Abstract
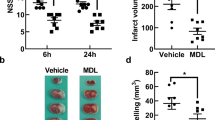
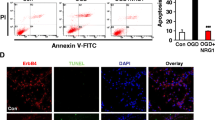
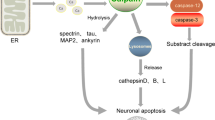
Calpains, cathepsins and caspases play crucial role in mediating cell death. In the present study we observed a cascade of events involving the three proteases during middle cerebral artery occlusion (MCAo) in Wistar rats. The rats were MCA occluded and reperfused at various time points. We observed a maximal increase in the levels of calpains during 1h and 12 h after reperfusion than permanently occluded rats. Further, these levels were reduced by 1st and 3rd day of reperfusion. Similarly the cathepsin-b levels were significantly increased during 1h and 12 h, of reperfusion, followed by activation of caspase-3 which reached maximal levels by 1st and 3rd day of reperfusion. The sequential activation of calpains, cathepsin-b and cleaved caspase-3 is evident by the Western blot analysis which was further confirmed by the cleavage of substrates like PSD-95 and spectrin. The differences in the regional distribution and elevation of these proteases at different reperfusion time periods indicates that differential mode of cell death occur in the brain during cerebral ischemia in rat model.











Similar content being viewed by others
References
Mc Conkey DJ (1998) Biochemical determinants of apoptosis and necrosis. Toxicol Lett 99:157–168
Haunstetter A, Izumo S (1998) Apoptosis: basic mechanisms and implications for cardiovascular disease. Circ Res 82:111–1129
Syntichaki P, Tavernarakis N (2003) The biochemistry of neuronal necrosis: rogue biology? Nat Rev Neurosci 4:672–684
Nicotera P, Leist M, Ferrando-may E (1998) Intracellular ATP, a switch in the decision between apoptosis and necrosis. Toxicol lett 102–103:139–142
Sapolsky RM, Trafton J, Tombaugh GC (1996) Excitotoxic neuron death, acidotic endangerment, and the paradox of acidotic protection. Adv Neurol 71:237–244
Syntichaki P, Samara C, Tavernarakis N (2005) The vacuolar H+ ATPase mediates intracellular acidification required for neurodegeneration in C. elegans. Curr Biol 15:1249–1254
Xiuli L, van Vleet T, Schnellmann RG (2004) The role of Calpain in oncotic cell death. Annu Rev Pharmacol Toxicol 44:349–70
Marta A-S, Samara C, Syntichki P, Tavernarakis N (2006) Lysosomal biogenesis and function is critical for necrotic cell death in Caenorhabditis elegans. The journal of Cell Biology 173:231–239
Yamashima T, Tonchev AB, Tsukada T, Saido TC, Imajoh-Ohmi S, Momoi T, Kominami E (2003) Sustained calpain activation is associated with lysosomal rupture executes necrosis of the post-ischemic CA1 neurons in primates. Hippocampus 13:791–800
Wang KK (2000) Calpain and caspase: can you tell the difference? Trends Neurosci 23:20–26
Lomgren K, Zhu C, Wang X, Karlsson JO, Leverin AL, Bahr BA, Mallard C, Hagberg H (2001) Synergistic activation of caspase-3 by m-calpain after neonatal hypoxia-ischemia: A mechanism of “pathologic apoptosis”. J Biol Chem 276:10191–10198
Kato M, Nonaka T, Maki M, Kikuchi H, Imajoh-Ohmi S (2000) Caspases cleaves the amino-terminal calpain inhibitory unit of calpastatin during apoptosis in human jurkat T cells. J Biochem 127:297–305
Longa EZ, Weinstein PR, Carlson S, Cummins R (1989) Reversible middle cerebral artery occlusion without craniectomy in rats. Stroke 20:84–91
Zhang L, Schallert T, Zhang ZG et al. (2002) A test for detecting long-term sensorimotor dysfunction in the mouse after focal cerebral ischemia. J Neurosci Meth 117:207–214
Cheng Du, Rong Hu, Csernansky CA, Hsu CY, Choi DW (1996) Very Delayed Infarction After Mild Focal Cerebral Ischemia: A Role for Apoptosis? Journal of Cerebral Blood Flow & Metabolism 16:195–201
Lankiewicz S, Marc LC, Truc BN, Krohn AJ, Poppe M, Cole GM, Saido TC, Prehn JH (2000) Activation of calpain I converts excitotoxic neuron death into a caspase-independent cell death. J Biol Chem 275:17064–17071
Xiuli L, van Vleet T, Schnellmann RG (2004) The role of Calpain in oncotic cell death. Annu Rev Pharmacol Toxicol 44:349–70
Stefan JR, Shi Y (2004) Molecular mechanisms of Caspases during apoptosis. Nature Reviews Molecular Cell biology 5:897–907
Charriaut-Marlangue C, Margaill I, Represa A, Popovici T, Plotkine M, Ben-Ari Y (1996) Apoptosis and necrosis after reversible focal ischemia: an in situ DNA fragmentation analysis. J Cereb Blood Flow Metab 16:186–194
Unal C, Dalkara T (2003). Intravenously administered propidium iodide labels necrotic cells in the intact mouse brain after injury. Cell Death and Differentiation 10:928–929
Unal-Çevik I, Munire K, Can A, Gürsoy-Özdemir Y, Dalkara T (2004) Apoptotic and Necrotic death mechanisms are concomitantly activated in the same cell after Cerebral Ischemia. Stroke 35:2189–2194
Gobeil S, Boucher CC, Nadeau D, Poirier GG (2001) Characterization of the necrotic cleavage of poly (ADP-ribose) polymerase (PARP-1): implication of lysosomal proteases. Cell Death Differ 8:588–94
Xiao YL, Rong Y, Baudry M (2000) Calpain-mediated degradation of PSD-95 in developing and adult rat brain. Neuroscience Letters 286:149–153
Yamashima T (2000) Implication of cysteine proteases calpain, cathepsin and caspase in ischemic neuronal death of primates. Prog Neurobiol 62:273–295
Robert WN, Xu A, Gada H, Guttmann RP, Siman R (2003) Cross talk between Calpain and Caspase proteolytic systems during neuronal apoptosis. J Biol Chem 16:1462–1467
Acknowledgements
Financial assistance from DBT and Senior Research fellowship from UGC is gratefully acknowledged. Generous gift of antibodies from Prof. Emmanuel Brouillet for providing PSD-95, Spectrin and Dr. Spencer for Calpain antibody is acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chaitanya, G.V., Babu, P.P. Activation of Calpain, Cathepsin-b and Caspase-3 during Transient Focal Cerebral Ischemia in Rat Model. Neurochem Res 33, 2178–2186 (2008). https://doi.org/10.1007/s11064-007-9567-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-007-9567-7