Summary
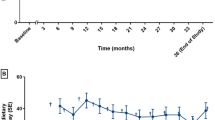
This study aimed to evaluate the response to and safety of an 8-day course of sapropterin dihydrochloride (6R-tetrahydrobiopterin or 6R-BH4) 10 mg/kg per day in patients with phenylketonuria (PKU), who have elevated blood phenylalanine (Phe) levels, and to identify a suitable cohort of patients who would respond to sapropterin dihydrochloride treatment with a reduction in blood Phe level. Eligible patients were aged ≥ 8 years, had blood Phe levels ≥ 450 μmol/L and were not adhering to a Phe-restricted diet. Suitable patients were identified by a ≥30% reduction in blood Phe level from baseline to day 8 following sapropterin dihydrochloride treatment. The proportion of patients who met these criteria was calculated for the overall population and by baseline Phe level (<600, 600 to <900, 900 to <1200 and ≥1200 μmol/L). In total, 485/490 patients completed the study and 20% (96/485) were identified as patients who would respond to sapropterin dihydrochloride. A reduction in Phe level was observed in all subgroups, although response was greater in patients with lower baseline Phe levels. Wide variability in response was seen across all baseline Phe subgroups. The majority of adverse events were mild and all resolved without complications. Sapropterin dihydrochloride was well tolerated and reduced blood Phe levels across all PKU phenotypes tested. Variability in reduction of Phe indicates that the response to sapropterin dihydrochloride cannot be predicted by baseline Phe level.
Similar content being viewed by others
References
Bernegger C, Blau N (2002) High frequency of tetrahydrobiopterin-responsiveness among hyperphenylalaninemias: a study of 1,919 patients observed from 1988 to 2002. Mol Genet Metab 77: 304–313.
Blau N, Erlandsen H (2004) The metabolic and molecular bases of tetrahydrobiopterin-responsive phenylalanine hydroxylase deficiency. Mol Genet Metab 82: 101–11.
Cerone R, Schiaffino MC, Fantasia AR, Perfumo M, Birk Moller L, Blau N (2004) Long-term follow-up of a patient with mild tetrahydrobiopterin-responsive phenylketonuria. Mol Genet Metab 81: 137–139.
Fiege B, Bonafe L, Ballhausen D, et al (2005) Extended tetrahydrobiopterin loading test in the diagnosis of cofactor-responsive phenylketonuria: a pilot study. Mol Genet Metab 86(Supplement 1): S91–S95.
Grier RE, Gahl WA, Cowan T, Bernardini I, McDowell GA, Rinaldo P (2004) Revised sections F7.5 (quantitative amino acid analysis) and F7.6 (qualitative amino acid analysis): American College of Medical Genetics Standards and Guidelines for Clinical Genetics Laboratories, 2003. Genet Med 6: 66–68.
Hennermann JB, Buhrer C, Blau N, Vetter B, Monch E (2005) Long-term treatment with tetrahydrobiopterin increases phenylalanine tolerance in children with severe phenotype of phenylketonuria. Mol Genet Metab 86(Supplement 1): S86–S90.
Holtzman NA, Kronmal RA, van Doorninck W, Azen C, Koch R (1986) Effect of age at loss of dietary control on intellectual performance and behavior of children with phenylketonuria. N Engl J Med 314: 593–598.
Kaufman S (1986) Unsolved problems in diagnosis and therapy of hyperphenylalaninemia caused by defects in tetrahydrobiopterin metabolism. J Pediatr 109: 572–578.
Kure S, Hou DC, Ohura T, et al (1999) Tetrahydrobiopterin-responsive phenylalanine hydroxylase deficiency. J Pediatr 135: 375–78.
Levy H, Milanowski A, Chakrapani A, et al (2006) A phase 3 study of the efficacy of sapropterin dihydrochloride (tetrahydrobiopterin, 6R-BH4) in reducing Phe levels in subjects with phenylketonuria. 56th Annual Meeting of the American Society of Human Genetics (ASHG), New Orleans, LA, USA. [Abstract].
Levy H, Milanowski A, Chakrapani A, et al (2007) Sapropterin dihydrochloride (tetrahydrobiopterin, 6R-BH4) is well tolerated and reduces blood phenylalanine levels in BH4-responsive phenylketonuric patients: results of a Phase 3, double-blind, placebo-controlled trial. American College of Medical Genetics (ACMG), Nashville, TN, USA. [Abstract].
Lindner M, Haas D, Mayatepek E, Zschocke J, Burgard P (2001) Tetrahydrobiopterin responsiveness in phenylketonuria differs between patients with the same genotype. Mol Genet Metab 73: 104–106.
Matalon R, Michals-Matalon K, Koch R, Grady J, Tyring S, Stevens RC (2005) Response of patients with phenylketonuria in the US to tetrahydrobiopterin. Mol Genet Metab 86(Supplement 1): S17–S21.
Michals K, Azen C, Acosta P, Koch R, Matalon R (1988) Blood phenylalanine levels and intelligence of 10-year-old children with PKU in the National Collaborative Study. J Am Diet Assoc 88: 1226–1229.
Muntau AC, Roschinger W, Habich M, et al (2002) Tetrahydrobiopterin as an alternative treatment for mild phenylketonuria. N Engl J Med 347: 2122–2132.
National Institutes of Health Consensus Development Conference Statement (2001) Phenylketonuria: screening and management, October 16–18, 2000. Pediatrics 108: 972–982.
Pietz J (1998) Neurological aspects of adult phenylketonuria. Curr Opin Neurol 11: 679–688.
Scriver CR, Kaufman S (2001) Hyperphenylalaninemia: phenylalanine hydroxylase deficiency. In: Scriver CR, Beaudet AL, Sly WS, Valle D, eds; Childs B, Kinzler KW, Vogelstein B, assoc. eds. The Metabolic and Molecular Bases of Inherited Disease, 8th edn. New York: McGraw-Hill, 1667–1709.
Shintaku H, Kure S, Ohura T, Kano Y, Ohwada M, Sugiyama N (2003) Diagnosis and long-term treatment of tetrahydrobiopterin-responsive hyperphenylalaninaemia with a mutant phenylalanine hydroxylase gene. J Inherit Metab Dis 26(Supplement 2): 16–23.
Walter JH, White FJ, Hall SK, et al (2002) How practical are recommendations for dietary control in phenylketonuria? Lancet 360: 55–57.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicating editor: Nenad Blau
Competing interests: None declared
References to electronic databases: Phenylketonuria (PKU): OMIM 261600.
Rights and permissions
About this article
Cite this article
Burton, B.K., Grange, D.K., Milanowski, A. et al. The response of patients with phenylketonuria and elevated serum phenylalanine to treatment with oral sapropterin dihydrochloride (6R-tetrahydrobiopterin): a phase II, multicentre, open-label, screening study. J Inherit Metab Dis 30, 700–707 (2007). https://doi.org/10.1007/s10545-007-0605-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10545-007-0605-z