Abstract
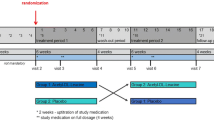
Cuban patients with Spinocerebellar Ataxia type 2 (SCA2) have reduced concentrations of zinc in serum and cerebrospinal fluid (CSF). To assess the effect and safety of zinc supplementation, 36 Cuban SCA2 patients were randomly assigned to receive daily either 50 mg ZnSO4 or placebo, together with neurorehabilitation therapy in a randomized, double-blind, placebo-controlled clinical trial during 6 months. Outcome measures included the changes of zinc levels in CSF and serum, ataxia score, oxidative stress and saccadic eye movements. At the end of the study, the Zinc-treated group showed: (i) a significant increase of the Zn levels in the CSF, (ii) mild decrease in the ataxia scale subscores for gait, posture, stance and dysdiadochocinesia (iii) reduction of lipid’s oxidative damage, and (iv) reduction of saccadic latency when compared with the placebo group. The treatment was safe and well tolerated by all subjects. This study demonstrated the efficacy and safety of Zn supplementation, combined with neurorehabilitation for SCA2 patients and therefore it may encourage further studies on the clinical effect of zinc supplementation in SCA2 based in the conduction of future clinical trials with higher number of subjects.




Similar content being viewed by others
References
Velázquez-Pérez L, Rodríguez-Labrada R, García-Rodríguez JC et al. (2011) Comprehensive review of spinocerebellar ataxia type 2. Cerebellum doi:10.1007/s12311-011-0265-2
Velazquez Perez L, Cruz GS, Santos Falcon N et al (2009) Molecular epidemiology of spinocerebellar ataxias in Cuba: insights into SCA2 founder effect in Holguin. Neurosci Lett 454(2):157–160
Underwood BR, Rubinsztein DC (2008) Spinocerebellar ataxias caused by polyglutamine expansions: a review of therapeutic strategies. Cerebellum 7(2):215–221
Gonzalez C, Sánchez G, Quevedo AG et al (2005) Serum and cerebrospinal fluid levels of copper, iron and zinc in patiens with ataxia type SCA-2 from the province of Holguin in Cuba. Therapeutic Basic Dial Clin Neurosci 13(4):12–16
World Health Organization Committee (1996) Zinc. In: Trace elements in human nutrition and health. WHO, Geneva, pp 72–104
Black MM (2003) The evidence linking zinc deficiency with children’s cognitive and motor functioning. J Nutr 133:1473S–1476S
Wall MJ (2005) A role for zinc in cerebellar synaptic transmission? Cerebellum 4:224–229
Fosmire G (1990) Zn toxicity. Am J Clin Nutr 51:225–227
Hooper PL, Visconti L, Garry PJ et al (1980) Zinc lowers high-density lipoprotein-cholesterol levels. J Am Med Assoc 244:1960–1961
Haase H, Overbeck S, Rink L (2008) Zinc supplementation for the treatment or prevention of disease: Current status and future perspectives. Exp Gerontol 48:394–408
Schmitz-Hubsch T, du Montcel ST, Baliko L et al (2006) Scale for the assessment and rating of ataxia: development of a new clinical scale. Neurology 66(11):1717–1720
Esterbaver H, Cheeseman K (1990) Determination of aldehydic lipid peroxidation product: Malondialdehyde and 4-hydroxynonenal. Meth Enzymol 186:407–421
Erdelmeier I, Gerard D, Yadan J et al (1998) Reactions of N methyl-2-phenyl-indole with malondialdehyde and 4-hydroxialkenals. Mechanistic aspects of the colorimetric assay of lipid peroxidation. Chem Res Toxicol 11(10):1184–1194
Mannheim B (1987) Biochemica information. A revised biochemical reference source enzymes for routine, 1st edn. Boehringer Mannheim, Berlin, pp 15–16
Shukla G, Hussain T, Chandra S (1987) Possible role of superoxide dismutase activity and lipid peroxide levels in cadmium neurotoxicity: in vivo and in vitro studies in growing rats. Life Sci 14:2215–2225
Witko-Sarsat V, Gausson V, Descamps-Latscha B (2003) Are advanced oxidation protein products potential uremic toxins? Kidney Int Suppl 84:S11–S14
Dvergsten CL, Fosmire GJ, Ollerich DA et al (1984) Alterations in the postnatal development of the cerebellar cortex due to zinc deficiency. II. Impaired maturation of Purkinje cells. Brain Res 318:11–20
Dvergsten CL, Johnson LA, Sandstead HH (1984) Alterations in the postnatal development of the cerebellar cortex due to zinc deficiency. III. Impaired dendritic differentiation of basket and stellate cells. Brain Res 318:21–26
Sensi SL, Paoletti P, Bush AI et al (2009) Zinc in the physiology and pathology of the CNS. Nat Rev Neurosci 10:780–791
Oteiza PI, Olin KL, Fraga CG et al (1995) Zinc deficiency causes oxidative damage to proteins, lipids and DNA in rat testes. J Nutr 125:823–829
Mackenzie GG, Keen CL, Oteiza PI (2002) Zinc status of human IMR-32 neuroblastoma cells influences their susceptibility to iron-induced oxidative stress. Dev Neurosci 24:125–133
Mackenzie GG, Zago MP, Keen CL et al (2002) Low intracellular zinc impairs the translocation of activated NF-kB to the nuclei in human neuroblastoma IMR-32 cells. J Biol Chem 277:34610–34617
Bao B, Prasad A, Beck F et al (2010) Zinc decreases C-reactive protein, lipid peroxidation, and inflammatory cytokines in elderly subjects: a potential implication of zinc as an atheroprotective agent. Am J Clin Nutr 91(6):1634–1641
Farinati F, Cardin R, D’inca R et al (2003) Zinc treatment prevents lipid peroxidation and increases glutathione availability in Wilson’s disease. J Lab Clin Med 141(6):372–377
Marjani A (2005) Plasma lipid peroxidation zinc and erythrocyte Cu-Zn superoxide dismutase enzyme activity in patients with type 2 diabetes mellitus in Gorgan City (South East of the Caspian Sea). Internet J Endocrinol 2(1):1647–1648
Velazquez-Perez L, Seifried C, Santos-Falcon N et al (2004) Saccade velocity is controlled by polyglutamine size in spinocerebellar ataxia 2. Ann Neurol 56(3):444–447
Velazquez-Perez L, Seifried C, Abele M et al (2009) Saccade velocity is reduced in presymptomatic spinocerebellar ataxia type 2. Clin Neurophysiol 120(3):632–635
Buttner N, Geschwind D, Jen JC et al (1998) Oculomotor phenotypes in autosomal dominant ataxias. Arch Neurol 55(10):1353–1357
Rodríguez-Labrada R, Velázquez-Pérez L, Seigfried C et al. (2011) Saccadic latency is prolonged in spinocerebellar ataxia type 2 and correlates with the frontal-ecutive dysfunctions. J Neurol Sci doi:10.1016/j.jns.2011.03.033
Hutton SB (2008) Cognitive control of saccadic eye movements. Brain Cogn 68(3):327–340
Agget P, Harries J (1979) Current status of zinc in health and disease states. Arch Dis Child 54:909–917
Golub M, Takeuchi P, Keen C et al (1996) Activity and attention in zinc-deprived adolescent monkeys. Am J Clin Nutr 64(6):908–915
Arnold LE, DiSilvestro RA (2005) Zinc in attention-deficit/hyperactivity disorder. J Child Adolesc Psychopharm 15(4):619–627
Acknowledgment
We are grateful to the SCA2 patients, the Cuban Ministry of Health and the Iberoamerican multidisciplinary network for the movement disorders (RIBERMOV) for their cooperation.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Velázquez-Pérez, L., Rodríguez-Chanfrau, J., García-Rodríguez, J.C. et al. Oral Zinc Sulphate Supplementation for Six Months in SCA2 Patients: A Randomized, Double-Blind, Placebo-Controlled Trial. Neurochem Res 36, 1793–1800 (2011). https://doi.org/10.1007/s11064-011-0496-0
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-011-0496-0