquet-Ehrismann R, Mackie EJ, Pearson CA, Sakakura T: An extracellular matrix protein involved in tissue interactions during fetal development and oncogeuesis. Cell 147: 131–139, 1986
Crossin KL: Cytotactin binding: inhibition of stimulated proliferation and intracellular alkalinization in fibroblasts. Proc Natl Acad Sci USA 88: 11403–11407, 1991
Ehrismann RC, Chiquet M: Tenascins: regulation and putative functions during pathological stress. J Pathol 200: 488–499, 2003
Fischer D, Brown-Ludi M, Schulthess T, Chiquet-Ehrismann R: Concerted action of tenascin-C domains in cell adhesion, antiadhesion and promotion of neurite outgrowth. J Cell Sci 110: 1513–1522, 1997
Giese A, Berens ME, Loo MA, et al: Contrasting migratory response of astrocytoma ceils to tenascin mediated by different integrins. J Cell Sci 109:2161–2168, 1996
Gladson CL: The extracellular matrix of gliomas: modulation of cell function. J Neuropathol Exp Neurol 58: 1029–1040, 1999
Herold-Mende Mueller, MM, Bonsanto MM, et al: Clinical impact and functional aspects of tenascin-c expression during glioma progression. Int J Cancer 98: 362–369, 2002
Higuchi M, Ohnishi T, Arita N, et al: Expression of tenascin in human gliomas: its relation to histological malignancy, tumor dedifferentiation and angiogenesis. Acta Neuropathol 85: 481–487, 1993
Huang W, Chiquet-Ehrismann R, Moyano V, et al: Interference of tenascin-C with syndecan-4 binding to fibronectin blocks cell adhesion and stimulates tumor cell proliferation. Cancer Res 61: 8586–8594, 2001
Ishihara A, Yoshida T, Tamaki H, Sakakukra T: Tenascin expression in cancer cells and stroma of human breast cancer and its prognostic significance. Clin Cancer Res 1: 1035–1041, 1995
Kim CH, Bak KH, Kim JM, et al: Expression of tenascin-C in astrocytic tumors: its relevance to proliferation and angiogenesis. Surg Neurol 54: 235–240, 2000
Kumar P, Wang J, Bernabeu C: CD 105 and angiogenesis. J Pathol 178:363–366, 1996
Kumar S, Ghellal A, Li C, et al: Vascular density determined using CD 105 antibody correlates with tumor prognosis. Cancer Res 59: 856–861, 1999
Lotz MM, Burdsal CA, Erickson HP, Mc Clay DR: Ceil adhesion to fibronectin and tenascin: quantitative measurements of initial binding and subsequent strengthening response. J Cell Biol 109: 1795–1805. 1989
Mai J, Sameni M, Mikkelsen T, Sloane BF: Degradation of extracellular matrix protein tenascin-C by cathepsin B: an interaction involved in the progression of gliomas. Biol Chem 383: 1407–1413, 2002
McLendon RE, Wikstrand J, Matthews Mr, et al: Glioma associated antigen expression in oligodendroglial neoplasms: tenascin and epidermal growth factor receptor. J Histochem Cytochem 48: 1103–1110, 2000
Orend G, Huang W, Olayioye MA, et al: Tenascin-C blocks cellcycle progression of anchorage-dependent fibroblasts on fibronectin through inhibition of syndecan-4. Oncogene 22: 3917–3926, 2003
Pilch H, Schaffer U, Schienger K, et al: Expression of tenascin in human cervical cancer: association of tenascin expression with clinicopathological parameters. Gynecol Oncol 73: 415–421, 1999
Roijani AM, Dorovini-Zis K: Glomeruloid vascular structures in glioblastoma multiforme: an immunohistochemical and ultrastructural study. J Neurosurg 85:1078–1084, 1996
Scheetz FJ, L’Loyd PJ: The tenascin family of ECM glycoproteins: structure, function, and regulation during embryonic development and tissue remodeling. Developmental Dynamics 218:235–259, 2000
Schenk S, Chuquet-Ehrismann R, Battegay EJ: The fibrinogen globe of tenascin-C promotes basic fibroblast growth factorinduced endothelial cell elongation. Mol Biol Cell 10: 2933–2943, 1999
Tanaka F, Yanagihara OK, Kawano Y, et al: Evaluation of angiogenesis in non-small cell lung cancer: comparison between anti-CD34 antibody and anti-CD 105 antibody. Clin Cancer Res 7: 3410–3415, 2001
Tanaka K, Hiraiwa N, Hashimoto H, et al: Tenascin -C regulates angiogenesis in tumor through the regulation of vascular endothelial growth factor expression. Int J Cancer 108: 31–40, 2004
Uhm JH, Gladson LC, Rao JS: The role of integrins in the malignant phenotype of gliomas. Front Biosci 4: 188–199, 1999
Ventimiglia JB, Wikstrand CJ, Ostrowski LE, et al: Tenascin expression in human glioma cell lines and normal tissues. J Neuroimmunol. 36:41–45, 1992
Vitolo D, Paradiso P, Uccini S, et al: Expression of adhesion molecules and extracellular matrix in glioblastomas: relation to angiogenesis and spread. Histopathology 28: 521–528, 1996
Wehrle-Haller B, Chiquet M: Dual function of tenascin: simultaneous promotion of neural growth and inhibition of glial migration. J Cell Sci 106: 597–610, 1993
Yoshyida T, Yoshimura E, Numata H, et al: Involvement of tenascin-C on proliferation and migration of laryngeal carcinoma cells. Virchows Arch 435: 496–500, 1993
Zagzag D, Friedlander DR, Miller DC, et al: Tenascin expression in astrocytomas correlates with angiogenesis. Cancer Res 55: 907–914, 1995
Zagzag D, Capo V: Angiogenesis in the central nervous system: a role for endothelial growth factor/vascular permeability factor and tenascin-C, common molecular effectors in cerebral neoplastic and non-neoplastic “angiogenic diseases”. Histol Histopathol 17: 301–321, 2002
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Behrem, S., Zarković, K., Eškinja, N. et al. Distribution pattern of Tenascin-C in glioblastoma: Correlation with angiogenesis and tumor cell proliferation. Pathol. Oncol. Res. 11, 229–235 (2005). https://doi.org/10.1007/BF02893856
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02893856