Abstract
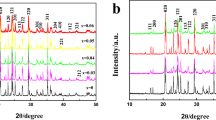
Uncoated and La2O3-coated LiNiO2 cathode materials were synthesized by polymeric sol gel process using metal nitrate precursors at 600 °C for 10 h. The structure and electrochemical properties of the surface-coated LiNiO2 materials were characterized by using X-ray diffraction (XRD), scanning electron microscopy (SEM), cyclic voltammetry, charge/discharge and electrochemical impedance spectroscopy techniques. X-ray powder diffraction and SEM result show that no significant bulk structural differences were observed between the lanthanum oxide coated and pristine LiMn2O4. The galvanostatic charge/discharge studies on the uncoated and lanthanum oxide-coated LiNiO2-positive material at 0.5-C rate in the potential range between 3 and 4.5 V revealed that lanthanum oxide-coated positive electrode material has enhanced charge/discharge capacities; 2.0 wt.% of lanthanum oxide-coated LiNiO2-positive material has satisfied the structural stability, high reversible capacity and high electrochemical performances.







Similar content being viewed by others
References
Liu HK, Wang GX, Guo Z, Wang J, Konstantinov K (2006) J Nanosci Nanotechnol 6:1–15
Whittingham MS (2004) Chem Rev 104:4271
Tarascon JM, Armond M (2001) Nature 414:359
Zhecheva E, Mladenov M, Stoyanova R, Vassilev S (2006) J Power Sources 162:823
Liu HS, Yang Y, Zhang JJ (2006) J Power Sources 162:644
Reddy MV, Subba Rao GV, Chowdari BVR (2006) J Power Sources 160:1369
Dahn JR, Sacken UV, Juzkow MW, Jannaby HA (1991) Electrochem Soc 138:2207
Lee SM, Oh SH, Ahn JP, Cho WI, Jang H (2006) J Power Sources 159:1334–1339
Zhao HL, Gao L, Qiu WH, Zhang XH (2004) J Power Sources 132:195
Fey GTK, Lu CZ, Prem KT, Chang YC (2005) Surf Coat Technol 199:22
Wang H, Tang A, Huang K, Liu S (2010) Trans Nonferrous Metals Soc China 20:803–808
Chowdari BVR, Subba Rao GV, Chow S (2002) J Solid State Electrochem 6:565–567
Ha HW, Jeong KH, Yun NJ, Hongb MZ, Kim K (2005) Electrochim Acta 50:3764–3769
Deng XR, Hu GR, Du K, Peng ZD, Gao XG, Yang YN (2008) Mater Chem Phys 109:469–474
Ma X, Wang C, Han X, Sun J (2008) J Alloys Compd 453:352–355
Guo R, Shi P, Cheng X, Sun L (2009) Electrochim Acta 54:5796–5803
Arumugam D, Paruthimal Kalaignan G (2010) Mater Res Bull 45:1825–1831
Arumugam D, Paruthimal Kalaignan G (2011) Thin Solid Films 520:338–343
Myung ST, Izumi K (2005) Chem Mater 17:3695
Kannan AM, Rabenbery L, Manthiram A (2003) J Electrochem Soc 6:16
Cao H, Xia BJ, Zhang Y, Xu NX (2005) Solid State Ion 176:911
Caballero A, Cruz M, Hernan L, Melero M, Morales J, Castellon ER (2005) J Power Sources 150:192
Hewston TA, Chamberland BL (1987) J Phys Chem Solids 48:97
Kayalvizhi M, John Berchmans L (2010) E-J Chem 7(S1):S137–S142
Sathiyamoorthi R, Vasudevan T (2007) Electrochem Commun 9:416–424
Arumugam D, Paruthimal Kalaignan G, Manisankar P (2008) Solid State Ion 179:580–586
Ohzuku T, Ueda A, Nagayama MJ (1993) Electrochem Soc 140:1862
Prado G, Rougier E, Fournes L, Delmas C (2000) J Electrochem Soc 147(8):2880–2887
Ye XR, Jia DZ, Yu JQ, Xin XQ, Xue Z (1999) Adv Mater 11:941
Sivaprakash S, Majumder SB (2010) J Electrochem Soc 157(4):A418–A422
Smart MC, Luchi BL, Ratnakumar BV (2008) J Electrochem Soc 155(8):A557–A568
Chen CH, Liu J, Amine K (2001) J Power Sources 96:321
Acknowledgments
The authors thank the Department of Physics, Alagappa University, Karaikudi for providing the XRD analysis to carry out this research work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mohan, P., Kalaignan, G.P. Electrochemical performance of La2O3-coated layered LiNiO2 cathode materials for rechargeable lithium-ion batteries. Ionics 19, 895–902 (2013). https://doi.org/10.1007/s11581-012-0821-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11581-012-0821-x