Abstract
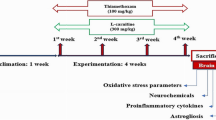
Wernicke’s encephalopathy (WE) is a serious neuropsychiatric syndrome caused by chronic alcoholism and thiamine (T) deficiency. Our aim was to shed more light on the pathophysiology of WE, by introducing a modified in vivo experimental model of WE and by focusing on changes provoked in the total antioxidant status (TAS) and three crucial brain enzyme activities in adult rats. Rats were placed on ethanol (EtOH) consumption (20 % v/v) for a total of 5 weeks. By the end of the third week, rats were fed a T-deficient diet (TDD) and were treated with pyrithiamine (PT; 0.25 mg/kg) for the remaining 2 weeks. Following the induction of WE symptomatology, rats were treated with three consecutive (every 8 h) injections of saline or T (100 mg/kg) and were sacrificed. Brain homogenates were generated and used for spectrophotometrical evaluation of TAS and enzymatic activities. Additionally, in vitro experiments were conducted on brain homogenates or pure enzymes incubated with T or neuromodulatory antioxidants. Pre-exposure to EtOH provided a successful protocol modification that did not affect the expected time of WE symptomatology onset. Administration of T ameliorated this symptomatology. WE provoked oxidative stress that was partially limited by T administration, while T itself also caused oxidative stress to a smaller extent. Brain acetylcholinesterase (AChE) was found inhibited by WE and was further inhibited by T administration. In vitro experiments demonstrated a potential neuroprotective role for L-carnitine (Carn). Brain sodium-potassium adenosine triphosphatase (Na+,K+-ATPase) activity was found increased in WE and was reduced to control levels by in vivo T administration; this increase was also evident in groups exposed to PT or to TDD, but not to EtOH. In vitro experiments demonstrated a potential neuroprotective role for this Na+,K+-ATPase stimulation through T or L-cysteine (Cys) administration. Brain magnesium adenosine triphosphatase (Mg2+-ATPase) activity was found decreased by prolonged exposure to EtOH, but was not affected by the experimental induction of WE. Our data suggest that T administration inhibits AChE, which is also found inhibited in WE. Moreover, increased brain Na+,K+-ATPase activity could be a marker of T deficiency in WE, while combined T and antioxidant co-supplementation of Cys and/or Carn could be neuroprotective in terms of restoring the examined crucial brain enzyme activities to control levels.




Similar content being viewed by others
Notes
TDD was purchased from Mucedola (Italy) and contained: corn starch, casein vitamin free, sucrose, corn oil, cellulose powder, dicalcium phosphate, potassium citrate, calcium carbonate, D,L-methionine, ethoxyquin, vitamin A, vitamin D3, vitamin E, copper and selenium.
PT is an inhibitor of T-pyrophosphokinase, the enzyme that converts T to its active form.
References
Anzalone S, Vetreno RP, Ramos RL, Savage LM (2010) Cortical cholinergic abnormalities contribute to the amnesic state induced by pyrithiamine-induced thiamine deficiency in the rat. Eur J Neurosci 32:847–858
Arendt T, Schugens MM, Bigl V (1990) The cholinergic system and memory: amelioration of ethanol-induced memory deficiency by physostigmine in rat. Acta Neurobiol Exp (Wars) 50:251–261
Bimpis A, Papalois A, Tsakiris S, Kalafatakis K, Zarros A, Gkanti V, Skandali N, Al-Humadi H, Kouzelis C, Liapi C (2013) Modulation of crucial adenosinetriphosphatase activities due to U-74389G administration in a porcine model of intracerebral hemorrhage. Metab Brain Dis, in press
Bowler K, Tirri R (1974) The temperature characteristics of synaptic membrane ATPases from immature and adult rat brain. J Neurochem 23:611–613
Butterworth RF (1986) Cerebral thiamine-dependent enzyme changes in experimental Wernicke’s encephalopathy. Metab Brain Dis 1:165–175
Carageorgiou H, Pantos C, Zarros A, Mourouzis I, Varonos D, Cokkinos D, Tsakiris S (2005) Changes in antioxidant status, protein concentration, acetylcholinesterase, (Na+,K+)- and Mg2+ -ATPase activities in the brain of hyper- and hypothyroid adult rats. Metab Brain Dis 20:129–139
Carageorgiou H, Tzotzes V, Pantos C, Mourouzis C, Zarros A, Tsakiris S (2004) In vivo and in vitro effects of cadmium on adult rat brain total antioxidant status, acetylcholinesterase, (Na+,K+)-ATPase and Mg2+ -ATPase activities: protection by L-cysteine. Basic Clin Pharmacol Toxicol 94:112–118
Carageorgiou H, Zarros A, Tsakiris S (2003) Selegiline long-term effects on brain acetylcholinesterase, (Na+,K+)-ATPase activities, antioxidant status and learning performance of aged rats. Pharmacol Res 48:245–251
Desjardins P, Butterworth RF (2005) Role of mitochondrial dysfunction and oxidative stress in the pathogenesis of selective neuronal loss in Wernicke’s encephalopathy. Mol Neurobiol 31:17–25
EEC Council (1986) EEC Council Directive 86/609/EEC of 24 November 1986 on the approximation of laws, regulations and administrative provisions of the Member States regarding the protection of animals used for experimental and other scientific purposes. Off J Eur Union L358:1–28
Ellman GL, Courtney KD, Andres V Jr, Featherstone RM (1961) A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem Pharmacol 7:88–95
Gibson GE, Zhang H (2002) Interactions of oxidative stress with thiamine homeostasis promote neurodegeneration. Neurochem Int 40:493–504
Hakim AM, Pappius HM (1983) Sequence of metabolic, clinical, and histological events in experimental thiamine deficiency. Ann Neurol 13:365–375
Harper CG, Giles M, Finlay-Jones R (1986) Clinical signs in the Wernicke-Korsakoff complex: a retrospective analysis of 131 cases diagnosed at necropsy. J Neurol Neurosurg Psychiatry 49:341–345
Hazell AS, Butterworth RF (2009) Update of cell damage mechanisms in thiamine deficiency: focus on oxidative stress, excitotoxicity and inflammation. Alcohol Alcohol 44:141–147
Hazell AS, Pannunzio P, Rama Rao KV, Pow DV, Rambaldi A (2003) Thiamine deficiency results in downregulation of the GLAST glutamate transporter in cultured astrocytes. Glia 43:175–184
Hazell AS, Todd KG, Butterworth RF (1998) Mechanisms of neuronal cell death in Wernicke’s encephalopathy. Metab Brain Dis 13:97–122
Homewood J, Bond NW (1999) Thiamin deficiency and Korsakoff’s syndrome: failure to find memory impairments following nonalcoholic Wernicke’s encephalopathy. Alcohol 19:75–84
Huang HM, Chen HL, Gibson GE (2010) Thiamine and oxidants interact to modify cellular calcium stores. Neurochem Res 35:2107–2116
Husain K, Somani SM (1998) Effect of exercise training and chronic ethanol ingestion on cholinesterase activity and lipid peroxidation in blood and brain regions of rat. Prog Neuropsychopharmacol Biol Psychiatry 22:411–423
Iwata H (1982) Possible role of thiamine in the nervous system. Trends Pharmacol Sci 3:171–173
Jamal M, Ameno K, Ameno S, Morishita J, Wang W, Kumihashi M, Ikuo U, Miki T, Ijiri I (2007) Changes in cholinergic function in the frontal cortex and hippocampus of rat exposed to ethanol and acetaldehyde. Neuroscience 144:232–238
Jankowska-Kulawy A, Bielarczyk H, Pawełczyk T, Wróblewska M, Szutowicz A (2010) Acetyl-CoA and acetylcholine metabolism in nerve terminal compartment of thiamine deficient rat brain. J Neurochem 115:333–342
Langlais PJ, Zhang SX (1997) Cortical and subcortical white matter damage without Wernicke’s encephalopathy after recovery from thiamine deficiency in the rat. Alcohol Clin Exp Res 21:434–443
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Manzo L, Locatelli C, Candura SM, Costa LG (1994) Nutrition and alcohol neurotoxicity. Neurotoxicology 15:555–565
Martin PR, Singleton CK, Hiller-Sturmhofel S (2003) The role of thiamine deficiency in alcoholic brain disease. Alcohol Res Health 27:134–142
Matsuda T, Cooper JR (1981) Thiamine as an integral component of brain synaptosomal membranes. Proc Natl Acad Sci U S A 78:5886–5889
Matsuda T, Iwata H (1987) Decrease of high affinity ouabain binding in rat cerebellum and hypothalamus by thiamin deficiency. Brain Res 437:375–378
Matsushima K, MacManus JP, Hakim AM (1997) Apoptosis is restricted to the thalamus in thiamine-deficient rats. Neuroreport 8:867–870
Moloney B, Leonard BE (1984) Pre-natal and post-natal effects of alcohol in the rat–II. Changes in gamma-aminobutyric acid concentration and adenosine triphosphatase activity in the brain. Alcohol Alcohol 19:137–140
Mousseau DD, Rao VL, Butterworth RF (1996) Na+,K+-ATPase activity is selectively increased in thalamus in thiamine deficiency prior to the appearance of neurological symptoms. Eur J Pharmacol 300:191–196
Navarro D, Zwingmann C, Hazell AS, Butterworth RF (2005) Brain lactate synthesis in thiamine deficiency: a re-evaluation using 1H-13C nuclear magnetic resonance spectroscopy. J Neurosci Res 79:33–41
Oner P, Cinar F, Koçak H, Gürdöl F (2002) Effect of exogenous melatonin on ethanol-induced changes in Na+,K+- and Ca2+-ATPase activities in rat synaptosomes. Neurochem Res 27:1619–1623
Pires RG, Pereira SR, Oliveira-Silva IF, Franco GC, Ribeiro AM (2005) Cholinergic parameters and the retrieval of learned and re-learned spatial information: a study using a model of Wernicke-Korsakoff Syndrome. Behav Brain Res 162:11–21
Roland JJ, Savage LM (2009) The role of cholinergic and GABAergic medial septal/diagonal band cell populations in the emergence of diencephalic amnesia. Neuroscience 160:32–41
Ruano MJ, Sánchez-Martín MM, Alonso JM, Hueso P (2000) Changes of acetylcholinesterase activity in brain areas and liver of sucrose- and ethanol-fed rats. Neurochem Res 25:461–470
Schenker S, Henderson GI, Hoyumpa AM Jr, McCandless DW (1980) Hepatic and Wernicke’s encephalopathies: current concepts of pathogenesis. Am J Clin Nutr 33:2719–2726
Sechi G, Serra A (2007) Wernicke’s encephalopathy: new clinical settings and recent advances in diagnosis and management. Lancet Neurol 6:442–455
Syapin PJ, Chen J, Alkana RL (1985) Effect of norepinephrine on inhibition of mouse brain (Na+,K+)-stimulated, (Mg++)-dependent, and (Ca++)-dependent ATPase activities by ethanol. Alcohol 2:145–148
Tallaksen CM, Taubøll E (2000) Excitatory effect of thiamin on CA1 pyramidal neurones in rat hippocampal slices in vitro. Eur J Neurol 7:693–698
Tanphaichitr V (1999) Thiamin. In: Shils ME, Olson JA, Shike M, Ross AC (eds) Modern nutrition in health and disease, 9th edn. Williams & Wilkins, Baltimore, pp 381–389
Thomson AD, Marshall EJ (2006) The natural history and pathophysiology of Wernicke’s Encephalopathy and Korsakoff’s Psychosis. Alcohol Alcohol 41:151–158
Tsakiris S (2001) Effects of L-phenylalanine on acetylcholinesterase and Na+,K+-ATPase activities in adult and aged rat brain. Mech Ageing Dev 122:491–501
Tsakiris S, Angelogianni P, Schulpis KH, Behrakis P (2000) Protective effect of L-cysteine and glutathione on rat brain Na+,K+-ATPase inhibition induced by free radicals. Z Naturforsch [C] 55:271–277
Tsakiris T, Angelogianni P, Tesseromatis C, Tsakiris S, Schulpis KH (2008) Effect of L-carnitine administration on the modulated rat brain protein concentration, acetylcholinesterase, Na+,K+-ATPase and Mg2+-ATPase activities induced by forced swimming. Br J Sports Med 42:367–372
Victor M, Adams RD, Collins GH (1971) The Wernicke-Korsakoff syndrome. A clinical and pathological study of 245 patients, 82 with post-mortem examinations. Contemp Neurol Ser 7:1–206
Victor M, Adams RD, Collins GH (1989) The Wernicke-Korsakoff Syndrome and Related Neurologic Disorders due to Alcohol and Malnutrition, 2nd edn. Davies FA, Philadelphia
Vorhees CV, Schmidt DE, Barrett RJ (1978) Effects of pyrithiamin and oxythiamin on acetylcholine levels and utilization in rat brain. Brain Res Bull 3:493–496
Wernicke C (1881) Lehrbuch der Gehirnkrankheiten fur Aerzte und Studirende. Volume II. Fisher-Verlag, Kassel
Zarros A, Liapi C, Galanopoulou P, Marinou K, Mellios Z, Skandali N, Al-Humadi H, Anifantaki F, Gkrouzman E, Tsakiris S (2009) Effects of adult-onset streptozotocin-induced diabetes on the rat brain antioxidant status and the activities of acetylcholinesterase, (Na+,K+)- and Mg2+-ATPase: modulation by L-cysteine. Metab Brain Dis 24:337–348
Zarros A, Theocharis S, Skandali N, Tsakiris S (2008) Effects of fulminant hepatic encephalopathy on the adult rat brain antioxidant status and the activities of acetylcholinesterase, (Na+,K+)- and Mg2+-ATPase: comparison of the enzymes’ response to in vitro treatment with ammonia. Metab Brain Dis 23:255–264
Acknowledgments
The authors wish to acknowledge their appreciation to Dr Foteini Anifantaki and Mr Konstantinos Zissis for their assistance.
Conflict of interest
No conflicts of interest exist.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zarros, A., Liapi, C., Al-Humadi, H. et al. Experimentally-induced Wernicke’s encephalopathy modifies crucial rat brain parameters: the importance of Na+,K+-ATPase and a potentially neuroprotective role for antioxidant supplementation. Metab Brain Dis 28, 387–396 (2013). https://doi.org/10.1007/s11011-013-9394-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-013-9394-2