Abstract
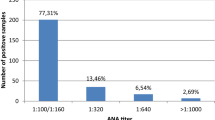
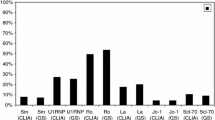
The aim of this study was to investigate the types and prevalence of disease-specific antinuclear antibodies (ANAs) and their relationship to rheumatic diseases in the general Japanese population. An immunofluorescence (IF) method was used for the first screening of ANA levels in serum samples obtained from 2181 residents of a small Japanese town. Individuals positive for IF-ANA were then further tested for disease-specific ANAs using eight enzyme immunoassays. Physical status and the presence of illness were determined by means of questionnaires and medical examinations. Based on the result of the IF-ANA assay, the rates of positive samples at 1:40 and 1:160 dilutions were 26.0 and 9.5%, respectively, with females have significantly higher positivity rates than males (P < 0.0001). Among 566 IF-ANA-positive individuals, 100 individuals were found to have 114 disease-specific ANAs. Anti-SSA/Ro, anti-centromere, and anti-U1RNP antibodies were detected in 58, 30, and 11 individuals, respectively, but anti-Sm, anti-Scl-70, and anti-Jo-1 antibodies were undetectable. Questionnaires and medical examinations revealed that among 60 disease-specific ANA-positive individuals that were available for testing, six had Sjögren’s syndrome (SS), five were suspected of having SS, and five had rheumatoid arthritis. Surprisingly, 34 (57%) of the disease-specific ANA-positive individuals were clinically healthy. Anti-SSA/Ro, anti-centromere, and anti-U1RNP antibodies were quite frequent among clinically healthy Japanese subjects, although anti-Sm, anti-Scl-70, and anti-Jo-1 antibodies were not. Of the 60 individuals who tested positive for disease-specific ANAs, 30% (18/60) actually manifested systemic rheumatic diseases, while 50% showed no detectable signs or symptoms of rheumatic diseases.


Similar content being viewed by others
References
Tan EM. Antinuclear antibodies: diagnostic markers for autoimmune diseases and probes for cell biology. Adv Immunol. 1989;44:93–151.
Fujimoto M, Shimozuma M, Yazawa N, Kubo M, Ihn H, Sato S, et al. Prevalence and clinical relevance of 52-kDa and 60-kDa Ro/SS-A autoantibodies in Japanese patients with systemic sclerosis. Ann Rheum Dis. 1997;56:667–70.
Cleymaet JE, Nakamura RM. Indirect immunofluorescent antinuclear antibody tests: comparison of sensitivity and specificity of different substrates. Am J Clin Pathol. 1972;58:388–93.
Kozin F, Fowler M, Koethe SM. A comparison of the sensitivities and specificities of different substrates for the fluorescent antinuclear antibody test. Am J Clin Pathol. 1980;74:785–90.
Tan EM, Feltkamp TE, Smolen JS, Butcher B, Dawkins R, Fritzler MJ, et al. Range of antinuclear antibodies in “healthy” individuals. Arthritis Rheum. 1997; 40:1601–11.
Hayashi N, Kawamoto T, Mukai M, Morinobu A, Koshiba M, Kondo S, et al. Detection of antinuclear antibodies by use of an enzyme immunoassay with nuclear HEp-2 cell extract and recombinant antigens: comparison with immunofluorescence assay in 307 patients. Clin Chem. 2001;47:1649–59.
von Muhlen CA, Tan EM. Autoantibodies in the diagnosis of systemic rheumatic diseases. Semin Arthritis Rheum. 1995;24:323–58.
de Vlam K, De Keyser F, Verbruggen G, Vandenbossche M, Vanneuville B, D’Haese D, et al. Detection and identification of antinuclear autoantibodies in the serum of normal blood donors. Clin Exp Rheumatol. 1993;11:393–7.
Manoussakis MN, Tzioufas AG, Silis MP, Pange PJ, Goudevenos J, Moutsopoulos HM. High prevalence of anti-cardiolipin and other autoantibodies in a healthy elderly population. Clin Exp Immunol. 1987;69:557–65.
Azizah MR, Azila MN, Zulkifli MN, Norita TY. The prevalence of antinuclear, anti-dsDNA, anti-Sm and anti-RNP antibodies in a group of healthy blood donors. Asian Pac J Allergy Immunol. 1996;14:125–8.
Juby AG, Davis P, McElhaney JE, Gravenstein S. Prevalence of selected autoantibodies in different elderly subpopulations. Br J Rheumatol. 1994;33:1121–4.
Ruffatti A, Calligaro A, Del Ross T, Bertoli MT, Doria A, Rossi L, et al. Anti-double-stranded DNA antibodies in the healthy elderly: prevalence and characteristics. J Clin Immunol. 1990;10:300–3.
Murakami A, Kojima K, Ohya K, Imamura K, Takasaki Y. A new conformational epitope generated by the binding of recombinant 70-kd protein and U1 RNA to anti-U1 RNP autoantibodies in sera from patients with mixed connective tissue disease. Arthritis Rheum. 2002;46:3273–82.
Alspaugh MA, Tan EM. Antibodies to cellular antigens in Sjogren’s syndrome. J Clin Invest. 1975;55:1067–73.
Wold RT, Young FE, Tan EM, Farr RS. Deoxyribonucleic acid antibody: a method to detect its primary interaction with deoxyribonucleic acid. Science. 1968;161:806–7.
Hansen KE, Arnason J, Bridges AJ. Autoantibodies and common viral illnesses. Semin Arthritis Rheum. 1998;27:263–71.
Muller S, Boire G, Ossondo M, Ricchiuti V, Smadja D, Vernant JC, et al. IgG autoantibody response in HTLV-I-infected patients. Clin Immunol Immunopathol. 1995;77:282–90.
Vitali C, Bombardieri S, Moutsopoulos HM, Balestrieri G, Bencivelli W, Bernstein RM, et al. Preliminary criteria for the classification of Sjogren’s syndrome. Results of a prospective concerted action supported by the European community. Arthritis Rheum. 1993;36:340–7.
Conrad K, Mehlhorn J. Diagnostic and prognostic relevance of autoantibodies in uranium miners. Int Arch Allergy Immunol. 2000;123:77–91.
Peene I, Meheus L, Veys EM, De Keyser F. Detection and identification of antinuclear antibodies (ANA) in a large and consecutive cohort of serum samples referred for ANA testing. Ann Rheum Dis. 2001;60:1131–6.
Ruffatti A, Artifoni L, Glorioso S, Calligaro A, Doria A, Gambari P, et al. Prevalence of anticentromere antibody in blood relatives of anticentromere positive patients. J Rheumatol. 1985;12:940–3.
Soma Y, Takehara K, Ishibashi Y. Clinical distribution of anticentromere antibody in Japanese patients. Dermatologica. 1989;178:16–9.
Spiewak R, Stojek N. Antinuclear antibodies among eastern-Polish rural inhabitants. Ann Agric Environ Med. 2003;10:207–9.
Keech CL, McCluskey J, Gordon TP. Transfection and overexpression of the human 60-kDa Ro/SS-A autoantigen in HEp-2 cells. Clin Immunol Immunopathol. 1994;73:146–51.
Tan EM, Smolen JS, McDougal JS, Butcher BT, Conn D, Dawkins R, et al. A critical evaluation of enzyme immunoassays for detection of antinuclear autoantibodies of defined specificities. I. Precision, sensitivity, and specificity. Arthritis Rheum. 1999;42:455–64.
Tan EM, Smolen JS, McDougal JS, Fritzler MJ, Gordon T, Hardin JA, et al. A critical evaluation of enzyme immunoassay kits for detection of antinuclear autoantibodies of defined specificities. II. Potential for quantitation of antibody content. J Rheumatol. 2002;29:68–74.
Adebajo AO, Charles P, Maini RN, Hazleman BL. Autoantibodies in malaria, tuberculosis and hepatitis B in a west African population. Clin Exp Immunol 1993;92:73–6.
Clifford BD, Donahue D, Smith L, Cable E, Luttig B, Manns M, et al. High prevalence of serological markers of autoimmunity in patients with chronic hepatitis C. Hepatology. 1995;21:613–9.
Li L, Chen M, Huang DY, Nishioka M. Frequency and significance of antibodies to chromatin in autoimmune hepatitis type I. J Gastroenterol Hepatol. 2001;16:245–7.
Maricq HR, Harper FE, Khan MM, Tan EM, LeRoy EC. Microvascular abnormalities as possible predictors of disease subsets in Raynaud phenomenon and early connective tissue disease. Clin Exp Rheumatol. 1983;1:195–205.
Bernstein RM, Callender ME, Neuberger JM, Hughes GR, Williams R. Anticentromere antibody in primary biliary cirrhosis. Ann Rheum Dis. 1982;41:612–4.
Arbuckle MR, McClain MT, Rubertone MV, Scofield RH, Dennis GJ, James JA, et al. Development of autoantibodies before the clinical onset of systemic lupus erythematosus. N Engl J Med. 2003;349:1526–33.
Acknowledgments
We thank Mr. Tsutomu Iwanami and Ms. Sanae Ohgaki (Health Center of Y Town) for their excellent technical support of the health examinations. We also thank Mr. Hiroshi Suno, Mr. Jiro Arai, and Mr. Yasushi Ishihara (Medical & Biological Laboratories), Mr. Kazuo Ohkubo (Mitsubishi Kagaku Iatron), Mr. Kotaro Banno (Dade Behring), Mr. Takahiko Tsuchida (Abbott Japan), and Mr. Tatsuya Nomura (Fujirebio) for providing reagent kits and technical advice. This research was supported in part by a Grant-in-Aid for Scientific Research (12922105) from the Japan Society for the Promotion of Science and by research grants (044-0211-1558) from the Ministry of Health, Labour and Welfare of Japan.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Hayashi, N., Koshiba, M., Nishimura, K. et al. Prevalence of disease-specific antinuclear antibodies in general population: estimates from annual physical examinations of residents of a small town over a 5-year period. Mod Rheumatol 18, 153–160 (2008). https://doi.org/10.1007/s10165-008-0028-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10165-008-0028-1