Abstract
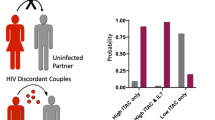
We recruited 59 individuals of known HIV serostatus after informed consent however, 44 were serodiscordant heterosexual partners [serodiscordant seronegative (SSN group) and serodiscordant seropositive (SSP group)] while 15 were seronegative healthy individuals (SNH). In the case–control study we choose to determine Malondialdehyde (MDA) concentration as a marker of lipid peroxidation index (oxidative stress) spectrophotometrically and quantify HIV mRNA by Real Time-nucleic acid sequence based amplification assay (RT-NASBA). Here our result show for the first time a high concentration of lipid peroxidation product (MDA, 116.6%) with a significant (P < 0.05) increase in HIV serodiscordant seropositive subjects over their seronegative partners. However, Spearman rank correlation statistics of SSP group showed a positive correlation value (P < 0.01, r = 0.89) between MDA and mRNA and a negative correlation between MDA and T-cell ratio (P < 0.01, r = 0.96).The study may strongly indicate a possible lipid peroxidation product threshold for predicting HIV infection and progression in serodiscordant heterosexual partners.


Similar content being viewed by others
References
Dutton G. Infected non progressors are providing clues to the control, and potentially the eradication of HIV. Science. 2006;20(11):27–31.
Smith RA. The body: couples 1998. http://www.thebody.com/encyclo/couples.
Fowke KR, Nagelkerke NJ, Kimani J, Simonsen JN, et al. Resistance to HIV-1 infection among persistently seronegative prostitute in Nairobi, Kenya. Lancet. 1997;349(9052):649–50.
AIDS SA.Natural resistance to HIV/AIDS. AIDS SA. 1998;6(3):1–5.
Pantaleo G, Graziosi C, Fauci AS. The immunopathogenesis of human immunodeficiency virus infection. N Engl J Med. 1993;328:327–35.
Levy JA. Pathogenesis of human immunodeficiency virus infection. Microbiol Rev. 1993;57:183–289.
Laeyendecker O, li X, Arroyo, M. The effect of HIV subtype on rapid disease progression in Rakai, uganda. 13th Conference on retroviruses and opportunistic infections. (No.44LB); 2006.
Quinn TC, Wawer MJ, Sewankambo N, Serwadda D, Wabwire-Mangen F, Li C, et al. Viral load and heterosexual transmission of human immunodeficiency virus type 1. Rakai Project Study Group. N Engl J Med. 2000;342:921–9.
Stapraws SI, Dailey PJ, Rosenthal A, Horton C, Grant RM, Lerche N, Feinberg MB. Simian immunodeficiency virus course in predicting the extent of virus replication during primary infection. J Virol. 1999;73(6):4829–39.
Gray RH, Wawer MJ, Sewankampo NK, Brookmeyer R, Serwadda D, Wabwire-Mangen F, et al. Probability of HIV-1 transmission per coital act in monogamous, heterosexual, HIV-1-discordant couples in Rakai, Uganda. Lancet. 2001;357(9263):1149–53.
Saksela K, Stevens C, Rubinstein P, Baltimore D. Human immunodeficiency virus type 1 mRNA expression in peripheral blood cells predicts disease progression independently of the numbers of CD4+ lymphocytes. Proc Natl Acad Sci USA. 1994;91:1104–8.
Michael NL, Mo T, Merzouki A, O’Shaughnessy M, Oster C, Burke DS, et al. Human immunodeficiency virus type 1 cellular RNA load and splicing patterns predict disease progression in a longitudinally studied cohort. J Virol. 1995;69:1868–77.
Furtado MR, Kingsley LA, Wolinsky SM. Changes in the viral mRNA expression pattern correlate with a rapid rate of CD4+ T-cell number decline in human immunodeficiency virus type 1-infected individuals. J Virol. 1995;69:2092–100.
Saksela K, Stevens CE, Rubinstein P, Taylor PE, Baltimore D. HIV-1 messenger RNA in peripheral blood mononuclear cells as an early marker for risk for progression to AIDS. Ann Intern Med. 1995;123:641–8.
Moseson M, Zeleniuch-Jacquotte A, Belsito DV, Shore RE, Marmor M, Pasternack B. The potential role of nutritional factors in the induction of immunologic abnormalities in HIV-positive homosexual men. J Acquir Immune Defic Syndr. 1989;2:235–47.
Burack JH, Barrett DC, Stall RD, Chesney MA, Ekstrand ML, Coates TJ. Depressive symptoms and CD4 lymphocyte decline among HIV-infected men. JAMA. 1993;270:2568–73.
Vittinghoff E, Hessol NA, Bacchetti P, Fusaro RE, Holmberg SD, Buchbinder SP. Cofactors for HIV disease progression in a cohort of homosexual and bisexual men. J Acquir Immune Defic Syndr. 2001;27:308–14.
Uchida A. Activation of stress signalling pathways by end product of lipid peroxidation. J Biol chem. 2000;274:2234–42.
Aust SD, Svingen BA. Lipid peroxidation in cellular membrane. In: Pryor Wa, editor. Free radicals in biology. New York: Academic press; 1982. p. 91–113.
Chan DC, Kim PS. HIV entry and its inhibition. Cell. 1998;93(5):681–4.
Lee PS, Xia J, Lewis M, Han M. Zn2+ promotes the self-association of human immunodeficiency virus type-1 integrase in vitro. Biochemistry. 1997;36:173–80.
Boom R, Sol CJ, van der Noordaa J. Rapid and simple method for purification of nucleic acids. Clin Microbiol. 1990;28:495–503.
Wallin B, Roseengren B, Shetzer HG, Camego G. Lipoprotein oxidation and measurement of thiobarbituric acid reacting substance (TBARS) formation in a single micro plate: its use for evaluation of antioxidants. Ann Biochem. 1993;208:10–5.
Harrigan PR, Bloor S, Larder BA. Relative replicative fitness of zidovudine-resistant human immunodeficiency virus type 1 isolates in vitro. J Virol. 1998;72:3773–8.
Learmont JC. Immunologic and virologic status after 14–18 years of infection with an attenuated strain of HIV- 1. N Engl J Med. 1999;340:1715–22.
Theodorou I, Meyer L, Magierowska M, Katlama C, Rouzious C. HIV-1 infection in an individual homozygous for CCR5 delta 32. Seroco study group. Lancet. 1997;349(9060):1219–20.
Liu R, Paxlon WA, Choe S, Ceradini D, Martin SR, Horuk R, MacDonald ME, Stuhlmam H, Koup RA, Landau NR. Homozygous defect in HIV-1 coreceptor accounts for resistance of some multiply-exposed individuals to HIV-1 infection. Cell. 1996;86:367–77.
Deek SG, Casper C, Carlenor EN. Neutralizing antibody responses against autologous and heterogolous viruses in acute versus chronic human immunodeficiency virus (HIV) infection: evidence for a constraint on the ability of HIV to completely evade neutralizing antibody responses. J Virol. 2006;80:6155–64.
Abram ME, Ferris AL, Shao W, Alvord WG, Hughes SH. Nature, position, and frequency of mutations made in a single cycle of HIV-1 replication. J Virol. 2010;84:9864–78.
Malim HM. Natural resistance to HIV infection: The Vif–APOBEC interaction. C R Biologies. 2006;329(11):871–5.
Cohen J. HIV natural resistance field finally overcomes resistance. Science. 2009;326(5959):1476–7.
John WP, Lloyd AH, Akhlaq AF. Cyclooxygenases, lipoxygenases, and epoxygenases in CNS: their role and involvement in neurological disorders. Brain Res Rev. 2006;52(2):201–43.
Domenico P, Virginia MY, John Q, et al. Increased F2-isoprostanes in Alzheimer’s disease: evidence for enhanced lipid peroxidation in vivo. FASEB J. 1998;12:1777–83.
He X, Jittiwat J, Kim JH, Jenner AM, Farooqui AA, Patel SC, Ong WY. Apolipoprotein D modulates F2-isoprostane and 7-ketocholesterol formation and has a neuroprotective effect on organotypic hippocampal cultures after kainate-induced excitotoxic injury. Neur Lett. 2009;455(3):183–6.
Attord C, Schoffer H. Antioxidant and lipid peroxidation products in HIV-1 infected patients with associated skin disease. Eur J Dermatol. 2006;4:148–53.
Modjarrad K, Chamot E, Vermund S. Impact of small reductions in plasma HIV RNA levels on the risk of heterosexual transmission and disease progression. AIDS. 2008;22(16):2179–85.
Schreck R, Rieber P, Baeuerle PA. Reactive oxygen intermediates as apparently widely used messengers in the activation of the NF–kB transcription factor and HIV–1. EMBO J. 1991;10:2247–2258.3.
Das UN, Podma M, Sogar PS, Ramesh G, Koratkar R. Stimulation of free radical generation in human leucocytes by various agents including tumor necrosis factor is a calmodulin-dependent process. Biochem Biophys Res Commun. 1990;167:1030–6.
Westendrop MO, Shatrov VA, SchulzeOsthof K, Frank R, Kraft M, Los M, Krammer PH, Droge W, et al. HIV-1 Tat potentiates TNF-induced NF-kappa B activation and cytotoxicity by altering the cellular redox state. EMBO J. 1995;14(3):546–54.
Droge W, Eck HP, Mihm S. Oxidant-antioxidant status in human immuno deficiency virus infection. In: Packer L, editor. Oxygen radicalism biological systems. San Diego: Academic Press; 1994. p. 594–601.
Wanchu A, Rana SV, Pallikkuth S, Sachdeva RK. Oxidative stress in HIV-infected individuals: a cross-sectional study. AIDS Res Hum Retroviruses. 2009;25:1307–11.
Ogunro PS, Ogungbamigbe TO, Ajala MO, Egbewale BE. Total antioxidant status and lipid peroxidation in HIV-1 infected patients in a rural area of south western Nigeria. Afr J Med Med Sci. 2005;34:221–5.
Suresh DR, Annam V, Pratibha K, Prasad BV. Total antioxidant capacity–a novel early bio-chemical marker of oxidative stress in HIV infected individuals. J Biomed Sci. 2009;16:61.
Burchill SA, Pereboltel L, Johnston C, Top B, Selby P. Comparison of the RNA-amplification based methods RT-PCR and NASBA for the detection of circulating tumour cells. Br J Cancer. 2000;86(1):102–9. (22 ref.).
Dewar RL, Highbarger HC, Sarmiento MD, Todd JA, Vasudevachari MB, Davey RT Jr, et al. Application of branched DNA signal amplification to monitor human immunodeficiency virus type 1 burden in human plasma. J Infect Dis. 1994;170:1172–9.
Schneider, et al. Real-time nucleic acid sequence-based amplification is more convenient than real-time PCR for quantification of Plasmodium falciparum. J Clin Microbiol. 2005;43(1):402–5.
Keightley, et al. Real-time NASBA detection of SARS-associated coronavirus and comparison with real-time reverse transcription-PCR. J Med Virol. 2005;77(4):602–8.
Kievits, et al. NASBA isothermal enzymatic in vitro nucleic acid amplification optimized for the diagnosis of HIV-1 infection. J Virol Methods. 1991;35(3):273–86.
Collins, et al. Detection of highly pathogenic and low pathogenic avian influenza subtype H5 (Eurasian lineage) using NASBA. J Virol Methods. 2002;103(2):213–25.
Collins, et al. A method to detect major serotypes of foot-and-mouth disease virus. Biochem Biophys Res Commun. 2002;297(2):267–74.
Cardier JE, Egidio R, Soyano Andres. Lipid Peroxidation and changes in T lymphocyte subsets and lymphocyte proliferative response in experimental iron overload. J Immunopharmacol. 1995;17(4):705–17.
Conflict of Interest
There is no potential conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ibeh, B.O., Obidoa, O. & Nwuke, C. Lipid Peroxidation Correlates with HIVmRNA in Serodiscordant Heterosexual HIVpartners of Nigerian Origin. Ind J Clin Biochem 26, 249–256 (2011). https://doi.org/10.1007/s12291-011-0120-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12291-011-0120-8