Abstract
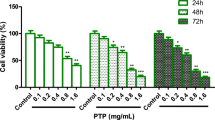
This study is the first to investigate the anticancer effect of Schisandra chinensis polysaccharide (SCP) in renal cell carcinoma (RCC) cells. The results revealed that SCP treatment showed high cytotoxic potency in Caki-1 cells by inducing apoptosis, which is associated with the disruption of mitochondrial membrane potential (MMP), release of cytochrome c into the cytosol, increase of Bax/Bcl-2 ratio, activation of caspase-3/9, and cleavage of poly(ADP-ribose) polymerase (PARP). Furthermore, pan-caspase inhibitor (z-VAD-fmk) significantly blocked SCP-induced apoptosis and PARP cleavage in Caki-1 cells. As well, we also observed that SCP inhibited the phosphorylation of ERK1/2, whereas it had no significant inhibition effect on the phospho-p38 and phospho-JNK activity. All the above parameters provided scientific evidence that SCP induced mitochondrial-mediated apoptosis in Caki-1 cells through the inactivation of ERK pathways, which may shed further light on its potential application as a cancer chemopreventive agent against RCC.





Similar content being viewed by others
References
Motzer RJ, Bander NH, Nanus DM. Renal-cell carcinoma. N Engl J Med. 1996;335:865–75.
Jemal A, Siegel R, Ward E, Murray T, Xu J, Smigal C, et al. Cancer statistics, 2006. CA Cancer J Clin. 2006;56:106–30.
Rini BI, Rathmell WK, Godley P. Renal cell carcinoma. Curr Opin Oncol. 2008;20:300–6.
Janzen NK, Kim HL, Figlin RA, Belldegrun AS. Surveillance after radical or partial nephrectomy for localized renal cell carcinoma and management of recurrent disease. Urol Clin North Am. 2003;30:843–52.
Cheng L, Zhang S, MacLennan GT, Lopez-Beltran A, Montironi R. Molecular and cytogenetic insights into the pathogenesis, classification, differential diagnosis, and prognosis of renal epithelial neoplasms. Hum Pathol. 2009;40:10–29.
Newman DJ, Cragg GM, Snader KM. Natural products as sources of new drugs over the period 1981–2002. J Nat Prod. 2003;66:1022–37.
Liu GT. Pharmacological actions and clinical use of Fructus schizandrae. Chin Med J. 1989;102:740–9.
Choi YW, Takamatsu S, Khan SI, Srinivas PV, Ferreira D, Zhao J, et al. Schisandrene, a dibenzocyclooctadiene lignan from Schisandra chinensis: structure-antioxidant activity relationships of dibenzocyclooctadiene lignans. J Nat Prod. 2006;69:356–9.
Panossian A, Wikman G. Pharmacology of Schisandra chinensis Bail.: an overview of Russian research and uses in medicine. J Ethnopharmacol. 2008;118:183–212.
Ikeya Y, Miki E, Okada M, Mitsuhashi H, Chai JG, Benzoylgomisin Q, et al. Two new lignans from Schisandra sphenanthera Rehd. et Wils. Chem Pharm Bull. 1990;38:1408–11.
Ikeya Y, Taguchi H, Yosioka I, Kobayashi H. The constituents of Schizandra chinensis Baill. I. Isolation and structure determination of five new lignans, Gomisin A, B, C, F, and G, and the absolute structure of schizandrin. Chem Pharm Bull. 1979;27:1383–94.
He XG, Lian LZ, Lin LZ. Analysis of lignan constituents from Schisandra chinensis by liquid chromatography–electrospray mass spectrometry. J Chromatogr A. 1997;757:81–7.
Yan S, Yang LQ, Zhao T, Zhao JL, Zou YM. Latest research progress on polysaccharide from Schisandra chinensis (Turcz.) Baill. J Jiangsu Univ. 2009;19:366–8.
Cummings BS, Kinsey GR, Bolchoz LJ, Schnellmann RG. Identification of caspase-independent apoptosis in epithelial and cancer cells. J Pharmacol Exp Ther. 2004;310:126–34.
Bai XC, Ma DZ, Liu AL, Shen XY, Wang QMJ, Liu YJ, et al. Rheb activates mTOR by antagonizing its endogenous inhibitor, FKBP38. Science. 2007;318:977–80.
Li M, Zhao L, Liu J, Liu AL, Zeng WS, Luo SQ, et al. Hydrogen peroxide induces G2 cell cycle arrest and inhibits cell proliferation in osteoblasts. Anat Rec (Hoboken). 2009;292:1107–13.
Yang J, Xiao YL, He XR, Qiu GF, Hu XM. Aesculetin-induced apoptosis through a ROS-mediated mitochondrial dysfunction pathway in human cervical cancer cells. J Asian Nat Prod Res. 2010;12:185–93.
Green DR, Reed JC. Mitochondria and apoptosis. Science. 1998;281:1309–12.
Cheung HY, Cheung SH, Li J, Cheung CS, Lai WP, Fong WF, et al. Andrographolide isolated from Andrographis paniculata induces cell cycle arrest and mitochondrial-mediated apoptosis in human leukemic HL-60 cells. Planta Med. 2005;71:1106–11.
Green DR. Kroemer GThe pathophysiology of mitochondrial cell death. Science. 2004;305:626–9.
Xie CY, Yang W, Li M, Ying J, Tao SJ, Li K, et al. Cell apoptosis induced by delta-elemene in colorectal adenocarcinoma cells via a mitochondrial-mediated pathway. Yakugaku Zasshi. 2009;129:1403–13.
Budihardjo I, Oliver H, Lutter M, Luo X, Wang X. Biochemical pathways of caspase activation during apoptosis. Annu Rev Cell Dev Biol. 1999;15:269–90.
Lowe SW, Lin AW. Apoptosis in cancer. Carcinogenesis. 2000;21:485–95.
Tang XL, Yang XY, Jung HJ, Kim SY, Jung SY, Choi DY, et al. Asiatic acid induces colon cancer cell growth inhibition and apoptosis through mitochondrial death cascade. Biol Pharm Bull. 2009;32:1399–405.
Petrache I, Choi ME, Otterbein LE, Chin BY, Mantell LL, Horowitz S, et al. Mitogen-activated protein kinase pathway mediates hyperoxia-induced apoptosis in cultured macrophage cells. Am J Physiol. 1999;277:L589–95.
Acknowledgements
This study was supported by the fund of Heilongjiang province science and technology plan project GC05C40701.
Conflicts of interest
None
Author information
Authors and Affiliations
Corresponding author
Additional information
Shi-Jian Liu and Hai-Ming Qu contributed equally to this paper.
Rights and permissions
About this article
Cite this article
Liu, SJ., Qu, HM. & Ren, YP. SCP, a polysaccharide from Schisandra chinensis, induces apoptosis in human renal cell carcinoma Caki-1 cells through mitochondrial-dependent pathway via inhibition of ERK activation. Tumor Biol. 35, 5369–5374 (2014). https://doi.org/10.1007/s13277-014-1699-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-014-1699-1