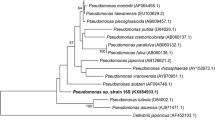
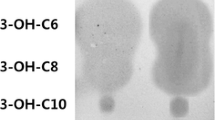
Abstract
In Gram-negative bacteria, the acyl-homoserine lactones (acyl-HSLs) are the main signaling substances employed in cell-to-cell communication systems. This paper describes the chemical characterization of acyl-HSLs produced by the worldwide-spread phytopathogen Pantoea ananatis (Serrano 1928) by using gas chromatography-electron impact mass spectrometry. The absolute configuration of the major identified substance, (S)-(−)-N-hexanoyl-HSL, was determined with gas chromatography-flame ionization detection with a chiral capillary column. Biological activities of extracts, fractions, and synthetic products were evaluated with the specific reporter Agrobacterium tumefaciensNTL4(pZLR4) in β-galactosidase expression assays.


Similar content being viewed by others
References
Azad, H. R., Holmes, G. J., and Cooksey, D. A. 2000. A new leaf blotch disease of sudangrass caused by Pantoea ananas and Pantoea stewartii. Plant Dis. 84:973–979.
Bainton, N. J., Stead, P., Chhabra, S. R., Bycroft, B. W., Salmond, G. P., Stewart, G. S., and Williams, P. 1992. N-(3-Oxohexanoyl)-l-homoserine lactone regulates carbapenem antibiotic production in Erwinia carotovora. Biochem. J. 288:997–1004.
Blosser, R. S. and Gray, K. M. 2000. Extraction of violacein from Chromobacterium violaceum provides a new quantitative bioassay for N-acyl homoserine lactone autoinducers. J. Microbiol. Methods. 40:47–55.
Bodman, S. B. V. and Farrand, S. K. 1995. Capsular polysaccharide biosynthesis and pathogenicity in Erwinia stewartii require induction by an N-acylhomoserine lactone autoinducer. J. Bacteriol. 177:5000–5008.
Cha, C., Gao, P., Chen, Y-C., Shaw, P. D., and Farrand, S. K. 1998. Production of acyl-homoserine lactone quorum-sensing signals by Gram-negative plant-associated bacteria. Mol. Plant–Microbe Interact. 11:1119–1129.
Chhabra, S. R., Stead, P., Bainton, N. J., Salmond, G. P. C., Stewart, G. S. A. B., Williams, P., and Bycroft, B. W. 1993. Autoregulation of carbapenem biosynthesis in Erwinia carotovora by analogs of N-(3-oxohexanoyl)- l-homoserine lactone. J. Antibiot. 46:441–454.
Cother, E. J., Reinke, R., McKenzie, C., Lanoiselet, V. M., and Noble, D. H. 2004. An unusual stem necrosis of rice caused by Pantoea ananas and the first record of this pathogen on rice in Australia. Australas. Plant Pathol. 33:495–503.
Coutinho, T. A., Preisig, O., Mergaert, J., Cnockaert, M. C., Riedel, K.-H., Swings, J., and Wingfield, M. J. 2002. Bacterial blight and dieback of eucalyptus species, hybrids, and clones in South Africa. Plant Dis. 86:20–25.
Eberhard, A., Burlingame, A. L., Eberhard, C., Kenyon, G. L., Nealson, K. H., and Oppenhemer, N. J. 1981. Structural identification of autoinducer of Photobacterium fischeri luciferase. Biochemistry 20:2444–2449.
Engebrecht, J. and Silverman, M. 1984. Identification of genes and gene products necessary for bacterial bioluminescence. Proc. Natl. Acad. Sci. USA 81:4154–4158.
Fuqua, C. and Greenberg, E. P. 1998. Self perception in bacteria: quorum sensing with acylated homoserine lactones. Curr. Opin. Microbiol. 1:183–189.
Hauben, L., Moore, E. R., Vauterin, L., Steenackers, M., Mergaert, J., Verdonck, L., and Swings, J. 1998. Phylogenetic position of phytopathogens within the Enterobacteriaceae. Syst. Appl. Microbiol. 21:384–397.
Horng, Y. T., Deng, S. C., Daykin, M., Soo, P. C., Wei, J. R., Luh, K. T., Ho, S. W., Swift, S., Lai, H. C., and Williams, P. 2002. The LuxR family protein SpnR functions as a negative regulator of N-acylhomoserine lactone-dependent quorum sensing in Serratia marcescens. Mol. Microbiol. 45:1655–1671.
Jones, S., Yu, B., Bainton, N. J., Birdsall, M., Bycroft, B. W., Chhabra, S. R., Cox, A. J. R., Golby, P., Reeves, P. J., Stephens, S., Winson, M. K., Salmond, G. P. C., Stewart, G. S. A. B., and Williams, P. 1993. The lux autoinducer regulates the production of exoenzyme virulence determinants in Erwinia carotovora and Pseudomonas aeruginosa. EMBO J. 12:2477–2482.
Lao, W., Kjelleberg, S., Kumar, N., Denys, R., Read, R. W., and Steinberg, P. 1999. 13C NMR study of N-acyl-S-homoserine lactone derivatives. Magn. Reson. Chem. 37:157–158.
Lithgow, J. K., Wilkinson, A., Hardman, A., Rodelas, B., Wisniewski-Dyé, F., Williams, P., and Downie, J. A. 2000. The regulatory locus cinRI in Rhizobium leguminosarum controls a network of quorum-sensing loci. Mol. Microbiol. 37:81–97.
Marketon, M. M., Gronquist, M. R., Eberhard, A., and González, J. E. 2002. Characterization of the Sinorhizobium meliloti sinR/sinI locus and the production of novel N-acyl homoserine lactones. J. Bacteriol. 184:5686–5695.
Morohoshi, T., Inaba, T., Kato, N., Kanai, K., and Ikeda, T. 2004. Identification of quorum-sensing signal molecules and the LuxRI homologues in the fish pathogen Edwardsiella tarda. J. Biosci. Bioeng. 98:274–281.
Paccola-Meirelles, L. D., Ferreira, A. S., Meirelles, W. F., Marriel, I. E., and Casela, C. R. 2001. Detection of a bacterium associated with a leaf spot disease of maize in Brazil. J. Phytopathol. 149:275–279.
Pearson, J. P., Gray, K. M., Passador, L., Tucker, K. D., Eberhard, A., Iglewski, B. H., and Greenberg, E. P. 1994. Structure of the autoinducer required for expression of Pseudomonas aeruginosa virulence genes. Proc. Natl. Acad. Sci. USA 91:197–201.
Piper, K. R., Bodman, S. B. V., and Farrand, S. K. 1993. Conjugation factor of Agrobacterium tumefaciens regulates Ti plasmid transfer by autoinduction. Nature 362:448–450.
Pomini, A. M., Araújo, W. L., Manfio, G. P., and Marsaioli, A. J. 2005. Acyl-homoserine lactones from Erwinia psidii R. IBSBF 435T, a guava phytopathogen (Psidium guajava L.). J. Agric. Food Chem. 53:6262–6265.
Ravn, L., Christensen, A. B., Molin, S., Givskov, M. and Gram, L. 2001. Methods for detecting acylated homoserine lactones produced by Gram-negative bacteria and their application in studies of AHL-production kinetics. J. Microbiol. Methods 44:239–251.
Roche, D. M., Byers, J. T., Smith, D. S., Glansdorp, F. G., Spring, D. R., and Welch, M. 2004. Communications blackout? Do N-acylhomoserine lactone-degrading enzymes have any role in quorum sensing? Microbiology 150:2023–2028.
Schaefer, A. L., Val, D. L., Hamzelka, B. L., Cronan, J. E., and Greenberg, E. P. 1996. Generation of cell-to-cell signals in quorum-sensing: acyl-homoserine lactone synthase activity of a purified Vibrio fischeri LuxI protein. Proc. Natl. Acad. Sci. USA 93:9505–9509.
Serrano, F. B. 1928. Bacterial fruitlet brown-rot of pineapple in the Philippines. Philipp. J. Sci. 36:271–305.
Suga, H. and Smith, K. M. 2003. Molecular mechanisms of bacterial quorum-sensing as a new drug target. Curr. Opin. Chem. Biol. 7:586–591.
Venturi, V., Venuti, C., Devescovi, G., Lucchese, C., Friscina, A., Degrassi, G., Aguilar, C., and Mazzucchi, U. 2004. The plant pathogen Erwinia amylovora produces acyl-homoserine lactone signal molecules in vitro and in planta. FEMS Microbiol. Lett. 241:179–183.
Walcott, R. R., Gitaitis, R. D., Castro, A. C., Sanders, F. H. Jr., and Diaz-Perez, J. C. 2002. Natural infestation of onion seed by Pantoea ananatis, causal agent of center rot. Plant Dis. 86:106–111.
Whitehead, N. A., Barnard, A. M., Slater, H., Simpson, N. J. L., and Salmond, G. P. C. 2001. Quorum-sensing in Gram-negative bacteria. FEMS Microbiol. Rev. 25:365–404.
Whithers, H., Swift, S., and Williams, P. 2001. Quorum sensing as an integral component of gene regulatory networks in Gram-negative bacteria. Curr. Opin. Microbiol. 4:186–193.
Zhang, L., Murphy, P. J., Kerr, A. and Tate, M. E. 1993. Agrobacterium conjugation and gene regulation by N-acyl- l-homoserine lactones. Nature 362:446–448.
Acknowledgments
The authors are indebted to FAPESP for financial support and for a scholarship awarded to A. M. Pomini (process n. 03/09357-7). We wish to express our gratitude to Carol H. Collins (IQ/Unicamp) for revising the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pomini, A.M., Araújo, W.L. & Marsaioli, A.J. Structural Elucidation and Biological Activity of Acyl-homoserine Lactones from the Phytopathogen Pantoea ananatis Serrano 1928. J Chem Ecol 32, 1769–1778 (2006). https://doi.org/10.1007/s10886-006-9108-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-006-9108-x