Abstract
Background
Treatment of HCV infection in patients with thalassemia major (TM) is limited by the lack of large clinical trials and concerns about ribavirin-induced hemolysis.
Methods
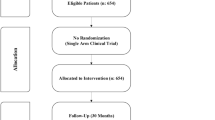
We conducted a prospective, randomized, open-label study to determine efficacy and tolerability of pegylated-interferon alfa 2b (1.5 μg/kg/week) alone (group A) or with ribavirin (12–15 mg/kg/day; group B) in patients with TM and chronic HCV infection. Patients with genotype 1 or 4 HCV were treated for 48 weeks and those with genotype 3 or 2 HCV for 24 weeks. Early viral response (EVR; after 12 weeks of treatment), end-of-treatment virological response (ETR) and sustained virological response (SVR; 6 months after stopping therapy) were assessed.
Results
Of 40 patients, 20 each were allocated to the two treatment groups. EVR rates in group A and B were 15 (75%) and 18 (90%), respectively. ETR occurred in 17/20 (85%) patients in each group. SVR occurred in 8 (40%) patients in group A and 14 (70%) in group B. Blood transfusion requirements increased in one patient in group A and four patients in group B. One patient in group A had severe sepsis and one in group B had nephrotic syndrome. Two patients in each group required reduction in drug dose.
Conclusions
In patients with TM and chronic HCV infection, pegylated interferon alfa 2b and ribavirin combination therapy achieves a higher SVR rate than pegylated interferon alone, and is well tolerated except for an increase in blood transfusion requirement.
Similar content being viewed by others
References
Angelucci E. Antibodies to hepatitis C virus in thalassemia. Haematologica 1994;79:353–355.
Wonke B, Hoffbrand AV, Brown D, Dusheiko G. Antibody to hepatitis C virus in multiply transfused patients with thalassaemia major. J Clin Pathol 1990;43:638–640.
Marwaha RK, Bansal D, Sharma S, et al. Seroprevalence of hepatitis C and B virus in multiply transfused beta-thalassemics: results from a thalassemic day care unit in north India. Vox Sang 2003;85:119–120.
Jaiswal SP, Chitnis DS, Jain AK, Inamdar S, Porwal A, Jain SC. Prevalence of hepatitis viruses among multitransfused homogenous thalassaemia patients. Hepatol Res 2001;19:247–253.
Agarwal MB, Malkan GH, Bhave AA, et al. Antibody to hepatitis-C virus in multi-transfused thalassaemics — Indian experience. J Assoc Physicians India 1993;41:195–197.
Angelucci E, Muretto P, Nicolucci A, et al. Effects of iron overload and hepatitis C virus positivity in determining progression of liver fibrosis in thalassemia following bone marrow transplantation. Blood 2002;100:17–21.
Di Marco V, Capra M, Gagliardotto F, et al. Liver disease in chelated transfusion-dependent thalassemics: the role of iron overload and chronic hepatitis C. Haematologica 2008;93:1243–1246.
Borgna-Pignatti C, Rugolotto S, De Stefano P, et al. Survival and complications in patients with thalassemia major treated with transfusion and deferoxamine. Haematologica 2004;89:1187–1193.
Borgna-Pignatti C, Cappellini MD, De Stefano P, et al. Cardiac morbidity and mortality in deferoxamine- or deferiprone-treated patients with thalassemia major. Blood 2006;107:3733–3737.
Zurlo MG, De Stefano P, Borgna-Pignatti C, et al. Survival and causes of death in thalassaemia major. Lancet 1989;2:27–30.
Dixit NM, Layden-Almer JE, Layden TJ, Perelson AS. Modelling how ribavirin improves interferon response rates in hepatitis C virus infection. Nature 2004;432:922–924.
Bodenheimer HC Jr, Lindsay KL, Davis GL, Lewis JH, Thung SN, Seeff LB. Tolerance and efficacy of oral ribavirin treatment of chronic hepatitis C: a multicenter trial. Hepatology 1997;26:473–477.
De Franceschi L, Fattovich G, Turrini F, et al. Hemolytic anemia induced by ribavirin therapy in patients with chronic hepatitis C virus infection: role of membrane oxidative damage. Hepatology 2000;31:997–1004.
Di Bisceglie AM, Bacon BR, Kleiner DE, Hoofnagle JH. Increase in hepatic iron stores following prolonged therapy with ribavirin in patients with chronic hepatitis C. J Hepatol 1994;21:1109–1112.
Li CK, Chan PK, Ling SC, Ha SY. Interferon and ribavirin as frontline treatment for chronic hepatitis C infection in thalassaemia major. Br J Haematol 2002;117:755–758.
Telfer PT, Garson JA, Whitby K, et al. Combination therapy with interferon alpha and ribavirin for chronic hepatitis C virus infection in thalassaemic patients. Br J Haematol 1997;98:850–855.
Harmatz P, Jonas MM, Kwiatkowski JL, et al. Safety and efficacy of pegylated interferon alfa-2a and ribavirin for the treatment of hepatitis C in patients with thalassaemia. Haematologica 2008;93:1247–1251.
Sood A, Midha V, Hissar S, et al. Comparison of low-dose pegylated interferon versus standard high-dose pegylated interferon in combination with ribavirin in patients with chronic hepatitis C with genotype 3: an Indian experience. J Gastroenterol Hepatol 2008;23:203–207.
Tover JL, Buti M, Segarra A, Majo J, Esteban R. De novo nephrotic syndrome following pegylated interferon alfa 2b/ribavirin therapy for chronic hepatitis C infection. Int Urol Nephrol 2008;40:539–541.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sood, A., Sobti, P., Midha, V. et al. Efficacy and safety of pegylated IFN alfa 2b alone or in combination with ribavirin in thalassemia major with chronic hepatitis C. Indian J Gastroenterol 29, 62–65 (2010). https://doi.org/10.1007/s12664-010-0014-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12664-010-0014-3