Opinion statement
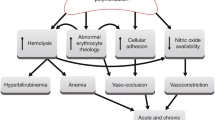
Stroke is one of the major complications in children with sickle cell disease (SCD). Ischemic stroke is associated with small asymptomatic subcortical infarcts to large territorial lesions causing major disability. Intracranial hemorrhages may be caused by aneurysm rupture or by leakage from moyamoya vessels or venous sources. There have been no acute stroke treatment studies in SCD, but hydration and exchange transfusion are often recommended. However, there is an evidence base for primary and to some extent secondary stroke prevention. Primary prevention of stroke was demonstrated in the Stroke Prevention Trial in Sickle Cell Anemia (STOP), in which children with transcranial Doppler (TCD) mean blood flow velocities of 200 cm/second (previously shown to indicate high stroke risk) or higher were randomized to either regular blood transfusions or no transfusion. The study showed a very significant 90% reduction in first stroke with transfusion. In STOP2, discontinuing transfusions after 30 months or more (even with normal TCD) resulted in a high rate of reversion to abnormal TCD values and stroke. TCD screening of all children with SCD, and initiation and maintenance of chronic transfusion to maintain hemoglobin S below 30% in the high-risk group, is the only proven prevention strategy for stroke in SCD. Hydroxyurea is being studied as secondary stroke prevention at this time. No recommendation specific to SCD regarding the use of antiplatelet agents or anticoagulants in ischemic stroke can be made. Bone marrow transplantation can be curative for SCD, and limited data support its use to prevent stroke in SCD.
Similar content being viewed by others
References and Recommended Reading
Ohene-Frempong K, Weiner SJ, Sleeper LA, et al.: Cerebrovascular accidents in sickle cell disease: rates and risk factors. Blood 1998, 91:288–294.
Prengler M, Pavlakis SG, Prohovnik I, Adams RJ: Sickle cell disease: the neurologic complications. Ann Neurol 2002, 51:543–552.
Moser FG, Miller ST, Bello JA, et al.: The spectrum of brain MR abnormalities in sickle cell disease: a report from the co-operative study of sickle cell disease. Am J Neuroradiol 1996, 17:965–972.
Powars DR, Conti PS, Wong WY, et al.: Cerebral vasculopathy in sickle cell anemia: diagnostic contribution of positron emission tomography. Blood 1999, 93:71–79.
Adams RJ, Nichols FT, McKie V, et al.: Cerebral infarction in sickle cell anemia: mechanism based on CT and MRI. Neurology 1988, 38:1012–1017.
Preul MC, Cendes F, Just N, Mohr G: Intracranial aneurysms and sickle cell anemia: multiplicity and propensity for the vertebrobasilar territory. Neurosurgery 1998, 42:971–977.
Pegelow CH: Stroke in children with sickle cell anemia: aetiology and treatment. Paediatr Drugs 2001, 3:421–432.
Ng WH, Yeo TT, Seow WT: Non-traumatic spontaneous acute epidural hematoma--report of two cases and review of the literature. J Clin Neurosci 2004, 7:791–794.
Hoppe C, Klitz W, Cheng S, et al.: Gene interactions and stroke risk in children with sickle cell anemia. Blood 2004, 103:2391–2396.
Hoppe C, Klitz W, Noble J, et al.: Distinct HLA associations by stroke subtype in children with sickle cell anemia. Blood 2003, 101:2865–2869.
Platt OS: Preventing stroke in sickle cell anemia. N Engl J Med 2005, 353:2743–2745.
Ware RE, Zimmerman SA, Sylvestre PB, et al.: Prevention of secondary stroke and resolution of transfusional iron overload in children with sickle cell anemia using hydroxyurea and phlebotomy. J Pediatr 2004, 145:346–352. This study from Duke University assessed the efficacy of hydroxyurea and phlebotomy secondary stroke prevention and iron overload as an alternative to chronic transfusion therapy. Thirty-five children with stroke and SCD were put on hydroxyurea alone or concurrently with transfusion therapy until full-dose hydroxyurea was reached. Stroke recurrence rate was 5.7 events per 100 patient-years, but children receiving overlapping hydroxyurea therapy had only 3.6 events per 100 patient-years.
Kirkham FJ, deBaun MR: Stroke in children with sickle cell disease. Curr Treat Options Neurol 2004, 6:357–375.
Nowak-Gottl U, Straeter R, Sebire G, Kirkham F: Antithrombotic drug treatment of pediatric patients with ischemic stroke. Paediatr Drugs 2003, 5:167–175.
Soman T, Rafay MF, Hune S, et al.: The risks and safety of clopidogrel in pediatric arterial ischemic stroke. Stroke 2006, 37:1120–1122.
Thirumalai SS, Shubin RA: Successful treatment of stroke in a child using recombinant tissue plasminogen activator. J Child Neurol 2000, 15:558.
Anson JA, Koshy M, Ferguson L, et al.: Subarachnoid hemorrhage in sickle cell disease. J Neurosurg 1991, 75:552–558.
Vicari P, Choairy CC, Siufi GC, et al.: Embolization of intracranial aneurysms and sickle cell disease. Am J Hematol 2004, 76:83–84.
Molyneux A, Kerr I, Stratton I, et al.: International Subarachnoid Aneurysm Trial (ISAT) of neurosurgical clipping versus endovascular coiling in 2143 patients with ruptured intracranial aneurysms: a randomised trial. Lancet 2002, 360:1267–1274.
Adams RJ: Stroke prevention and treatment in sickle cell disease. Arch Neurol 2001, 58:565–568.
Adams RJ, McKie VC, Hsu L, et al.: Prevention of a first stroke by transfusions in children with sickle cell anemia and abnormal results on transcranial Doppler ultrasonography. N Eng J Med 1998, 339:5–11.
Adams RJ: Lessons from Stroke Prevention Trial in Sickle Cell Anemia (STOP) Study. J Child Neurol 2000, 15:344–349.
Wong WY, Powars DR: Overt and incomplete (silent) cerebral infarction in sickle cell anemia: diagnosis and management. Hematol Oncol Clin N Am 2005, 19:839–855.
Adams RJ, Brambilla D: Optimizing Primary Stroke Prevention in Sickle Cell Anemia (STOP 2) Trial Investigators: Discontinuing prophylactic transfusions used to prevent stroke in sickle cell disease. N Engl J Med 2005, 353:2769–2778. This is a randomized, controlled study to address the duration of chronic transfusion therapy required before it can be stopped safely and without reverting back to the increased stroke risk. The study was stopped after 79 patients were enrolled; of the 41 patients who did not receive transfusions, high-risk TCD changes developed in 14, and two other patients had a stroke. Neither of these events occurred in the 38 patients who continued to receive transfusions.
Cohen AR, Martin MB, Silber JH, et al.: A modified transfusion program for prevention of stroke in sickle cell disease. Blood 1992, 79:1657–1661.
The Management of Sickle Cell Disease. NIH Publication No. 02-2117. Bethesda, MD: National Institutes of Health; 2002.
Brittenham GM, Badman DG: Noninvasive measurement of iron: report of NIDDK workshop. Blood 2003, 101:15–19.
Bernaudin F, Verlhac S, Coic L, et al.: Long-term followup of pediatric sickle cell disease patients with abnormal high velocities on transcranial Doppler. Pediatr Radiol 2005, 35:242–248.
Gulbis B, Haberman D, Dufour D, et al.: Hydroxyurea for sickle cell disease in children and for prevention of cerebrovascular events: the Belgian experience. Blood 2005, 105:2685–2690.
Nietert PJ, Abboud MR, Silverstein MD, et al.: Bone marrow transplantation versus periodic prophylactic blood transfusion in sickle cell patients at high risk of ischemic stroke: a decision analysis. Blood 2000, 95:3057–3064.
Pegelow CH, Adams RJ, McKie V, et al.: Risk of recurrent stroke in patients with sickle cell disease treated with erythrocyte transfusions. J Pediatr 1995, 126:896–899.
Stroke with Transfusions Changing to Hydroxyurea (SWiTCH) Trial. http://clinicaltrials.gov/show/nct00122980. Accessed August 8, 2006.
Abboud MR, Atweh GF: Prevention and management of strokes in patients with sickle cell disease. Curr Hematol Rep 2006, 5:15–22.
Walters MC, Storb R, Patience M, et al.: Impact of bone marrow transplantation for symptomatic sickle cell disease: an interim report. Blood 2000, 95:1918–1924.
Silent Cerebral Infarct Multi-center Clinical Trial. http://clinicaltrials.gov/show/NCT00072761. Accessed August 8, 2006.
Aspirin Prophylaxis in Sickle Cell Disease. http://clinicaltrials.gov/show/nct00178464. Accessed August 8, 2006.
SALT: Alternative Donor Bone Marrow and Cord Blood Transplantation for High-risk Sickle Cell Disease. http://www.clinicaltrials.gov/show/nct00257543. Accessed August 8, 2006.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Mehta, S.H., Adams, R.J. Treatment and prevention of stroke in children with sickle cell disease. Curr Treat Options Neurol 8, 503–512 (2006). https://doi.org/10.1007/s11940-006-0040-8
Issue Date:
DOI: https://doi.org/10.1007/s11940-006-0040-8