Abstract
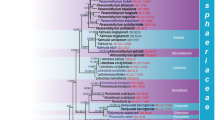
The neglected false truffle species Hydnotrya bailii Soehner (Ascomycetes, Discinaceae) is re-described and separated from its sister taxon Hydnotrya tulasnei by morphological and phylogenetic analyses based on internal transcribed spacer rDNA sequences. The most distinct morphological and ecological characters are small globose, rather than kidney-like, ascomata as known from the sister taxon H. tulasnei, strictly monoseriate ascospores and montane habitats. Phylogenetic analyses resulted in two clearly separated clusters that revealed the ectomycorrhizal specificity of H. bailii to Picea abies and that H. tulasnei is preferably associated to Fagus sylvatica. We also show that H. bailii was already present in mycorrhizal samples but until now could not be correctly assigned. Our analyses also indicate cryptic diversity within H. cerebriformis and other, morphologically not yet characterized, Hydnotrya groups. An emended determination key for all Hydnotrya species known from Central Europe is provided.






Similar content being viewed by others
References
Albee-Scott SR (2007) Does secotioid inertia drive the evolution of false-truffles? Mycol Res 111:1030–1039. doi:10.1016/j.mycres.2007.08.008
Altschul SF, Gish W, Miller W, Myers EW, Lipman DJ (1990) Basic local alignment search tool. J Mol Biol 215:403–410
Binder M, Bresinsky A (2002) Derivation of a polymorphic lineage of Gasteromycetes from boletoid ancestors. Mycologia 94:85–98
Brock M, Döring H, Bidartondo MI (2009) How to know unknown fungi: the role of a herbarium. New Phytol 181:719–724. doi:10.1111/j.1469-8137.2008.02703.x
Castresana J (2000) Selection of conserved blocks from multiple alignments for their use in phylogenetic analysis. Mol Biol Evol 17:540–552
Dickie IA, Dentinger BTM, Avis PG, McLaughlin DJ, Reich PB (2009) Ectomycorrhizal fungal communities of oak savanna are distinct from forest communities. Mycologia. doi:10.3852/08-178
Felsenstein J (1981) Evolutionary trees from DNA sequences: a maximum likelihood approach. J Mol Evol 17:368–376
Fitch WM (1971) Towards defining the course of evolution: minimal change for a specified tree topology. Syst Zool 20:406–416
Gardes M, Bruns TD (1993) ITS primers with enhanced specificity for basidiomycetes application to the identification of mycorrhizae and rusts. Mol Ecol 2:113–118. doi:10.1111/j.1365-294X.1993.tb00005.x
Göker M, García-Blázquez G, Voglmayr H, Tellería MT, Martín MP (2009) Molecular taxonomy of phytopathogenic fungi: a case study in Peronospora. PLoS One 4:e6319. doi:10.1371/journal.pone.0006319
Hawker L (1954) British hypogeous fungi. Phil Trans Roy Soc London 237:429–546
Hesse R (1890) Die Hypogaeen Deutschlands Band II. Verlag L Hoffstetter, Halle
Hibbett DS, Pine EM, Langer E, Langer G, Donoghue MJ (1997) Evolution of gilled mushrooms and puffballs inferred from ribosomal DNA sequences. PNAS 94:12002–12006
Hollos L (1911) Magyarország földalatti gombái, Szarvasgombaféléi (Fungi Hypogaei Hungariae). Budapest, Kiadja
Ishida TA, Nara K, Hogetsu T (2007) Host effects on ectomycorrhizal fungal communities: insight from eight host species in mixed conifer-broad leaf forests. New Phytol 174:430–440. doi:10.1111/j.1469-8137.2007.02016.x
Laesso T, Hansen K (2007) Truffle trouble: what happened to the Tuberales. Mycol Res 111:1075–1099. doi:10.1016/j.mycres.2007.08.004
Lack B (2003) Hydnotrya cubispora in Wales. Field Mycol 4:4
Lee C, Grasso C, Sharlow MF (2002) Multiple sequence alignment using partial order graphs. Bioinformatics 18:452–464
Montecchi A, Sarasini M (2000) Fungi ipogei d’Europa. AMB, Trento
Moser M (1963) Ascomyceten (Schlauchpilze) Band IIa. Gustav Fischer Verlag, Stuttgart
Münzenberger B, Bubner B, Wöllecke J, Sieber TN, Bauer R, Fladung M, Hüttl RF (2009) The ectomycorrhizal morphotype Pinirhiza sclerotia is formed by Acephala macrosclerotiorum sp. nov., a close relative of Phialocephala fortinii. Mycorrhiza, doi:10.1007/s00572-009-0239-0
Ogura-Tsujita Y, Yukawa T (2008) Epipactis helleborine shows strong mycorrhizal preference towards ectomycorrhizal fungi with contrasting geographic distributions in Japan. Mycorrhiza 18:331–338. doi:10.1007/s00572-008-0187-0
Peintner U, Iotti M, Klotz P, Bonuso E, Zambonelli A (2007) Soil fungal communities in a Castanea sativa (chestnut) forest producing large quantities of Boletus edulis sensu lato (porcini): where is the mycelium of porcini? Environ Microbiol 9:880–889. doi:10.1111/j.1462-2920.2006.01208.x
Smith SE, Read DJ (2008) Mycorrhizal symbiosis, Academic press
Soehner E (1959) Tuberaceen Studien V, Mitteilungen der Botanischen Staatssammlung München
Stamatakis A (2006) RAxML-VI-HPC: maximum likelihood-based phylogenetic analyses with thousands of taxa and mixed models. Bioinformatics 22:2688–2690
Stamatakis A, Hoover P, Rougemont J (2008) A rapid bootstrap algorithm for the RAxML web servers. Syst Biol 75:758–771. doi:10.1080/10635150802429642
Swofford DL (2002) PAUP*: Phylogenetic Analysis Using Parsimony (*and Other Methods), Version 4.0 b10. Sinauer Associates, Sunderland
Szemere L (1965) Die unterirdischen Pilze des Karpatenbeckens. Akadémiai Kiadó, Budapest
Thiers HD (1984) The secotioid syndrome. Mycologia 76:1–8
Tedersoo L, Kõljalg U, Hallenberg N, Larsson KH (2003) Fine scale distribution of ectomycorrhizal fungi and roots across substrate layers including coarse woody debris in a mixed forest. New Phytol 159:153–165. doi:10.1046/j.0028-646x.2003.00792.x
Tedersoo L, Hansen K, Perry BA, Kjøller R (2006) Molecular and morphological diversity of pezizalean ectomycorrhiza. New Phytol 170:581–596. doi:10.1111/j.1469-8137.2006.01678.x
Trappe JM (1975) Generic synonyms in the Tuberales. Mycotaxon 2:109–122
Vohnik M, Fendrych M, Kolarik M, Gryndler M, Hrselova H, Albrechtova J, Vosatka M (2007) The ascomycete Meliniomyces variabilis isolated from a sporocarp of Hydnotrya tulasnei (Pezizales) intracellularly colonises roots of ecto- and ericoid mycorrhizal host plants. Czech Mycol 59:215–226
White TJ, Bruns TD, Lee S, Taylor J (1990) Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In: Innis MA, Gelfand DH, Sninsky JJ, White TJ (eds) PCR Protocols: a guide to methods and applications. Academic Press, New York, pp 315–322
Zeller SM, Dodge CW (1924) Leucogaster and Leucophlebs in North America. Ann Mo Bot Gard 11:389–410
Zhang BC (1991) Morphology, cytology and taxonomy of Hydnotrya cerebriformis (Pezizales). Mycotaxon 42:155
Acknowledgements
We thank M. Roth, Müncheberg for technical assistance.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
These files are unfortunately not in the Publisher's archive anymore:
-
Supplementary 1 (NEX 73.3 kb)
-
Supplementary 2 (NEX 138 kb)
Rights and permissions
About this article
Cite this article
Stielow, B., Bubner, B., Hensel, G. et al. The neglected hypogeous fungus Hydnotrya bailii Soehner (1959) is a widespread sister taxon of Hydnotrya tulasnei (Berk.) Berk. & Broome (1846). Mycol Progress 9, 195–203 (2010). https://doi.org/10.1007/s11557-009-0625-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11557-009-0625-1