Abstract
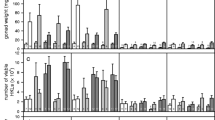
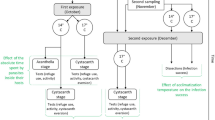
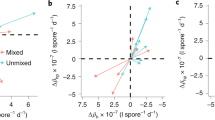
Environmental variation can alter the probability of parasitic infection or the fitness consequence of infection, and thus has the potential to dramatically alter the dynamics of host parasite coevolution. Here we investigated the effect of a changing temperature on host-parasite interactions using the crustacean Daphnia magna and its bacterial parasite Pasteuria ramosa. By reciprocally varying (1) the temperature at which exposure to parasites occurred and (2) the temperature at which within-host parasite growth occurred, and measuring several fitness-related traits, we show that while there are temperature combinations that favour either host or parasite, there are also conditions that favour neither, that is, negative fitness consequences for the host without fitness benefits for the parasite. This result highlights the importance of considering a heterogeneous rather than static environment in coevolutionary studies, while also showing support for an optimal virulence strategy in castrating parasites.


Similar content being viewed by others
References
Anderson RM, May RM (1979) Population biology of infectious diseases: Part I. Nature 280:361–367
Anderson RM, May RM (1982) Coevolution of hosts and parasites. Parasitology 85:411–426
Bell G (1982) The masterpiece of nature: the evolution and genetics of sexuality. University of California Press, Berkeley (CA)
Byers DL (2005) Evolution in heterogeneous environments and the potential of maintenance of genetic variation in traits of adaptive significance. Genetica 123:107–124
Duncan AB, Little TJ (2007) Parasite-driven genetic change in a natural population of Daphnia. Evolution 61:796–803
Ebert D, Weisser WW (1997) Optimal killing for obligate killers: the evolution of life histories and virulence of semelparous parasites. Proc Roy Soc Lond B 264:985–991
Ebert D, Rainey P, Embley T, Scholz D (1996) Development, life cycle, ultrastructure and phylogenetic position of Pasteuria ramosa Metchnikoff 1888: rediscovery of an obligate endoparasite of Daphnia magna Straus. Philos Trans R Soc Lond B 351:1689–1701
Ebert D, Carius HJ, Little T, Decaestecker E (2004) The evolution of virulence when parasites cause host castration and gigantism. Am Nat 164:S19–S32
Fels D, Kaltz O (2006) Temperature-dependent transmission and latency of Holospora undulata, a micronucleus-specific parasite of the ciliate Paramecium caudatum. Proc R Soc Lond B Biol Sci 273:1031–1038
Gillespie JH, Turelli M (1989) Genotype-environment interactions and the maintenance of polygenic variation. Genetics 121:129–138
Hamilton WD (1980) Sex versus non-sex versus parasite. Oikos 35:282–290
Klüttgen B, Dülmer U, Engels M, Ratte HT (1994) ADaM, an artificial freshwater for the culture of zooplankton. Water Res 28:743–746
Laine A-L (2008) Temperature-mediated patterns of local adaptation in a natural plant-pathogen metapopulation. Ecol Lett 11:327–337
Lazzaro BP, Little TJ (2008) Immunity in a variable world. Philos Trans R Soc Lond B 12:15–26
Little TJ (2002) The evolutionary significance of parasitism: do parasite-driven genetic dynamics occur ex silico? J Evol Biol 15:1–9
Little TJ, Chadwick W, Watt K (2008) Parasite variation and the evolution of virulence in a Daphnia-microparasite system. Parasitol 135:303–308
Mackinnon MJ, Read AF (2004) Virulence in malaria: an evolutionary viewpoint. Philos Trans R Soc Lond B Biol Sci 359:965–986
Mitchell SE, Read AF, Little TJ (2004) The effect of a pathogen epidemic on the genetic structure and reproductive strategy of the crustacean Daphnia magna. Ecol Lett 7:848–858
Mitchell SE, Rogers ES, Little TJ, Read AF (2005) Host-parasite and genotype-by-environment interactions: Temperature modifies potential for selection by a sterilizing pathogen. Evolution 59:70–80
O’Keefe KJ, Antonovics J (2002) Playing by different rules: the evolution of virulence in sterilizing pathogens. Am Nat 159:597–605
Peters RH, de Bernardi R (1987) Why Daphnia? In: Peters RH, de Bernardi R (eds) Daphnia. Consiglio nazionale delle ricerche, Istituto italiano di idrobiologia, Verbania Pallanza
SAS (2000) SAS OnlineDoc® , Version 8. SAS Institute Inc
Thomas MB, Blanford S (2003) Thermal biology in insect-parasite interactions. Trends Ecol Evol 18:344–350
Vale PF, Little TJ (2009) Measuring parasite fitness under genetic and thermal variation. Heredity (in press)
Vale PF, Stjernman M, Little TJ (2008) Temperature-dependent costs of parasitism and maintenance of polymorphism under genotype-by-environment interactions. J Evol Biol 21:1418–1427
Acknowledgments
We thank Andrew McKay for laboratory assistance, and Sarah Reece and two anonymous reviewers for manuscript comments. D.E.A. and T.J.L. are funded by a Wellcome Trust Senior Research Fellowship to T.J.L.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Allen, D.E., Little, T.J. Dissecting the effect of a heterogeneous environment on the interaction between host and parasite fitness traits. Evol Ecol 25, 499–508 (2011). https://doi.org/10.1007/s10682-010-9413-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10682-010-9413-2