Abstract
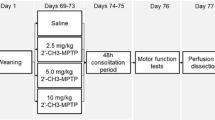
Parkinson’s disease (PD) is characterized by a progressive degeneration of dopamine (DA) neurons and gradual worsening of motor symptoms. The investigation of progressive degenerative mechanisms and potential neuroprotective strategies relies on experimental models of the chronic neuropathology observed in human. The present study investigated the progressive nature of neurodegeneration in the 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine/probenecid (MPTPp) chronic mouse model of PD. MPTP (25 mg/kg) plus probenecid (250 mg/kg) were administered twice a week for 5 weeks. We evaluated behavioral deficits (olfactory and motor impairment), neurodegeneration (loss of tyrosine hydroxylase (TH)-positive cells in the substantia nigra pars compacta, SNc), biochemical markers of functional impairment in the caudate-putamen (CPu) (striatal enkephalin mRNA, DA and DOPAC levels), and glial reactivity (CD11b and GFAP immunoreactivity in the SNc and CPu) at progressive time-points (after 1, 3, 7, and 10 administrations of MPTPp). Olfactory dysfunction already appeared after the 1st MPTPp injection, whereas motor dysfunction appeared after the 3rd and worsened upon subsequent administrations. Moreover, starting after three MPTPp injections, we observed a gradual decline of TH-positive cells in the SNc, and a gradual raise of enkephalin mRNA in the CPu. Striatal DA levels reduction was not different among all time-points evaluated, whereas DOPAC levels were similarly reduced after 1–7 MPTP injections, but were further decreased after the 10th injection. Reactive microglia and astroglia were observed in both the SNc and CPu from the 1st MPTPp administration. In the SNc, gliosis displayed a gradual increase over the treatment. After 2 months, TH, DA, DOPAC, and reactive glia in the SNc were still altered in MPTPp-treated mice as compared to controls. By showing a progressive development of behavioral deficits and nigral neurodegeneration, together with impairment of biochemical parameters and gradual increase of glial response, results suggest that the chronic MPTPp protocol is a model of progressive PD, which may be suitable to investigate chronic pathological processes and neuroprotective strategies in PD.







Similar content being viewed by others
References
Alvarez-Fischer D, Guerreiro S, Hunot S, Saurini F, Marien M, Sokoloff P, Hirsch EC, Hartmann A, Michel PP (2008) Modelling Parkinson-like neurodegeneration via osmotic minipump delivery of MPTP and probenecid. J Neurochem 107:701–711
Anglade P, Mouatt-Prigent A, Agid Y, Hirsch E (1996) Synaptic plasticity in the caudate nucleus of patients with Parkinson’s disease. Neurodegeneration 5:121–128
Araki T, Mikami T, Tanji H, Matsubara M, Imai Y, Mizugaki M, Itoyama Y (2001) Biochemical and immunohistological changes in the brain of 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP)-treated mouse. Eur J Pharm Sci 12:231–238
Barcia C, Fernández Barreiro A, Poza M, Herrero MT (2003) Parkinson’s disease and inflammatory changes. Neurotox Res 5:411–418
Barcia C, Sánchez Bahillo A, Fernández-Villalba E, Bautista V, Poza Y, Poza M, Fernández-Barreiro A, Hirsch EC, Herrero MT (2004) Evidence of active microglia in substantia nigra pars compacta of parkinsonian monkeys 1 year after MPTP exposure. Glia 46:402–419
Berendse HW, Booij J, Francot CM, Bergmans PL, Hijman R, Stoof JC, Wolters EC (2001) Subclinical dopaminergic dysfunction in asymptomatic Parkinson’s disease patients’ relatives with a decreased sense of smell. Ann Neurol 50:34–41
Bezard E, Jaber M, Gonon F, Boireau A, Bloch B, Gross CE (2000a) Adaptive changes in the nigrostriatal pathway in response to increased 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine-induced neurodegeneration in the mouse. Eur J Neurosci 12:2892–2900
Bezard E, Dovero S, Imbert C, Boraud T, Gross CE (2000b) Spontaneous long-term compensatory dopaminergic sprouting in MPTP-treated mice. Synapse 38:363–368
Blanchard V, Chritin M, Vyas S, Savasta M, Feuerstein C, Agid Y, Javoy-Agid F, Raisman-Vozari R (1995) Long-term induction of tyrosine hydroxylase expression: compensatory response to partial degeneration of the dopaminergic nigrostriatal system in the rat brain. J Neurochem 64:1669–1679
Blandini F, Armentero MT, Martignoni E (2008) The 6-hydroxydopamine model: news from the past. Parkinsonism Relat Disord 2:S124–S129
Braak H, Del Tredici K, Rüb U, de Vos RA, Jansen Steur EN, Braak E (2003) Staging of brain pathology related to sporadic Parkinson’s disease. Neurobiol Aging 24:197–211
Carta AR, Tronci E, Pinna A, Morelli M (2005) Different responsiveness of striatonigral and striatopallidal neurons to l-DOPA after a subchronic intermittent l-DOPA treatment. Eur J Neurosci 21:1196–1204
Carter RJ, Lione LA, Humby T, Mangiarini L, Mahal A, Bates GP, Dunnett SB, Morton AJ (1999) Characterization of progressive motor deficits in mice transgenic for the human Huntington’s disease mutation. J Neurosci 19:3248–3257
Chase TN, Oh JD (2000) Striatal dopamine- and glutamate-mediated dysregulation in experimental parkinsonism. Trends Neurosci 23:S86–S91
Chesselet MF, Fleming S, Mortazavi F, Meurers B (2008) Strengths and limitations of genetic mouse models of Parkinson’s disease. Parkinsonism Relat Disord 2:S84–S87
Colburn RW, DeLeo JA, Rickman AJ, Yeager MP, Kwon P, Hickey WF (1997) Dissociation of microglial activation and neuropathic pain behaviors following peripheral nerve injury in the rat. J Neuroimmunol 79:163–175
Dentresangle C, Le Cavorsin M, Savasta M, Leviel V (2001) Increased extracellular DA and normal evoked DA release in the rat striatum after a partial lesion of the substantia nigra. Brain Res 893:178–185
Dervan AG, Meshul CK, Beales M, McBean GJ, Moore C, Totterdell S, Snyder AK, Meredith GE (2004) Astroglial plasticity and glutamate function in a chronic mouse model of Parkinson’s disease. Exp Neurol 190:145–156
Dluzen DE, McDermott JL, Anderson LI, Kucera J, Joyce JN, Osredkar T, Walro JM (2004) Age-related changes in nigrostriatal dopaminergic function are accentuated in ± brain-derived neurotrophic factor mice. Neuroscience 128:201–208
Drucker-Colín R, García-Hernández F (1991) A new motor test sensitive to aging and dopaminergic function. J Neurosci Methods 39:153–161
Elsworth JD, Taylor JR, Sladek JR Jr, Collier TJ, Redmond DE Jr, Roth RH (2000) Striatal dopaminergic correlates of stable parkinsonism and degree of recovery in old-world primates 1 year after MPTP treatment. Neuroscience 95:399–408
Fernagut PO, Chalon S, Diguet E, Guilloteau D, Tison F, Jaber M (2003) Motor behaviour deficits and their histopathological and functional correlates in the nigrostriatal system of dopamine transporter knockout mice. Neuroscience 116:1123–1130
Fleming SM, Salcedo J, Fernagut PO, Rockenstein E, Masliah E, Levine MS, Chesselet MF (2004) Early and progressive sensorimotor anomalies in mice overexpressing wild-type human alphasynuclein. J Neurosci 24:9434–9440
Fornai F, Schlüter OM, Lenzi P, Gesi M, Ruffoli R, Ferrucci M, Lazzeri G, Busceti CL, Pontarelli F, Battaglia G, Pellegrini A, Nicoletti F, Ruggieri S, Paparelli A, Südhof TC (2005) Parkinson-like syndrome induced by continuous MPTP infusion: convergent roles of the ubiquitin-proteasome system and alpha-synuclein. Proc Natl Acad Sci USA 102:3413–3418
Forno LS, DeLanney LE, Irwin I, Di Monte D, Langston JW (1992) Astrocytes and Parkinson’s disease. Prog Brain Res 94:429–436
Gerfen CR (2003) D1 dopamine receptor supersensitivity in the dopamine-depleted striatum animal model of Parkinson’s disease. Neuroscientist 9:455–462
Gerfen CR, Engber TM, Mahan LC, Susel Z, Chase TN, Monsma FJ Jr, Sibley DR (1990) D1 and D2 dopamine receptor-regulated gene expression of striatonigral and striatopallidal neurons. Science 250:1429–1432
Haehner A, Hummel T, Hummel C, Sommer U, Junghanns S, Reichmann H (2007) Olfactory loss may be a first sign of idiopathic Parkinson’s disease. Mov Disord 22:839–842
Hald A, Lotharius J (2005) Oxidative stress and inflammation in Parkinson’s disease: is there a causal link? Exp Neurol 193:279–290 (Review)
Hirsch EC, Breidert T, Rousselet E, Hunot S, Hartmann A, Michel PP (2003) The role of glial reaction and inflammation in Parkinson’s disease. Ann N Y Acad Sci 991:214–228
Jackson-Lewis V, Jakowec M, Burke RE, Przedborski S (1995) Time course and morphology of dopaminergic neuronal death caused by the neurotoxin 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine. Neurodegeneration 4:257–269
Jenner P (2008) Functional models of Parkinson’s disease: a valuable tool in the development of novel therapies. Ann Neurol 2:S16–S29
Kohutnicka M, Lewandowska E, Kurkowska-Jastrzebska I, Członkowski A, Członkowska A (1998) Microglial and astrocytic involvement in a murine model of Parkinson’s disease induced by 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP). Immunopharmacology 39:167–180
Kurkowska-Jastrzebska I, Wrońska A, Kohutnicka M, Członkowski A, Członkowska A (1999) The inflammatory reaction following 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine intoxication in mouse. Exp Neurol 156:50–61
Liberatore GT, Jackson-Lewis V, Vukosavic S, Mandir AS, Vila M, McAuliffe WG, Dawson VL, Dawson TM, Przedborski S (1999) Inducible nitric oxide synthase stimulates dopaminergic neurodegeneration in the MPTP model of Parkinson disease. Nat Med 5:1403–1409
Linder JC, Klemfuss H, Groves PM (1987) Acute ultrastructural and behavioral effects of 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) in mice. Neurosci Lett 82:221–226
Manning-Bog AB, Langston JW (2007) Model fusion, the next phase in developing animal models for Parkinson’s disease. Neurotox Res 11:219–240 Review
Matsuura K, Kabuto H, Makino H, Ogawa N (1997) Pole test is a useful method for evaluating the mouse movement disorder caused by striatal dopamine depletion. J Neurosci Methods 73:45–48
McGeer PL, McGeer EG (2008) Glial reactions in Parkinson’s disease. Mov Disord 23:474–483
McGeer PL, Itagaki S, Boyes BE, McGeer EG (1988) Reactive microglia are positive for HLA-DR in the substantia nigra of Parkinson’s and Alzheimer’s disease brains. Neurology 38:1285–1291
Meredith GE, Kang UJ (2006) Behavioral models of Parkinson’s disease in rodents: a new look at an old problem. Mov Disord 21:1595–1606 Review
Meredith GE, Totterdell S, Petroske E, Santa Cruz K, Callison RC Jr, Lau YS (2002) Lysosomal malfunction accompanies alpha-synuclein aggregation in a progressive mouse model of Parkinson’s disease. Brain Res 22:156–165
Meredith GE, Dervan AG, Totterdell S (2005) Activated microglia persist in the substantia nigra of a chronic MPTP mouse model of Parkinson’s disease. In: Bolam JP, Ingham, Magill PJ (eds) The basal ganglia VIII. Springer Science, Singapore, pp 341–347
Meredith GE, Totterdell S, Potashkin JA, Surmeier DJ (2008) Modeling PD pathogenesis in mice: advantages of a chronic MPTP protocol. Parkinsonism Relat Disord 2:S112–S115
Mitra N, Mohanakumar KP, Ganguly DK (1992) Dissociation of serotoninergic and dopaminergic components in acute effects of 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine in mice. Brain Res Bull 28:355–364
Mitsumoto Y, Watanabe A, Mori A, Koga N (1998) Spontaneous regeneration of nigrostriatal dopaminergic neurons in MPTP-treated C57BL/6 mice. Biochem Biophys Res Commun 248:660–663
Novikova L, Garris BL, Garris DR, Lau YS (2006) Early signs of neuronal apoptosis in the substantia nigra pars compacta of the progressive neurodegenerative mouse 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine/probenecid model of Parkinson’s disease. Neuroscience 140:67–76
Ogawa N, Hirose Y, Ohara S, Ono T, Watanabe Y (1985) A simple quantitative bradykinesia test in MPTP-treated mice. Res Commun Chem Pathol Pharmacol 50:435–441
Ogawa N, Mizukawa K, Hirose Y, Kajita S, Ohara S, Watanabe Y (1987) MPTP-induced parkinsonian model in mice: biochemistry, pharmacology and behavior. Eur Neurol 1:16–23
Olanow CW (2007) The pathogenesis of cell death in Parkinson’s disease. Mov Disord 17:S335–S342
Olanow CW, Kieburtz K, Schapira AH (2008) Why have we failed to achieve neuroprotection in Parkinson’s disease? Ann Neurol 2:S101–S110
Paxinos G, Franklin KBJ (eds) (2001) The mouse brain in stereotaxic coordinates, 2nd edn. Academic Press, San Diego
Perez XA, Parameswaran N, Huang LZ, O’Leary KT, Quik M (2008) Pre-synaptic dopaminergic compensation after moderate nigrostriatal damage in non-human primates. J Neurochem 105:1861–1872
Petroske E, Meredith GE, Callen S, Totterdell S, Lau YS (2001) Mouse model of Parkinsonism: a comparison between subacute MPTP and chronic MPTP/probenecid treatment. Neuroscience 106:589–601
Quinn LP, Perren MJ, Brackenborough KT, Woodhams PL, Vidgeon-Hart M, Chapman H, Pangalos MN, Upton N, Virley DJ (2007) A beam-walking apparatus to assess behavioural impairments in MPTP-treated mice: pharmacological validation with R-(−)-deprenyl. J Neurosci Methods 164:43–49
Robinson TE, Mocsary Z, Camp DM, Whishaw IQ (1994) Time course of recovery of extracellular dopamine following partial damage to the nigrostriatal dopamine system. J Neurosci 14:2687–2696
Rousselet E, Joubert C, Callebert J, Parain K, Tremblay L, Orieux G, Launay JM, Cohen-Salmon C, Hirsch EC (2003) Behavioral changes are not directly related to striatal monoamine levels, number of nigral neurons, or dose of parkinsonian toxin MPTP in mice. Neurobiol Dis 14:218–228
Schintu N, Frau L, Ibba M, Caboni P, Garau A, Carboni E, Carta AR (2009) PPAR-gamma mediated neuroprotection in a chronic mouse model of Parkinson’s disease. Eur J Neurosci 29:954–963
Schneider B, Zufferey R, Aebischer P (2008) Viral vectors, animal models and new therapies for Parkinson’s disease. Parkinsonism Relat Disord 2:S169–S171
Sedelis M, Schwarting RK, Huston JP (2001) Behavioral phenotyping of the MPTP mouse model of Parkinson’s disease. Behav Brain Res 125:109–125
Serradj N, Jamon M (2007) Age-related changes in the motricity of the inbred mice strains 129/sv, C57BL/6j. Behav Brain Res 177:80–89
Smeyne RJ, Jackson-Lewis V (2005) The MPTP model of Parkinson’s disease. Brain Res Mol Brain Res 134:57–66
Sofroniew MV (2005) Reactive astrocytes in neural repair and protection. Neuroscientist 11:400–407
Sonsalla PK, Zeevalk GD, German DC (2008) Chronic intraventricular administration of 1-methyl-4-phenylpyridinium as a progressive model of Parkinson’s disease. Parkinsonism Relat Disord 2:S116–S118
Tansey MG, McCoy MK, Frank-Cannon TC (2007) Neuroinflammatory mechanisms in Parkinson’s disease: potential environmental triggers, pathways, and targets for early therapeutic intervention. Exp Neurol 208:1–25
Wu DC, Jackson-Lewis V, Vila M, Tieu K, Teismann P, Vadseth C, Choi DK, Ischiropoulos H, Przedborski S (2002) Blockade of microglial activation is neuroprotective in the 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine mouse model of Parkinson disease. J Neurosci 22:1763–1771
Yazdani U, German DC, Liang CL, Manzino L, Sonsalla PK, Zeevalk GD (2006) Rat model of Parkinson’s disease: chronic central delivery of 1-methyl-4-phenylpyridinium (MPP+). Exp Neurol 200:172–183
Zhou C, Huang Y, Przedborski S (2008) Oxidative stress in Parkinson’s disease: a mechanism of pathogenic and therapeutic significance. Ann N Y Acad Sci 147:93–104
Author information
Authors and Affiliations
Corresponding author
Additional information
Nicoletta Schintu and Lucia Frau equally contributed to the work.
Rights and permissions
About this article
Cite this article
Schintu, N., Frau, L., Ibba, M. et al. Progressive Dopaminergic Degeneration in the Chronic MPTPp Mouse Model of Parkinson’s Disease. Neurotox Res 16, 127–139 (2009). https://doi.org/10.1007/s12640-009-9061-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12640-009-9061-x