Abstract
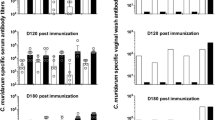
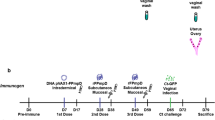
To validate the immune protective efficacy of pORF5 DNA vaccine and to analyze potential mechanisms related to this protection. In this study, pORF5 DNA vaccine was constructed and evaluated for its protective immunity in a mouse model of genital chlamydial infection. Groups of BALB/c mice were immunized intranasally with pORF5 DNA vaccine. Humoral and cell mediated immune responses were evaluated. The clearance ability of chlamydial challenge from the genital tract and the chlamydia-induced upper genital tract gross pathology and histopathological characterization were also detected. The results showed that the total and the IgG2a anti-pORF5 antibody levels in serum were significantly elevated after pcDNA3.1-pORF5 vaccination, as were the total antibody and IgA levels in vaginal fluids. pcDNA3.1-pORF5 induced a significantly high level of Th1 response as measured by robust gamma interferon (IFN-γ). Minimal IL-4 was produced by immune T cells in response to the re-stimulation with pORF5 protein or the inactive elementary body in vitro. pcDNA3.1-pORF5-vaccinated mice displayed significantly reduced bacterial shedding upon a chlamydial challenge and an accelerated resolution of infection. 100% of pcDNA3.1-pORF5 vaccinated mice successfully resolved the infection by day 24. pcDNA3.1-pORF5-immunized mice also exhibited protection against pathological consequences of chlamydial infection. The stimulated index was significantly higher than that of mice immunized with pcDNA3.1 and PBS (P<0.05). Together, these results demonstrated that immunization with pORF5 DNA vaccine is a promising approach for eliciting a protective immunity against a genital chlamydial challenge.
Article PDF
Similar content being viewed by others
References
Brunham R C, Rey-Ladino J. Immunology of Chlamydia infection: Implications for a Chlamydia trachomatis vaccine. Nat Rev Immunol, 2005, 5: 149–161, 15688042, 10.1038/nri1551, 1:CAS:528:DC%2BD2MXovFKnsA%3D%3D
Kilmarx P H, Mock P A, Levine W C. Effect of Chlamydia trachomatis co-infection on HIV shedding in genital tract secretions. Sex Transm Dis, 2001, 28(6): 347–348, 11403193, 10.1097/00007435-200106000-00008, 1:STN:280:DC%2BD3MzjsFOjtg%3D%3D
Smith J S, Muñoz N, Herrero R, et al. Evidence for Chlamydia trachomatis as a human papillomavirus cofactor in the etiology of invasive cervical cancer in Brazil and the Philippines. J Infect Dis, 2002, 185(3): 324–331, 11807714, 10.1086/338569
Tamim H, Finan R R, Sharida H E, et al. Cervicovaginal coinfections with human papillomavirus and Chlamydia trachomatis. Diagn Microbiol Infect Dis, 2002, 43(4): 277–281, 12151187, 10.1016/S0732-8893(02)00403-0
Wallin K L, Wiklund F, Luostarinen T, et al. A population-based prospective study of Chlamydia trachomatis infection and cervical carcinoma. Int J Cancer, 2002, 101(4): 371–374, 12209962, 10.1002/ijc.10639, 1:CAS:528:DC%2BD38XmvFWiu7c%3D
Igietseme J U, Murdin A. Induction of protective immunity against Chlamydia trachomatis genital infection by a vaccine based on major outer membrane protein-lipophilic immune response-stimulating complex. Infect Immun, 2000, 68(12): 6798–6806, 11083798, 10.1128/IAI.68.12.6798-6806.2000, 1:CAS:528:DC%2BD3MXjslCktLo%3D
Zhang D, Yang X, Berry J, et al. DNA vaccination with the major outer-membrane protein gene induces acquired immunity to Chlamydia trachomatis (mouse pneumontis) infection. J Infect Dis, 1997, 176(4): 1035–1040, 9333163, 1:CAS:528:DyaK2sXms12msbc%3D, 10.1086/516545
Zhang D J, Yang X, Shen C, et al. Characterization of immune responses following intramuscular DNA immunization with the MOMP gene of Chlamydia trachomatis mouse pneumonitis strain. Immunology, 1999, 96(2): 314–321, 10233711, 10.1046/j.1365-2567.1999.00682.x, 1:CAS:528:DyaK1MXhtFOisrs%3D
Brunham R C, Zhang D J. Transgene as vaccine for Chlamydia. Am Heart J, 1999, 138(5 Pt2): s519–522, 10539864, 10.1016/S0002-8703(99)70291-7, 1:CAS:528:DyaK1MXns1Cltrc%3D
Donati M, Sambri V, Comanducci M, et al. DNA immunization with pgp3 gene of Chlamydia trachomatis inhibits the spread of chlamydial infection from the lower to the upper genital tract in C3H/HeN mice. Vaccine, 2003, 21(11–12): 1089–1093, 12559784, 10.1016/S0264-410X(02)00631-X, 1:CAS:528:DC%2BD3sXmt1ahsg%3D%3D
Li Z, Chen D, Zhong Y, et al. The chlamydial plasmid-encoded protein pgp3 is secreted into the cytosol of Chlamydia-infected cells. Infect Immun, 2008, 76(8): 3415–3428, 18474640, 10.1128/IAI.01377-07, 1:CAS:528:DC%2BD1cXptF2itLs%3D
Li Z, Zhong Y, Lei L, et al. Antibodies from women urogenitally infected with C. trachomatis predominantly recognized the plasmid protein pgp3 in a conformation-dependent manner. BMC Microbiol, 2008, 8: 90–102, 18541036, 10.1186/1471-2180-8-90, 1:CAS:528:DC%2BD1cXotlCisrk%3D
Morrison R P, Caldwell H D. Immunity to murine chlamydial genital infection. Infect Immun, 2002, 70(6): 2741–2751, 12010958, 10.1128/IAI.70.6.2741-2751.2002, 1:CAS:528:DC%2BD38XktFGitrs%3D
Brunham R C, Kimani J, Bwayo J, et al. The epidemiology of Chlamydia trachomatis within a sexually transmitted diseases core group. J Infect Dis, 1996, 173: 950–956, 8603976, 1:CAS:528:DyaK28XisVCitbo%3D
Kimani J, Maclean I W, Bwayo J J, et al. Risk factors for Chlamydia trachomatis pelvic inflammatory disease among sex workers in Nairobi, Kenya. J Infect Dis, 1996, 173: 1437–1444, 8648217, 1:STN:280:DyaK283hslOnsA%3D%3D
Holland M J, Bailey R L, Conway D J, et al. T helper type-1 (Th1)/Th2 profiles of peripheral blood mononuclear cells (PBMC); responses to antigens of Chlamydia trachomatis in subjects with severe trachomatous scarring. Clin Exp Immunol, 1996, 105(3): 429–435, 8809130, 10.1046/j.1365-2249.1996.d01-792.x, 1:STN:280:DyaK28vgsV2qsg%3D%3D
Faal N, Bailey R L, Sarr I, et al. Temporal cytokine gene expression patterns in subjects with trachoma identify distinct conjunctival responses associated with infection. Clin Exp Immunol, 2005, 142(2): 347–353, 16232223, 10.1111/j.1365-2249.2005.02917.x, 1:STN:280:DC%2BD2Mrls1SktA%3D%3D
Morrison R P, Caldwell H D. Immunity to murine chlamydial genital infection. Infect Immun, 2002, 70(6): 2741–2751, 12010958, 10.1128/IAI.70.6.2741-2751.2002, 1:CAS:528:DC%2BD38XktFGitrs%3D
Yang X. Role of cytokines in Chlamydia trachomatis protective immunity and immunopathology. Curr Pharm Des, 2003, 9(1): 67–73, 12570676, 10.2174/1381612033392486, 1:CAS:528:DC%2BD3sXisVarsQ%3D%3D
Loomis W P, Stambach M N. T cell responses to Chlamydia trachomatis. Curr Opin Microbiol, 2002, 5(1): 87–91, 11834375, 10.1016/S1369-5274(02)00291-6, 1:CAS:528:DC%2BD38XhtVKqsbs%3D
Darville T, Andrews C W Jr, Rank R G. Does inhibition of tumor necroses factor alpha affect chlamydial genital tract infection in mice and guinea pigs? Infect Immun, 2000, 68(9): 5299–5305, 10948158, 10.1128/IAI.68.9.5299-5305.2000, 1:CAS:528:DC%2BD3cXmtVektr0%3D
Perfettini J L, Darville T, Gachelin G, et al. Effect of Chlamydia trachomatis infection and subsequent tumor necrosis factor alpha secretion on apoptosis in the murine genital tract. Infect Immun, 2000, 68(4): 2237–2244, 10722625, 10.1128/IAI.68.4.2237-2244.2000, 1:CAS:528:DC%2BD3cXitFels7w%3D
Wang S, Fan Y, Brunham R C, et al. IFN-gamma knockout mice show Th2-associated delayed-type hypersensitivity and inflammatory cells fail to localize and control chlamydial infection. Eur J Immunol, 1999, 29(11): 3782–3792, 10556835, 10.1002/(SICI)1521-4141(199911)29:11<3782::AID-IMMU3782>3.0.CO;2-B, 1:CAS:528:DyaK1MXnt1Ogurc%3D
Lu H, Yang X, Takeda K, et al. Chlamydia trachomatis mouse pneumonitis lung infection in IL-18 and IL-12 knockout mice: IL-12 is dominant over IL-18 for protective immunity. Mol Med, 2000, 6(7): 604–612, 10997341, 1:CAS:528:DC%2BD3cXmvFWqurw%3D
Rottenberg M E, Gigliotti Rothfuchs A C, Gigliotti D, et al. Regulation and role of IFN-gamma in the innate resistance to infection with Chlamydia pneumoniae. J Immunol, 2000, 164(9): 4812–4818, 10779789, 1:CAS:528:DC%2BD3cXivFWmurY%3D
Sharma M, Sethi S, Daftari S, et al. Evidence of Chlamydia infection in infertile women with fallopian tube obstruction. Indian J Pathol Microbiol, 2003, 46(4): 680–683, 15025381
Igietseme J U, Ramsey K H, Magee D M, et al. Resolution of murine chlamydial genital infection by the adoptive transfer of a biovar-specific, Th1 lymphocyte clone. Reg Immunol, 1993, 5(6): 317–324, 8068534, 1:STN:280:DyaK2czkslGqtg%3D%3D
Cotter T W, Meng Q, Shen Z L, et al. Protective efficacy of major outer membrane protein-specific immunoglobulin A (IgA) and IgG monoclonal antibodies in a murine model of Chlamydia trachomatis genital tract infection. Infect Immun, 1995, 63(12): 4704–4714, 7591126, 1:CAS:528:DyaK2MXpslClsLs%3D
Author information
Authors and Affiliations
Corresponding authors
Additional information
Supported by the National High-Tech Research and Development Program of China (Grant No. 2004AA2Z3530), the Hunan Province “Eleventh Five-Year Plan” Important Special Program (Grant No. 2006SK1001), and the National Institutes of Health, USA (Grant No. 1 R01 AI47997-01)
Rights and permissions
About this article
Cite this article
Li, Z., Wang, S., Wu, Y. et al. Immunization with chlamydial plasmid protein pORF5 DNA vaccine induces protective immunity against genital chlamydial infection in mice. SCI CHINA SER C 51, 973–980 (2008). https://doi.org/10.1007/s11427-008-0130-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-008-0130-9