Abstract
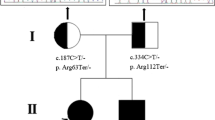
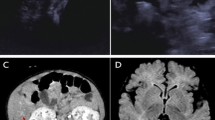
A child of consanguineous parents of Pakistani origin developed jaundice at 5 weeks and then, at 3 months, irritability, a prolonged prothrombin time, a low albumin, and episodes of hypoglycaemia. Investigation showed an elevated alanine aminotransferase with a normal γ-glutamyl-transpeptidase. Analysis of urine by electrospray ionisation tandem mass spectrometry (ESI-MS/MS) showed that the major peaks were m/z 480 (taurine-conjugated 3β-hydroxy-5-cholenoic acid) and m/z 453 (sulphated 3β-hydroxy-5-cholenoic acid). Analysis of plasma by gas chromatography-mass spectrometry (GC-MS) showed increased concentrations of 3β-hydroxy-5-cholenoic acid, 3β-hydroxy-5-cholestenoic acid and 27-hydroxycholesterol, indicating oxysterol 7α-hydroxylase deficiency. The patient was homozygous for a mutation (c.1249C>T) in CYP7B1 that alters a highly conserved residue in oxysterol 7α-hydroxylase (p.R417C) - previously reported in a family with hereditary spastic paraplegia type 5. On treatment with ursodeoxycholic acid (UDCA), his condition was worsening, but on chenodeoxycholic acid (CDCA), 15 mg/kg/d, he improved rapidly. A biopsy (after 2 weeks on CDCA), showed a giant cell hepatitis, an evolving micronodular cirrhosis, and steatosis. The improvement in liver function on CDCA was associated with a drop in the plasma concentrations and urinary excretions of the 3β-hydroxy-Δ5 bile acids which are considered hepatotoxic. At age 5 years (on CDCA, 6 mg/kg/d), he was thriving with normal liver function. Neurological development was normal apart from a tendency to trip. Examination revealed pes cavus but no upper motor neuron signs. The findings in this case suggest that CDCA can reduce the activity of cholesterol 27-hydroxylase - the first step in the acidic pathway for bile acid synthesis.




Similar content being viewed by others
References
Alvelius G, Hjalmarson O, Griffiths WJ et al (2001) Identification of unusual 7-oxygenated bile acid sulfates in a patient with Niemann-Pick disease, type C. J Lipid Res 42:1571–1577
Andersson S, Gustafsson N, Warner M, Gustafsson JA (2005) Inactivation of liver X receptor beta leads to adult-onset motor neuron degeneration in male mice. Proc Natl Acad Sci U S A 102:3857–3862
Axelson M, Mörk B, Aly A et al (1989) Concentrations of cholestenoic acids in plasma from patients with liver disease. J Lipid Res 30:1877–1882
Björkhem I, Eggertsen G (2001) Genes involved in initial steps of bile acid synthesis. Curr Opin Lipidol 12:97–103
Cao G, Liang Y, Broderick CL et al (2003) Antidiabetic action of a liver x receptor agonist mediated by inhibition of hepatic gluconeogenesis. J Biol Chem 278:1131–1136
Clayton PT (1983) The validation and application of an assay for the measurement of bile acids in the plasma of infants and children. MD Thesis. University of Cambridge
Clayton PT (2011) Disorders of bile acid synthesis. J Inherit Metab Dis 34:593–604
Clayton PT, Muller DP (1980) A simplified gas–liquid chromatographic method for the estimation of non-sulphated plasma bile acids. Clin Chim Acta 105:401–405
Clayton PT, Lake BD, Hall NA et al (1987) Plasma bile acids in patients with peroxisomal dysfunction syndromes; analysis by capillary gas chromatography – mass spectrometry. Eur J Pediatr 146:166–173
Clayton PT, Mills KA, Johnson AW et al (1996) Delta 4-3-oxosteroid 5 beta-reductase deficiency: failure of ursodeoxycholic acid treatment and response to chenodeoxycholic acid plus cholic acid. Gut 38:623–628
Cui YL, Zhang JL, Zheng QC et al (2013) Structural and dynamic basis of human cytochrome P450 7B1: a survey of substrate selectivity and major active site access channels. Chemistry 19:549–557
Davies JP, Winchester BG, Malcolm S (1993) Sequence variations in the first exon of alpha-galactosidase A. J Med Genet 30:658–663
Fink J (2006) Hereditary spastic paraplegia. Curr Neurol Neurosci Rep 6:65–76
Gao M, Liu D (2013) The liver X receptor agonist T0901317 protects mice from high fat diet-induced obesity and insulin resistance. AAPS J 15:258–266
Goizet C, Boukhris A, Durr A et al (2009) CYP7B1 mutations in pure and complex forms of hereditary spastic paraplegia type 5. Brain 132:1589–1600
Goodwin B, Watson MA, Kim H et al (2003) Differential regulation of rat and human CYP7A1 by the nuclear oxysterol receptor Liver X receptor-α. Mol Endocrinol 17:386–394
Griffiths WJ, Crick PJ, Wang Y et al (2013) Analytical strategies for characterization of oxysterol lipidomes: liver X receptor ligands in plasma. Free Radic Biol Med 59:69–84
Koopman BJ, Wolthers BG, van der Molen JC et al (1984) Capillary gas chromatographic determinations of urinary bile acids and bile alcohols in CTX patients proving the ineffectivity of ursodeoxycholic acid treatment. Clin Chim Acta 142:103–111
Lewis DF (2004) 57 varieties: the human cytochromes P450. Pharmacogenomics 5:305–318
Li-Hawkins J, Lund EG, Turley SD, Russell DW (2000) Disruption of the oxysterol 7alpha-hydroxylase gene in mice. J Biol Chem 275:16536–16542
Mathis U, Karlaganis G, Preisig R (1983) Monohydroxy bile salt sulfates: tauro-3 beta-hydroxy-5-cholenoate-3-sulfate induces intrahepatic cholestasis in rats. Gastroenterology 85:674–681
Meaney S, Heverin M, Panzenboeck U et al (2007) Novel route for the elimination of brain oxysterols across the blood–brain barrier: conversion into 7α-hydroxy-3-oxo-4-cholestenoic acid. J Lipid Res 48:944–951
Miller SA, Dykes DD, Polesky HF (1988) A simple salting out procedure for extracting DNA from human nucleated cells. Nucleic Acids Res 16:1215
Mills KA, Mushtaq I, Johnson AW et al (1998) A method for the quantitation of conjugated bile acids in dried blood spots using electrospray ionization-mass spectrometry. Pediatr Res 43:361–368
Mizuochi T, Kimura A, Suzuki M et al (2011) Successful heterozygous living donor liver transplantation for an oxysterol 7α-hydroxylase deficiency in a Japanese patient. Liver Transpl 17:1059–1065
Nishimaki-Mogami T, Une M, Fujino T et al (2004) Identification of intermediates in the bile acid synthetic pathway as ligands for the farnesoid X receptor. J Lipid Res 45:1538–1545
Paumgartner G, Beuers U (2004) Mechanisms of action and therapeutic efficacy of ursodeoxycholic acid in cholestatic liver disease. Clin Liver Dis 8:67–81
Pikuleva IA, Waterman MR (2013) Cytochromes p450: roles in diseases. J Biol Chem 288:17091–17098
Segev H, Honigman A, Rosen H, Leitersdorf E (2001) Transcriptional regulation of the human sterol 27-hydroxylase gene (CYP27) and promoter mapping. Atherosclerosis 156:339–347
Setchell KD, Schwarz M, O'Connell NC et al (1998) Identification of a new inborn error in bile acid synthesis: mutation of the oxysterol 7alpha-hydroxylase gene causes severe neonatal liver disease. J Clin Invest 102:1690–1703
Siam A, Brancale A, Simons C (2012) Comparative modeling of 25-hydroxycholesterol-7α-hydroxylase (CYP7B1): ligand binding and analysis of hereditary spastic paraplegia type 5 CYP7B1 mutations. J Mol Model 18:441–453
Song W, Chen J, Dean WL et al (1998) Purification and characterization of hamster liver microsomal 7alpha-hydroxycholesterol dehydrogenase. Similarity to type I 11beta-hydroxysteroid dehydrogenase. J Biol Chem 273:16223–16228
Stiles A, McDonald J, Bauman D, Russell D (2009) CYP7B1: one cytochrome P450, two human genetic diseases, and multiple physiological functions. J Biol Chem 284:28485–28489
Tsaousidou M, Ouahchi K, Warner TT et al (2008) Sequence alterations within CYP7B1 implicate defective cholesterol homeostasis in motor-neuron degeneration. Am J Hum Genet 82:510–515
Twisk J, de Wit EC, Princen HM (1995) Suppression of sterol 27-hydroxylase mRNA and transcriptional activity by bile acids in cultured rat hepatocytes. Biochem J 305:505–511
Ueki I, Kimura A, Nishiyori A et al (2008) Neonatal cholestatic liver disease in an Asian patient with a homozygous mutation in the oxysterol 7alpha-hydroxylase gene. J Pediatr Gastroenterol Nutr 46:465–469
Vlahcevic ZR, Jairath SK, Heuman DM et al (1996) Transcriptional regulation of hepatic sterol 27-hydroxylase by bile acids. Am J Physiol 270:G646–G652
Acknowledgments
The authors thank the family members for their participation. PTC and PBM were funded by Great Ormond Street Children’s Charity. PG is funded by a Wellcome Trust Senior Fellowship. Work in Swansea was supported by BBSRC. Work in Bordeaux was supported by the Agence Nationale de la Recherche (ANR) (Project 2010BLAN112601/LIGENAX), the Association Française contre les Myopathies (AFM) (14879/MNM2 2012), the Conseil Régional d’Aquitaine (CRA) (2011-0151/LIGENAX), The Association Strumpell-Lorrain (ASL) (2011–0135), and the Pôle de compétitivité Prod’Innov.
Competing interest
Dongling Dai, Philippa Mills, Emma Footitt, Paul Gissen, Patricia McClean, Jens Stahlschmidt, Isabelle Coupry, Julie Lavie, Fanny Mochel, Cyril Goizet, Tatsuki Mizuochi, Akihiko Kimura, Hiroshi Nittono, Karin Schwarz, Peter Crick, Yuqin Wang, William Griffiths and Peter Clayton declare that they are not paid personally, or for research, by any company involved in the manufacture, distribution or marketing of CDCA for medicinal use, nor do they hold shares in any such company. Cyril Goizet makes the following declaration: The work under consideration was financially supported by Agence Nationale de la Recherche (ANR) (Project 2010BLAN112601/LIGENAX), the Association Française contre les Myopathies (AFM) (14879/MNM2 2012), the Conseil Régional d’Aquitaine (CRA) (2011-0151/LIGENAX), The Association Strumpell-Lorrain (ASL) (2011–0135), and the Pôle de compétitivité Prod’Innov. This support was addressed to Pr Cyril Goizet. Pr Cyril Goizet received consulting fees from Raptor, Genzyme, Actelion and Shire. Pr Cyril Goizet received financial support for research activities from TKT5S, Shire, Genzyme, Association Française contre les myopathies (AFM), Association contre les maladies mitochondriales (AMMi), Association Strumpell-Lorrain (ASL), Connaitre les Syndromes Cérébelleux (CSC), Conseil régional d’Aquitaine (CRA), Agence Nationale pour la Recherche (ANR), Programme Hospitalier de Recherche Clinique (PHRC), and Registry of the European Huntington Disease Network (EHDN). Inscriptions and travels for congresses of Dr Cyril Goizet were funded by Shire, Genzyme, Actelion, Takeda. William Griiffiths, Peter Crick and Yuqun Wang declare that the development of the analytical methods used in Swansea for the analysis of cholesterol metabolites was funded by a grant from the UIK Research Council, BBSRC (grant no BB/I001735/1). Peter Clayton receives a salary from Great Ormond Street Hospital Children’s Charity and declares the following relevant financial activities outside the submitted work: Grant from Actelion for investigator led project on diagnosis and monitoring of Niemann-Pick C, fees for teaching on courses from Orphan Europe/Recordati Foundation for Rare Diseases, fees for lectures / consultancy from Merck Corp USA, Actelion, shares in Waters, Abbott, Abbvie.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by: K. Michael Gibson
Nomenclature and abbreviations
Some of the bile acids and oxysterols in this paper are referred to by the names in common use in the medical literature rather than the IUPAC recommended names. Thus 27-hydroxycholesterol is cholest-(25R)-5-ene-3β,26-diol / (25R)26-hydroxycholesterol; 7α,27-dihydroxycholesterol is cholest-(25R)-5-ene 3β,7α,26-triol; 7α-hydroxycholesterol is cholest-5-ene-3β,7α-diol; 3β-hydroxy-5-cholestenoic acid is 3β-hydroxycholest-5-en-26-oic acid; 3β-hydroxy-5-cholenoic acid is 3β-hydroxychol-5-en-24-oic acid. Chenodeoxycholic acid (CDCA) is 3α,7α-dihydroxy-5β-cholan-24-oic acid; ursodeoxycholic acid (UDCA) is 3α,7β-dihydroxy-5β-cholan-24-oic acid; cholic acid is 3α,7α,12α-trihydroxy-5β-cholan-24-oic acid.
Rights and permissions
About this article
Cite this article
Dai, D., Mills, P.B., Footitt, E. et al. Liver disease in infancy caused by oxysterol 7α-hydroxylase deficiency: successful treatment with chenodeoxycholic acid. J Inherit Metab Dis 37, 851–861 (2014). https://doi.org/10.1007/s10545-014-9695-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10545-014-9695-6