Abstract
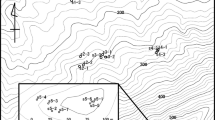
Widespread ecosystem change has led to declines in species world-wide. The loss of pollinators in particular constitutes a problem for ecosystem function and crop production. Understanding how landscape change affects pollinator movement, effective pollen flow, and plant and pollinator survival is therefore a global priority. In this study we investigated patterns of effective pollen flow, using wild cherry tree (Prunus avium) progeny arrays, to address two questions in three case studies: Do land-use types present different resistances to pollinator movement? Which pollinator travel path best explains the pollination data (straight lines, weighted straight lines, least cost paths or pair-wise resistance)? Trees and progeny arrays were genotyped and effective pollen flow and pollinator movement were estimated using the spatially explicit mating model. We found that pollinators did modify their travel paths in response to land-use type and arrangement, but the travel path that best described pollinator movement and the resistance rank of the land uses depended on the type and size of land-use patches and the landscape context. We propose a novel theoretical framework rooted in behavioural ecology, the resource model, for interpreting pollinator behaviour in heterogeneous landscapes. We conclude by discussing the importance and practicality of conservation and management strategies in which native and non-native land-use types together provide functional habitat and support ecosystem services across economic landscapes.

Similar content being viewed by others
References
Allen-Wardell G, Bernhardt P, Bitner R, Burquez A, Buchmann S, Cane J, Cox PA, Dalton V, Feinsinger P, Ingram M, Inouye D, Jones EC, Kennedy K, Kevan P, Koopowitz H, Medellin R, Medellin-Morales S, Nabhan GP, Pavlik B, Tepedino V, Torchio P, Walker S (1998) The potential consequences of pollinator declines on the conservation of biodiversity and stability of food crop yields. Conserv Biol 12(1):8–17
Bell W (1990) Searching behaviour patterns in insects. Annu Rev Entomol 35:447–467
Burnham KP, Anderson DR (1998) Model selection and inference: a practical information-theoretic approach. Springer-Verlag, New York
Carvell C, Jordan WC, Bourke AFG, Pickles R, Redhead JW, Heard MS (2012) Molecular and spatial analyses reveal links between colony-specific foraging distance and landscape-level resource availability in two bumblebee species. Oikos 121(5):734–742
Cipriani G, Lot G, Huang WG, Marrazzo MT, Peterlunger E, Testolin R (1999) AC/GT and AG/CT micosatellite repeats in peach (Prunus persica (L.) Batsch): isolation, characterization and cross-species amplification in Prunus. Theor Appl Genet 99:65–72
Cottrell JE, Vaughan SP, Connolly T, Sing L, Moodley DJ, Russell K (2009) Contemporary pollen flow, characterization of the maternal ecological neighbourhood and mating patterns in wild cherry (Prunus avium L.). Heredity 103:118–128
Cranmer L, McCollin D, Ollerton J (2012) Landscape structure influences pollinator movements and directly affects plant reproductive success. Oikos 121(4):562–568
Dirlewanger E, Capdeville G, Tauzin Y, Cosson P, Claverie J (2008) A Sweet Cherry (Prunus avium L.) linkage map and its comparison to other Prunus species. In: Burak M, Lang GA, Gulen H, Ipek A (eds) Proceedings of the Vth international cherry symposium, vols 1 and 2, Acta Horticulturae. pp 115–126
Doerr VAJ, Barrett T, Doerr ED (2011) Connectivity, dispersal behaviour and conservation under climate change: a response to Hodgson et al. J Appl Ecol 48(1):143–147
Downey S, Iezzoni A (2000) Polymorphic DNA markers in black cherry (Prunus serotina) are identified using sequences from sweet cherry, peach, and sour cherry. J Am Soc Hortic Sci 125:76–80
Dyer RJ, Chan DM, Gardiakos VA, Meadows CA (2012) Pollination graphs: quantifying pollen pool covariance networks and the influence of intervening landscape on genetic connectivity in the North American understory tree, Cornus florida L. Landsc Ecol 27:239–251
Fortuna MA, Garcia C, Guimaraes PR, Bascompte J (2008) Spatial mating networks in insect-pollinated plants. Ecol Lett 11(5):490–498
Guitian P, Guitian J, Navarro L (1993) Pollen transfer and diurnal versus nocturnal pollination in Lonicera etrusca. Acta Oecol Int J Ecol 14(2):219–227
Hagerty BE, Nussear KE, Esque TC, Tracy CR (2011) Making molehills out of mountains: landscape genetics of the Mojave Desert tortoise. Landsc Ecol 26(2):267–280
Hodges C, Miller R (1981) Pollinator flight directionality and the assessment of pollen returns. Oecologia 50:376–379
Jacobs JH, Clark SJ, Denholm I, Goulson D, Stoate C, Osborne JL (2009) Pollination biology of fruit-bearing hedgerow plants and the role of flower-visiting insects in fruit-set. Ann Bot 104(7):1397–1404
Jolivet C, Holtken AM, Liesebach H, Steiner W, Degen B (2012) Mating patterns and pollen dispersal in four contrasting wild cherry populations (Prunus avium L.). Eur J For Res 131:1055–1069
Jones E, Dornhaus A (2011) Predation risk makes bees reject rewarding flowers and reduce foraging activity. Behav Ecol Sociobiol 65:1505–1511
Joobeur T, Periam N, Vicente M, King G, Arus P (2000) Development of a second generation linkage map for almond using RAPD and SSR markers. Genome 43:649–655
Jules ES, Shahani P (2003) A broader ecological context to habitat fragmentation: why matrix habitat is more important than we thought. J Veg Sci 14(3):459–464
Klein EK, Carpentier FH, Oddou-Muratorio S (2011) Estimation of the whole variance of male fecundity from genotypes of progeny arrays: accuracy and limits of the Bayesian forward approach. Methods Ecol Evol 2:349–361
Kremen C, Williams NM, Aizen MA, Gemmill-Herren B, LeBuhn G, Minckley R, Packer L, Potts SG, Roulston T, Steffan-Dewenter I, Vázquez DP, Winfree R, Adams L, Crone EE, Greenleaf SS, Keitt TH, Klein AM, Regetz J, Ricketts TH (2007) Pollination and other ecosystem services produced by mobile organisms: a conceptual framework for the effects of land-use change. Ecol Lett 10(4):299–314
Lander TA, Bebber DP, Choy CTL, Harris SA, Boshier DH (2011) The circe principle explains how resource-rich land can waylay pollinators in fragmented landscapes. Curr Biol 21(15):1302–1307
Lavergne S, Thuiller W, Molina J, Debussche M (2005) Environmental and human factors influencing rare plant local occurrence, extinction and persistence: a 115-year study in the Mediterranean region. J Biogeogr 32:799–811
MacArthur RH, Pianka ER (1966) On the optimal use of a patchy environment. Am Nat 100:603–609
MacArthur RH, Wilson EO (1967) The theory of Island biogeography. Princeton University Press, Princeton
McRae BH, Dickson BG, Keitt TH, Shah VB (2008) Using circuit theory to model connectivity in ecology, evolution and conservation. Ecology 89(10):2712–2724
Meagher T (1986) Analysis of paternity within natural population of Chamaelirium luteum 1. Identification of most-likely male parent. Am Nat 128:199–215
Moilanen A, Hanski I (1998) Metapopulation dynamics: effects of habitat quality and landscape structure. Ecology 79:2503–2515
Ohashi K, Thomson J (2005) Efficient harvesting of renewing resources. Behav Ecol 16:592–605
Potts SG, Biesmeijer JC, Kremen C, Neumann P, Schweiger O, Kunin WE (2010) Global pollinator declines: trends, impacts and drivers. Trends Ecol Evol 25:345–353
Pullinger MG, Johnson CJ (2010) Maintaining or restoring connectivity of modified landscapes: evaluating the least-cost path model with multiple sources of ecological information. Landsc Ecol 25(10):1547–1560
Rands S, Whitney H (2011) Field margins, foraging distances and their impacts on nesting pollinator success. PLoS One 6(10):e25971
Ray N (2005) PATHMATRIX: a geographical information system tool to compute effective distances among samples. Mol Ecol Notes 5(1):177–180
Richard Y, Armstrong DP (2010) Cost distance modelling of landscape connectivity and gap-crossing ability using radio-tracking data. J Appl Ecol 47(3):603–610
Richards S, Williams N, Harder L (2009) Variation in pollination: causes and consequences for plant reproduction. Am Nat 174:382–398
Ricketts TH (2001) The matrix matters: effective isolation in fragmented landscapes. Am Nat 158(1):87–99
Ries L, Fletcher RJ, Battin J, Sisk TD (2004) Ecological responses to habitat edges: mechanisms, models, and variability explained. Annu Rev Ecol Evol Syst 35:491–522
Romero G, Antiqueira P, Koricheva J (2011) A meta-analysis of predation risk effects on pollinator behaviour. PLoS One 6(6):e20689
Russell K (2003) EUFORGEN technical guidelines for genetic conservation and use for wild cherry (Prunus avium). International Plant Genetic Resources Institute, Rome, p 6
Sonneveld T, Tobutt KR, Robbins TP (2003) Allele-specific PCR detection of sweet cherry self-incompatibility (S) alleles S1 to S16 using consensus and allele-specific primers. Theor Appl Genet 107(6):1059–1070
Spear SF, Balkenhol N, Fortin MJ, McRae BH, Scribner K (2010) Use of resistance surfaces for landscape genetic studies: considerations for parameterization and analysis. Mol Ecol 19(17):3576–3591
Stoeckel S, Grange J, Fernandez-Manjarres JF, Bilger I, Frascaria-Lacoste N, Mariette S (2006) Heterozygote excess in a self-incompatible and partially clonal forest tree species—Prunus avium L. Mol Ecol 15(8):2109–2118
Stoeckel S, Klein E, Oddou-Muratorio S, Musch B, Mariette S (2012) Microevolution of S-allele frequencies in wild cherry populations: respective impacts of negative frequency dependent selection and genetic drift. Evolution 66:486–504
Testolin R, Marrazzo T, Cipriani G, Quarta R, Verde I, Dettori MT, Pancaldi M, Sansavini S (2000) Microsatellite DNA in peach (Prunus persica L. Batsch) and its use in fingerprinting and testing the genetic origin of cultivars. Genome 43:512–520
Tischendorf L, Fahrig L (2000) How should we measure landscape connectivity? Landsc Ecol 15(7):633–641
van Etten J (2012) Gdistance: distances and movements on geographical grids. http://r-forge.r-project.org/projects/gdistance
Vandermeer J, Carvajal R (2001) Metapopulation dynamics and the quality of the matrix. Am Nat 158(3):211–220
Vaughan SP, Boskovic RI, Gisbert-Climent A, Russell K, Tobutt KR (2008) Characterisation of novel S-alleles from cherry (Prunus avium L.). Tree Genet Genomes 4(3):531–541
Winker K, Rappole J, Ramos M (1995) The use of movement data as an assay of habitat quality. Oecologia 101:211–216
Wolf S, Moritz RFA (2008) Foraging distance in Bombus terrestris L. (Hymenoptera: Apidae). Apidologie 39:419–427
Zeller KA, McGarigal K, Whiteley AR (2012) Estimating landscape resistance to movement: a review. Landsc Ecol 27(6):777–797
Acknowledgments
We thank Laurent Lévèque and Bénédicte Le Guérroué for field sampling and lab genotyping for the Neuillé population. Isabelle Bilger, Stéphane Matz and the French Office National des Forêts sampled at St-Gobain, and Jérôme Grange and Sandrine Toussaint helped to acquire genetic data for this population. This work was funded by grants to T. L. by INRA (EFPA department) and to S.M. and S.S. from Cemagref, and the Office National des Forêts.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lander, T.A., Klein, E.K., Stoeckel, S. et al. Interpreting realized pollen flow in terms of pollinator travel paths and land-use resistance in heterogeneous landscapes. Landscape Ecol 28, 1769–1783 (2013). https://doi.org/10.1007/s10980-013-9920-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10980-013-9920-y