Abstract
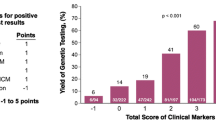
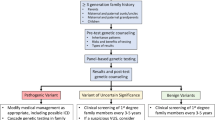
Recent advances have expanded our ability to conduct a comprehensive genetic evaluation for dilated cardiomyopathy (DCM). By evaluating recent literature, this review aims to bring the reader up-to-date on the genetic evaluation of DCM. Updated guidelines have been published. Mutations in BAG3, including a large deletion, were identified in 2 % of DCM. Truncating mutations in TTN were reported in 25 % of DCM. Two new genes have been reported with autosomal recessive DCM. These studies illustrate the role of improved technologies while raising the possibility of a complex genetic model for DCM. The inclusion of TTN has led to an increased genetic testing detection rate of 40 %. While our ability to identify disease-causing variants has increased, so has the identification of variants of unknown significance. A genetic evaluation for DCM must therefore address this complexity.
Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance
Hershberger RE, Morales A, Siegfried JD. Clinical and genetic issues in dilated cardiomyopathy: a review for genetics professionals. Gen Med. 2010;12:655–67.
Harakalova M, van Harssel JJ, Terhal PA, van Lieshout S, Duran K, Renkens I, et al. Dominant missense mutations in ABCC9 cause cantu syndrome. Nat Genet. 2012;44:793–6.
Lefeber DJ, de Brouwer AP, Morava E, Riemersma M, Schuurs-Hoeijmakers JH, Absmanner B, et al. Autosomal recessive dilated cardiomyopathy due to DOLK mutations results from abnormal dystroglycan O-mannosylation. PLoS Genet. 2011. DOLK mutations are reported as a cause of primarily non syndromic autosomal recessive DCM in 11 pediatric cases.
Vasan R, Larson MG, Levy D, Evans JC, Benjamin EJ. Distribution and categorization of echocardiographic measurements in relation to reference limits. The Framingham heart study: formulation of a height- and sex-specific classification and its prospective validation. Circulation. 1997;96:1863–73.
Burkett EL, Hershberger RE. Clinical and genetic issues in familial dilated cardiomyopathy. J Am Coll Cardiol. 2005;45:969–81.
Mestroni L, Maisch B, McKenna WJ, Schwartz K, Charron P, Rocco C, et al. Guidelines for the study of familial dilated cardiomyopathies. Eur Heart J. 1999;20:93–102.
Elliott P, Andersson B, Arbustini E, Bilinska Z, Cecchi F, Charron P, et al. Classification of the cardiomyopathies: a position statement from the European society of cardiology working group on myocardial and pericardial diseases. Eur Heart J. 2008;29:270–6.
Morales A, Pinto JR, Siegfried JD, Li D, Norton N, Hofmeyer M, et al. Late onset sporadic dilated cardiomyopathy caused by a cardiac troponin T mutation. Clin Trans Sci. 2010;3:219–26.
Vatta M, Mohapatra B, Jimenez S, Sanchez X, Faulkner G, Perles Z, et al. Mutations in Cypher/ZASP in patients with dilated cardiomyopathy and left ventricular noncompaction. J Am Coll Cardiol. 2003;42:2014–27.
Moller DV, Andersen PS, Hedley P, Ersboll MK, Bundgaard H, Moolman-Smook J, et al. The role of sarcomere gene mutations in patients with idiopathic dilated cardiomyopathy. Eur J Hum Genet. 2009;17:1241–9.
Sen-Chowdhry S, Lowe MD, Sporton SC, McKenna WJ. Arrhythmogenic right ventricular cardiomyopathy: clinical presentation, diagnosis, and management. Am J Med. 2004;117:685–95.
Sen-Chowdhry S, Syrris P, Prasad SK, Hughes SE, Merrifield R, Ward D, et al. Left-dominant arrhythmogenic cardiomyopathy: an under-recognized clinical entity. J Am Coll Cardiol. 2008;52:2175–87.
Rizzo S, Pilichou K, Thiene G, Basso C. The changing spectrum of arrhythmogenic (right ventricular) cardiomyopathy. Cell Tissue Res. 2012;348:319–23.
Maron BJ, Towbin JA, Thiene G, Antzelevitch C, Corrado D, Arnett D, et al. Contemporary definitions and classification of the cardiomyopathies: an American heart association scientific statement from the council on clinical cardiology, heart failure and transplantation committee; quality of care and outcomes research and functional genomics and translational biology interdisciplinary working groups; and council on epidemiology and prevention. Circulation. 2006;113:1807–16.
Hershberger RE, Siegfried JD. State of the art review. Update 2011: clinical and genetic issues in familial dilated cardiomyopathy. J Am Coll Cardiol. 2011;57:1641–9.
Sivasankaran S, Sharland GK, Simpson JM. Dilated cardiomyopathy presenting during fetal life. Cardiol Young. 2005;15:409–16.
Rampersaud E et al. Rare variant mutations identified in pediatric patients with dilated cardiomyopathy. Prog Pediatr Cardiol. 2011;31:39–47. Clinical and molecular genetic data from pediatric DCM cases were analyzed, showing that nonsynonymous rare variants, predominantly in sarcomeric genes, are causative of pediatric DCM.
Elkayam U. Clinical characteristics of peripartum cardiomyopathy in the United States: diagnosis, prognosis, and management. J Am Coll Cardiol. 2011;58:659–70.
Rajagopalan N, Attili AK, Bodiwala K, Bailey AL. Features of left ventricular noncompaction in peripartum cardiomyopathy: a case series. Int J Cardiol. 2012.
Michels M, Hoedemaekers YM, Kofflard MJ, Frohn-Mulder I, Dooijes D, Majoor-Krakauer D, et al. Familial screening and genetic counselling in hypertrophic cardiomyopathy: the Rotterdam experience. Neth Heart J. 2007;15:184–90.
Grunig E, Tasman JA, Kucherer H, Franz W, Kubler W, Katus HA. Frequency and phenotypes of familial dilated cardiomyopathy [see comments]. J Am Coll Cardiol. 1998;31:186–94.
Baig MK, Goldman JH, Caforio AP, Coonar AS, Keeling PJ, McKenna WJ. Familial dilated cardiomyopathy: cardiac abnormalities are common in asymptomatic relatives and may represent early disease. J Am Coll Cardiol. 1998;31:195–201.
Kushner JD, Nauman D, Burgess D, Ludwigsen S, Parks S, Pantely G, et al. Clinical characteristics of 304 kindreds evaluated for familial dilated cardiomyopathy. J Cardiac Failure. 2006;12:422–9.
Elliott P, O'Mahony C, Syrris P, Evans A, Rivera Sorensen C, Sheppard MN, et al. Prevalence of desmosomal protein gene mutations in patients with dilated cardiomyopathy. Circ Cardiovasc Genet. 2010;3:314–22.
Posch MG et al. A missense variant in desmoglein-2 predisposes to dilated cardiomyopathy. Mol Genet Metab. 2008;95:74–80.
Garcia-Pavia P, Syrris P, Salas C, Evans A, Mirelis JG, Cobo-Marcos M, et al. Desmosomal protein gene mutations in patients with idiopathic dilated cardiomyopathy undergoing cardiac transplantation: a clinicopathological study. Heart. 2011;97:1744. Mutations in desmosomal genes were identified in patients with advanced DCM, suggesting that the addition of these genes may improve the yield of genetic testing for DCM.
Murphy RT, Mogensen J, Shaw A, Kubo T, Hughes S, McKenna WJ. Novel mutation in cardiac troponin I in recessive idiopathic dilated cardiomyopathy. Lancet. 2004;363:371–2.
Towbin JA, Hejtmancik JF, Brink P, Gelb B, Zhu XM, Chamberlain JS, et al. X-linked dilated cardiomyopathy. Circulation. 1993;87:1854–65.
Zaragoza MV, Brandon MC, Diegoli M, Arbustini E, Wallace DC. Mitochondrial cardiomyopathies: how to identify candidate pathogenic mutations by mitochondrial DNA sequencing, MITOMASTER and phylogeny. Eur J Hum Genet. 2011;19:200–7.
Theis JL, Sharpe KM, Matsumoto ME, Chai HS, Nair AA, Theis JD, et al. Homozygosity mapping and exome sequencing reveal GATAD1 mutation in autosomal recessive dilated cardiomyopathy. Circ Cardiovasc Genet. 2011;4:585–94. Using exome sequencing and homozygosity mapping in a consanguineous family, a homozygous variant in a novel gene associated with autosomal recessive DCM was identified. The gene product, GATA zinc finger domain-containing protein 1, is relevant for gene expression.
Norton N, Li D, Reider MJ, Siegfried JD, Rampersaud E, Zuchner S, et al. Genome-wide studies of copy number variation and exome sequencing identify rare variants in BAG3 as a cause of dilated cardiomyopathy. Am J Hum Genet. 2011;88:273–82. Variants in a novel gene, including a large deletion, associated with autosomal dominant, non syndromic DCM are reported for the first time.
Herman DS, Lam L, Taylor MR, Wang L, Teekakirikul P, Christodoulou D, et al. Truncations of titin causing dilated cardiomyopathy. N Engl JMed. 2012;366:619–28. Comprehensive evaluation of TTN suggesting that truncating mutations cause 25% of DCM.
Norton N, Robertson PD, Rieder MJ, Zuchner S, Rampersaud E, Martin E, et al. Evaluating pathogenicity of rare variants from dilated cardiomyopathy in the Exome era. Circ Cardiovasc Genet. 2012;5:167–74.
Selcen D, Muntoni F, Burton BK, Pegoraro E, Sewry C, Bite AV, et al. Mutation in BAG3 causes severe dominant childhood muscular dystrophy. Ann Neurol. 2009;65:83–9.
Schonberger J, Wang L, Shin JT, Kim SD, Depreux FF, Zhu H, et al. Mutation in the transcriptional coactivator EYA4 causes dilated cardiomyopathy and sensorineural hearing loss. Nat Genet. 2005;37:418–22.
Gupta P, Bilinska ZT, Sylvius N, Boudreau E, Veinot JP, Labib S, et al. Genetic and ultrastructural studies in dilated cardiomyopathy patients: a large deletion in the lamin A/C gene is associated with cardiomyocyte nuclear envelope disruption. Basic Res Cardiol. 2010;105:365–77.
Norton N, Siegfried JD, Li D, Hershberger RE. Assessment of LMNA copy number variation in 58 probands with dilated cardiomyopathy. Clin Transl Sci. 2011;4:351–2.
Bang ML, Centner T, Fornoff F, Geach AJ, Gotthardt M, McNabb M, et al. The complete gene sequence of titin, expression of an unusual approximately 700-kDa titin isoform, and its interaction with obscurin identify a novel Z-line to I-band linking system. Circ Res. 2001;89:1065–72.
Guo W, Bharmal SJ, Esbona K, Greaser ML. Titin diversity—alternative splicing gone wild. J Biomed Biotechnol. 2010;2010:753675.
Guo W, Schafer S, Greaser ML, Radke MH, Liss M, Govindarajan T, et al. RBM20, a gene for hereditary cardiomyopathy, regulates titin splicing. Nat Med. 2012;18:766–73.
Brauch KM, Karst ML, Herron KJ, de Andrade M, Pellikka PA, Rodeheffer RJ, et al. Mutations in ribonucleic acid binding protein gene cause familial dilated cardiomyopathy. J Am Coll Cardiol. 2009;54:930–41.
Li D, Morales A, Gonzalez Quintana J, Norton N, Siegfried JD, Hofmeyer M, et al. Identification of novel mutations In RBM20 in patients with dilated cardiomyopathy. Clin Trans Sci. 2010;3:90–7.
Gerull B, Gramlich M, Atherton J, McNabb M, Trombitas K, Sasse-Klaassen S, et al. Mutations of TTN, encoding the giant muscle filament titin, cause familial dilated cardiomyopathy. Nat Genet. 2002;14:14.
Itoh-Satoh M, Hayashi T, Nishi H, Koga Y, Arimura T, Koyanagi T, et al. Titin mutations as the molecular basis for dilated cardiomyopathy. Biochem Biophys Res Commun. 2002;291:385–93.
Norton N, Li D, Rampersaud E, Morales A, Martin ER, Zuchner S, et al. Exome sequencing and genome-wide linkage analysis in 17 families demonstrates the complex contribution of TTN truncating variants to dilated cardiomyopathy. Circ Cardiovasc Genet. 2013;6:144–53.
Hershberger RE, Parks SB, Kushner JD, Li D, Ludwigsen S, Jakobs PM, et al. Coding sequence mutations identified in MYH7, TNNT2, SCN5A, CSRP3, LBD3, and TCAP from 313 patients with familial or idiopathic dilated cardiomyopathy. Clin Transl Sci. 2008;1:21–6.
Hershberger RE, Norton N, Morales A, Li D, Siegfried JD, Gonzalez-Quintana J. Coding sequence rare variants identified in MYBPC3, MYH6, TPM1, TNNC1, and TNNI3 from 312 patients with familial or idiopathic dilated cardiomyopathy. Circ Cardiovasc Genet. 2010;3:155–61.
Hershberger RE, Pinto JR, Parks SB, Kushner JD, Li D, Ludwigsen S, et al. Clinical and functional characterization of TNNT2 mutations identified in patients with dilated cardiomyopathy. Circ Cardiovasc Genet. 2009;2:306–13.
Hershberger RE. A glimpse into multigene rare variant genetics: triple mutations in hypertrophic cardiomyopathy. J Am Coll Cardiol. 2010;55:1454–5.
Kelly M, Semsarian C. Multiple mutations in genetic cardiovascular disease: a marker of disease severity? Circ Cardiovasc Genet. 2009;2:182–90.
Hershberger RE, Lindenfeld J, Mestroni L, Seidman CE, Taylor MR, Towbin JA. Genetic evaluation of cardiomyopathy—a heart failure society of America practice guideline. J Card Fail. 2009;15:83–97.
Hershberger RE, Cowan J, Morales A, Siegfried JD. Progress with genetic cardiomyopathies: screening, counseling, and testing in dilated, hypertrophic, and arrhythmogenic right ventricular dysplasia/cardiomyopathy. Circ Heart Fail. 2009;2:253–61.
Ackerman MJ, Priori SG, Willems S, Berul C, Brugada R, Calkins H, et al. HRS/EHRA expert consensus statement on the state of genetic testing for the channelopathies and cardiomyopathies. This document was developed as a partnership between the Heart Rhythm Society (HRS) and the European Heart Rhythm Association (EHRA). Hear Rhythm. 2011;8:1308–39. Recent clinical guidelines highlighting the value of genetic testing for DCM for diagnostic and prognostic purposes in some cases.
Lakdawala NK, Funke BH, Baxter S, Cirino AL, Roberts AE, Judge DP, et al. Genetic testing for dilated cardiomyopathy in clinical practice. J Card Fail. 2012;18:296–303. Examination of the role of genetic testing for DCM in clinical practice, including a quantification of variants of unknown significance.
Miller EM, Wang Y, Ware SM. Uptake of cardiac screening and genetic testing among hypertrophic and dilated cardiomyopathy families. J Genet Couns. 2012. Chart review of HCM and DCM cases exploring the implementation of cardiac screening and genetic testing guidelines.
Genetic information nondiscrimination act (GINA) of 2008. 2010 [cited 2010 December 17]. A brief history of GINA, with information for researchers and health care professionals. ]. Available from: http://www.genome.gov/24519851.
Aatre RD, Day SM. Psychological issues in genetic testing for inherited cardiovascular diseases. Circ Cardiovasc Genet. 2011;4:81–90. Discussion of psychosocial issues in cardiovascular genetic counseling, including proposed guidelines for counseling and testing in inherited cardiovascular disease.
Morales A, Cowan J, Dagua J, Hershberger RE. Family history: an essential tool for cardiovascular genetic medicine. Congest Heart Fail. 2008;14:37–45.
Nauman D, Morales A, Cowan J, Dagua J, Hershberger RE. The family history as a tool to identify patients at risk for dilated cardiomyopathy. Prog Cardiovasc Nurs. 2008;23:41–4.
Compliance with Ethics Guidelines
ᅟ
Conflict of Interest
Ana Morales declares that she has no conflict of interest. Ray E. Hershberger declares that he has no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is part of the Topical Collection on Congestive Heart Failure
Rights and permissions
About this article
Cite this article
Morales, A., Hershberger, R.E. Genetic Evaluation of Dilated Cardiomyopathy. Curr Cardiol Rep 15, 375 (2013). https://doi.org/10.1007/s11886-013-0375-1
Published:
DOI: https://doi.org/10.1007/s11886-013-0375-1