Summary
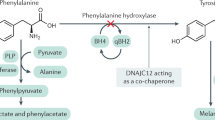
In phenylketonuria, mental retardation is prevented by a diet that severely restricts natural protein and is supplemented with a phenylalanine-free amino acid mixture. The result is an almost normal outcome, although some neuropsychological disturbances remain. The pathology underlying cognitive dysfunction in phenylketonuria is unknown, although it is clear that the high plasma concentrations of phenylalanine influence the blood–brain barrier transport of large neutral amino acids. The high plasma phenylalanine concentrations increase phenylalanine entry into brain and restrict the entry of other large neutral amino acids. In the literature, emphasis has been on high brain phenylalanine as the pathological substrate that causes mental retardation. Phenylalanine was found to interfere with different cerebral enzyme systems. However, apart from the neurotoxicity of phenylalanine, a deficiency of the other large neutral amino acids in brain may also be an important factor affecting cognitive function in phenylketonuria. Cerebral protein synthesis was found to be disturbed in a mouse model of phenylketonuria and could be caused by shortage of large neutral amino acids instead of high levels of phenylalanine. Therefore, in this review we emphasize the possibility of a different idea about the pathogenesis of mental dysfunction in phenylketonuria patients and the aim of treatment strategies. The aim of treatment in phenylketonuria might be to normalize cerebral concentrations of all large neutral amino acids rather than prevent high cerebral phenylalanine concentrations alone. In-depth studies are necessary to investigate the role of large neutral amino acid deficiencies in brain.

Similar content being viewed by others
Abbreviations
- BBB:
-
blood–brain barrier
- LNAA:
-
large neutral amino acids
- MRS:
-
magnetic resonance spectroscopy
- PAH:
-
phenylalanine hydroxylase
- PKU:
-
phenylketonuria
References
Anderson PJ, Wood SJ, Francis DE, Coleman L, Anderson V, Boneh A (2007) Are neuropsychological impairments in children with earl-treated phenylketonuria (PKU) related to white matter abnormalities or elevated phenylalanine levels? Dev Neuropsychol 32: 645–668.
Antoshechkin AG, Chentsova TV, Tatur VY, Naritsin DB, Railian GP (1991) Content of phenylalanine, tyrosine and their metabolites in CSF in phenylketonuria. J Inherit Metab Dis 14: 749–754. doi:10.1007/BF01799945.
Bauman ML, Kemper TL (1982) Morphologic and histoanatomic observations of the brain in untreated human phenylketonuria. Acta Neuropathol 58: 55–63. doi:10.1007/BF00692698.
Bonafé L, Blau N, Burlina AP, et al (2001) Treatable neurotransmitter deficiency in mild phenylketonuria. Neurology 57: 908–911.
Burlina AB, Bonafé L, Ferrari V, et al (2000) Measurement of neurotransmitter metabolites in the cerebrospinal fluid of phenylketonuric patients under dietary treatment. J Inherit Metab Dis 2000; 23: 313–316. doi:10.1023/A:1005694122277.
Butler IJ, O’Flynn ME, Seifert WE, Jr, Howell RR (1981) Neurotransmitter defects and treatment of disorders of hyperphenylalaninemia. J Pediatr 98: 729–733.
Cabib S, Pascucci T, Ventura R, Romano V, Puglisi-Allegra S (2003) The behavioral profile of severe mental retardation in a genetic mouse model of phenylketonuria. Behav Genet 33: 301–310. doi:10.1023/A:1023498508987.
Channon S, German E, Cassina C, Lee P (2004) Executive functioning, memory, and learning in phenylketonuria. Neuropsychology 18: 613–620. doi:10.1037/0894-4105.18.4.613.
Channon S, Mockler C, Lee P (2005) Executive functioning and speed of processing in phenylketonuria. Neuropsychology 19: 679–686. doi:10.1037/0894-4105.19.5.679.
Channon S, Goodman G, Zlotowitz S, Mockler C, Lee PJ (2007) Effects of dietary management of phenylketonuria on long-term cognitive outcome. Arch Dis Child 92: 213–218. doi:10.1136/adc.2006.104786.
Diamond A, Ciaramitaro V, Donner E, Djali S, Robinson MB (1994) An animal model of early-treated PKU. J Neurosci 14: 3072–3082.
Dyer CA, Kendler A, Philibotte T, et al (1996) Evidence for central nervous system glial cell plasticity in phenylketonuria. J Neuropathol Exp Neurol 55: 795–814. doi:10.1097/00005072-199607000-00005.
Fernstrom JD, Fernstrom MH (2007) Tyrosine, phenylalanine, and catecholamine synthesis and function in the brain. J Nutr 137 (Supplement 1): 1539S–1547S.
Glushakov AV, Glushakova O, Varshney M et al (2005) Long-term changes in glutamatergic synaptic transmission in phenylketonuria. Brain 128(2): 300–307. doi:10.1093/brain/awh354.
Guttler F, Lou H (1986) Dietary problems of phenylketonuria: effect on CNS transmitters and their possible role in behaviour and neuropsychological function. J Inherit Metab Dis 9 (Supplement 2): 169–177. doi:10.1007/BF01799701.
Horster F, Schwab MA, Sauer SW, et al (2006) Phenylalanine reduces synaptic density in mixed cortical cultures from mice. Pediatr Res 59: 544–548. doi:10.1203/01.pdr.0000203091.45988.8d.
Hughes JV, Johnson TC (1976) The effects of phenylalanine on amino acid metabolism and protein synthesis in brain cells in vitro. J Neurochem 26: 1105–1113. doi:10.1111/j.1471-4159.1976.tb06993.x.
Huijbregts SCJ, de Sonneville LMJ, van Spronsen FJ, Licht R, Sergeant JA (2002) The neuropsychological profile of early and continuously treated phenylketonuria: orienting, vigilance, and maintenance versus manipulation-functions of working memory. Neurosci Biobehav Rev 26: 697–712. doi:10.1016/S0149-7634(02)00040-4.
Huijbregts SCJ, de Sonneville LMJ, van Spronsen FJ, et al (2003) Motor function under lower and higher controlled processing demands in early and continuously treated phenylketonuria. Neuropsychology 17: 369–379. doi:10.1037/0894-4105.17.3.369.
Ikeda M, Levitt M, Udenfriend S (1965) Hydroxylation of phenylalanine by purified preparations of adrenal and brain tyrosine hydroxylase. Biochem Biophys Res Commun 18: 482–488. doi:10.1016/0006-291X(65)90778-3.
Iversen L (2006) Neurotransmitter transporters and their impact on the development of psychopharmacology. Br J Pharmacol 147 (Supplement 1): S82–S88. doi:10.1038/sj.bjp.0706428.
Johnston MV (2003) Brain plasticity in paediatric neurology. Eur J Paediatr Neurol 7: 105–113. doi:10.1016/S1090-3798(03)00039-4.
Joseph B, Dyer CA (2003) Relationship between myelin production and dopamine synthesis in the PKU mouse brain. J Neurochem 86: 615–626. doi:10.1046/j.1471-4159.2003.01887.x.
Leuzzi V, Pansini M, Sechi E, et al (2004) Executive function impairment in early-treated PKU subjects with normal mental development. J Inherit Metab Dis 27: 115–125. doi:10.1023/B:BOLI.0000028781.94251.1f.
Lykkelund C, Nielsen JB, Lou HC, et al (1988) Increased neurotransmitter biosynthesis in phenylketonuria induced by phenylalanine restriction or by supplementation of unrestricted diet with large amounts of tyrosine. Eur J Pediatr 148: 238–245. doi:10.1007/BF00441411.
Martynyuk AE, Glushakov AV, Sumners C, et al (2005) Impaired glutamatergic synaptic transmission in the PKU brain. Mol Gen Metab 86(Supplement 1): S34–S42. doi:10.1016/j.ymgme.2005.06.014.
McKean CM (1972) The effects of high phenylalanine concentrations on serotonin and catecholamine metabolism in the human brain. Brain Res 47: 469–476. doi:10.1016/0006-8993(72)90653-1.
Möller HE, Weglage J, Wiedermann D, Ullrich K (1998) Blood–brain barrier phenylalanine transport and individual vulnerability in phenylketonuria. J Cereb Blood Flow Metab 18: 1184–1191. doi:10.1097/00004647-199811000-00004.
Nishihira T, Takagi T, Mori S (1993) Amino acid imbalance and intracellular protein synthesis. Nutrition 9: 37–42.
Ogawa S, Ichinose H (2006) Effect of metals and phenylalanine on the activity of human tryptophan hydroxylase-2: comparison with that on tyrosine hydroxylase activity. Neurosci Lett 401: 261–265. doi:10.1016/j.neulet.2006.03.031.
Pardridge WM (1998) Blood–brain barrier carrier-mediated transport and brain metabolism of amino acids. Neurochem Res 23: 635–644. doi:10.1023/A:1022482604276.
Pascucci T, Ventura R, Puglisi-Allegra S, Cabib S (2002) Deficits in brain serotonin synthesis in a genetic mouse model of phenylketonuria. Neuroreport 13: 2561–2564. doi:10.1097/00001756-200212200-00036.
Pietz J, Fatkenheuer B, Burgard P, et al (1997) Psychiatric disorders in adult patients with early-treated phenylketonuria. Pediatrics 99: 345–350. doi:10.1542/peds.99.3.345.
Pietz J, Kreis R, Rupp A, et al (1999) Large neutral amino acids block phenylalanine transport into brain tissue in patients with phenylketonuria. J Clin Invest 103: 1169–1178. doi:10.1172/JCI5017.
Pietz J, Rupp A, Ebinger F, et al (2003) Cerebral energy metabolism in phenylketonuria: findings by quantitative in vivo 31P MR spectroscopy. Pediatr Res 53: 654–662. doi:10.1203/01.PDR.0000055867.83310.9E.
Pratt OEA (1980) New approach to the treatment of phenylketonuria. J Ment Defic Res 24: 203–217.
Puglisi-Allegra S, Cabib S, Pascucci T, et al (2000) Dramatic brain aminergic deficit in a genetic mouse model of phenylketonuria. Neuroreport 11: 1361–1364. doi:10.1097/00001756-200004270-00042.
Ramakers GJ (2002) Rho proteins, mental retardation and the cellular basis of cognition. Trends Neurosci 25: 191–199. doi:10.1016/S0166-2236(00)02118-4.
Schindeler S, Ghosh-Jerath S, Thompson S, et al (2007) The effects of large neutral amino acid supplements in PKU: an MRS and neuropsychological study. Mol Genet Metab 91: 48–54. doi:10.1016/j.ymgme.2007.02.002.
Schmidt E, Burgard P, Rupp A (1996) Effects of concurrent phenylalanine levels on sustained attention and calculation speed in patients treated early for phenylketonuria. Eur J Pediatr 155(Supplement 1): S82–S86. doi:10.1007/PL00014258.
Scriver CR (2001) Hyperphenylalaninemia: phenylalanine hydroxylase deficiency. In: Scriver CR, Beaudet AL, SlyWS, Valle D, eds; Childs B, Kinzler KW, Vogelstein B, assoc. eds. The Metabolic and Molecular Bases of InheritedDisease, 8th edn. New York: McGraw-Hill, 1667–1724.
Scriver CR, Waters PJ (1999) Monogenic traits are not simple: lessons from phenylketonuria. Trends Genet 15: 267–272. doi:10.1016/S0168-9525(99)01761-8.
Shefer S, Tint GS, Jean-Guillaume D, et al (2000) Is there a relationship between 3-hydroxy-3-methylglutaryl coenzyme a reductase activity and forebrain pathology in the PKU mouse? J Neurosci Res 61: 549–563. doi:10.1002/1097-4547(20000901)61:5<549::AID-JNR10>3.0.CO;2-0.
Shiman R, Akino M, Kaufman S (1971) Solubilization and partial purification of tyrosine hydroxylase from bovine adrenal medulla. J Biol Chem 246: 1330–1340.
Smith CB, Kang J (2000) Cerebral protein synthesis in a genetic mouse model of phenylketonuria. Proc Natl Acad Sci U S A 97: 11014–11019. doi:10.1073/pnas.97.20.11014.
Smith I, Knowles J (2000) Behaviour in early treated phenylketonuria: a systematic review. Eur J Pediatr 159(Supplement 2): S89–S93. doi:10.1007/PL00014392.
Smith QR, Momma S, Aoyagi M, Rapoport SI (1987) Kinetics of neutral amino acid transport across the blood–brain barrier. J Neurochem 49: 1651–1658. doi:10.1111/j.1471-4159.1987.tb01039.x.
Stemerdink BA, Kalverboer AF, van der Meere JJ, et al (2000) Behaviour and school achievement in patients with early and continuously treated phenylketonuria. J Inherit Metab Dis 23: 548–562. doi:10.1023/A:1005669610722.
Surtees R, Blau N (2000) The neurochemistry of phenylketonuria. Eur J Pediatr 159 (Supplement 2): S109–S113. doi:10.1007/PL00014370.
Ullrich K, Weglage J, Oberwittler C, et al (1994) Effect of l-dopa on pattern visual evoked potentials (P-100) and neuropsychological tests in untreated adult patients with phenylketonuria. J Inherit Metab Dis 17: 349–352. doi:10.1007/BF00711827.
van Spronsen FJ, van RM, Bekhof J, Koch R, Smit PG (2001) Phenylketonuria: tyrosine supplementation in phenylalanine-restricted diets. Am J Clin Nutr 73: 153–157.
Weglage J, Pietsch M, Funders B, Koch HG, Ullrich K (1996) Deficits in selective and sustained attention processes in early treated children with phenylketonuria—result of impaired frontal lobe functions? Eur J Pediatr 155: 200–204. doi:10.1007/BF01953938.
Weglage J, Wiedermann D, Moller H, Ullrich K (1998) Pathogenesis of different clinical outcomes in spite of identical genotypes and comparable blood phenylalanine concentrations in phenylketonurics. J Inherit Metab Dis 21: 181–182. doi:10.1023/A:1005328717095.
Weglage J, Wiedermann D, Denecke J, et al (2001) Individual blood–brain barrier phenylalanine transport determines clinical outcome in phenylketonuria. Ann Neurol 50: 463–467. doi:10.1002/ana.1226.
Welsh MC, Pennington BF, Ozonoff S, Rouse B, McCabe ER (1990) Neuropsychology of early-treated phenylketonuria: specific executive function deficits. Child Dev 61: 1697–1713. doi:10.2307/1130832.
White DA, Nortz MJ, Mandernach T, Huntington K, Steiner RD (2001) Deficits in memory strategy use related to prefrontal dysfunction during early development: evidence from children with phenylketonuria. Neuropsychology 15: 221–229. doi:10.1037/0894-4105.15.2.221.
White DA, Nortz MJ, Mandernach T, Huntington K, Steiner RD (2002) Age-related working memory impairments in children with prefrontal dysfunction associated with phenylketonuria. J Int Neuropsychol Soc 8(1): 1–11. doi:10.1017/S1355617702811018.
Zagreda L, Goodman J, Druin DP, McDonald D, Diamond A (1999) Cognitive deficits in a genetic mouse model of the most common biochemical cause of human mental retardation. J Neurosci 19: 6175–6182.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicating editor: Beat Thöny
Competing interests: None declared
References to electronic databases: Phenylketonuria: OMIM 261600.
Rights and permissions
About this article
Cite this article
van Spronsen, F.J., Hoeksma, M. & Reijngoud, DJ. Brain dysfunction in phenylketonuria: Is phenylalanine toxicity the only possible cause?. J Inherit Metab Dis 32, 46–51 (2009). https://doi.org/10.1007/s10545-008-0946-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10545-008-0946-2