Abstract
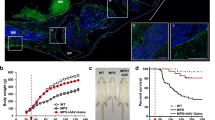
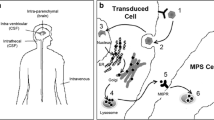
Mucopolysaccharidosis type II (MPS II; Hunter syndrome) is an X-linked inherited disorder caused by a deficiency of the enzyme iduronate-2-sulfatase (IDS), which results in the lysosomal accumulation of glycosaminoglycans (GAG) such as dermatan and heparan sulfate. Here, we report the generation of IDS knockout mice, a model of human MPS II, and an analysis of the resulting phenotype. We also evaluated the effect of gene therapy with a pseudotyped, recombinant adeno-associated virus 2/8 vector encoding the human IDS gene (rAAV-hIDS) in IDS-deficient mice. IDS activity and GAG levels were measured in serum and tissues after therapy. Gene therapy completely restored IDS activity in plasma and tissue of the knockout mice. The rescued enzymatic activity completely cleared the accumulated GAGs in all the tissues analyzed. This model can be used to explore the therapeutic potential of IDS replacement and other strategies for the treatment of MPS II. Additionally, AAV2/8 vectors have promising future clinical applications for the treatment of patients with MPS II.
Similar content being viewed by others
References
Birkenmeier, E.H., Davisson, M.T., Beamer, W.G., Ganschow, R.E., Vogler, C.A., Gwynn, B., Lyford, K.A., Maltais, L.M., and Wawrzyniak, C.J. (1989). Murine mucopolysaccharidosis type VII. Characterization of a mouse with beta-glucuronidase deficiency. J. Clin. Invest. 83, 1258–1266.
Braun, S.E., Aronovich, E.L., Anderson, R.A., Crotty, P.L., McIvor, R.S., and Whitley, C.B. (1993). Metabolic correction and cross-correction of mucopolysaccharidosis type II (Hunter syndrome) by retroviral-mediated gene transfer and expression of human iduronate-2-sulfatase. Proc. Natl. Acad. Sci. USA 90, 11830–11834.
Brooks, D.A., Kakavanos, R., and Hopwood, J.J. (2003). Significance of immune response to enzyme-replacement therapy for patients with a lysosomal storage disorder. Trends Mol. Med. 9, 450–453.
Cardone, M., Polito, V.A., Pepe, S., Mann, L., D’Azzo, A., Auricchio, A., Ballabio, A., and Cosma, M.P. (2006). Correction of Hunter syndrome in the MPS II mouse model by AAV2/8-mediated gene delivery. Hum. Mol. Genet. 15, 1225–1236.
Clarke, L.A., Russell, C.S., Pownall, S., Warrington, C.L., Borowski, A., Dimmick, J.E., Toone, J., and Jirik, F.R. (1997). Murine mucopolysaccharidosis type I: targeted disruption of the murine alpha-L-iduronidase gene. Hum. Mol. Genet. 6, 503–511.
Coppa, G.V., Gabrielli, O., Zampini, L., Pierani, P., Giorgi, P.L., Jezequel, A.M., Orlandi, F., Miniero, R., Busca, A., De Luca, T., et al. (1995). Bone marrow transplantation in Hunter syndrome (mucopolysaccharidosis type II): two-year follow-up of the first Italian patient and review of the literature. Pediatr. Med. Chir. 17, 227–235.
Daniele, A., Tomanin, R., Villani, G.R., Zacchello, F., Scarpa, M., and Di Natale, P. (2002). Uptake of recombinant iduronate-2-sulfatase into neuronal and glial cells in vitro. Biochim. Biophys. Acta 1588, 203–209.
Daya, S., and Berns, K.I. (2008). Gene therapy using adeno-associated virus vectors. Clin. Microbiol. Rev. 21, 583–593.
Di Francesco, C., Cracco, C., Tomanin, R., Picci, L., Ventura, L., Zacchello, E., Di Natale, P., Anson, D.S., Hopwood, J.J., Graham, F.L., et al. (1997). In vitro correction of iduronate-2-sulfatase deficiency by adenovirus-mediated gene transfer. Gene Ther. 4, 442–448.
Evers, M., Saftig, P., Schmidt, P., Hafner, A., McLoghlin, D.B., Schmahl, W., Hess, B., von Figura, K., and Peters, C. (1996). Targeted disruption of the arylsulfatase B gene results in mice resembling the phenotype of mucopolysaccharidosis VI. Proc. Natl. Acad. Sci. USA 93, 8214–8219.
Garcia, A.R., Pan, J., Lamsa, J.C., and Muenzer, J. (2007). The characterization of a murine model of mucopolysaccharidosis II (Hunter syndrome). J. Inherit. Metab. Dis. 30, 924–934.
Grimm, D., Pandey, K., Nakai, H., Storm, T.A., and Kay, M.A. (2006). Liver transduction with recombinant adeno-associated virus is primarily restricted by capsid serotype not vector genotype. J. Virol. 80, 426–439.
Hacein-Bey-Abina, S., von Kalle, C., Schmidt, M., Le Deist, F., Wulffraat, N., McIntyre, E., Radford, I., Villeval, J.L., Fraser, C.C., Cavazzana-Calvo, M., et al. (2003). A serious adverse event after successful gene therapy for X-linked severe combined immunodeficiency. N. Engl. J. Med. 348, 255–256.
Jung, S.C., Han, I.P., Limaye, A., Xu, R., Gelderman, M.P., Zerfas, P., Tirumalai, K., Murray, G.J., During, M.J., Brady, R.O., et al. (2001). Adeno-associated viral vector-mediated gene transfer results in long-term enzymatic and functional correction in multiple organs of Fabry mice. Proc. Natl. Acad. Sci. USA 98, 2676–2681.
Ko, J.Y., Ahn, Y.L., and Cho, B.N. (2003). Angiogenesis and white blood cell proliferation induced in mice by injection of a prolactin-expressing plasmid into muscle. Mol. Cells 15, 262–270.
Li, P., Thompson, J.N., Hug, G., Huffman, P., and Chuck, G. (1996). Biochemical and molecular analysis in a patient with the severe form of Hunter syndrome after bone marrow transplantation. Am. J. Med. Genet. 64, 531–535.
Li, H.H., Yu, W.H., Rozengurt, N., Zhao, H.Z., Lyons, K.M., Anagnostaras, S., Fanselow, M.S., Suzuki, K., Vanier, M.T., and Neufeld, E.F. (1999). Mouse model of Sanfilippo syndrome type B produced by targeted disruption of the gene encoding alpha-N-acetylglucosaminidase. Proc. Natl. Acad. Sci. USA 96, 14505–14510.
Liu, Q., and Muruve, D.A. (2003). Molecular basis of the inflammatory response to adenovirus vectors. Gene Ther. 10, 935–940.
Malatack, J.J., Consolini, D.M., and Bayever, E. (2003). The status of hematopoietic stem cell transplantation in lysosomal storage disease. Pediatr. Neurol. 29, 391–403.
Martin, R., Beck, M., Eng, C., Giugliani, R., Harmatz, P., Munoz, V., and Muenzer, J. (2008). Recognition and diagnosis of mucopolysaccharidosis II (Hunter syndrome). Pediatrics 121, e377–386.
Muenzer, J., Lamsa, J.C., Garcia, A., Dacosta, J., Garcia, J., and Treco, D.A. (2002). Enzyme replacement therapy in mucopolysaccharidosis type II (Hunter syndrome): a preliminary report. Acta Paediatr. 91, 98–99.
Muruve, D.A. (2004). The innate immune response to adenovirus vectors. Hum. Gene Ther. 15, 1157–1166.
Neufeld, E.F. (1991). Lysosomal storage diseases. Annu. Rev. Biochem. 60, 257–280.
Oh, H.J., Park, E.S., Kang, S., Jo, I., and Jung, S.C. (2004). Longterm enzymatic and phenotypic correction in the phenylketonuria mouse model by adeno-associated virus vectormediated gene transfer. Pediatr. Res. 56, 278–284.
Ohshima, T., Murray, G.J., Swaim, W.D., Longenecker, G., Quirk, J.M., Cardarelli, C.O., Sugimoto, Y., Pastan, I., Gottesman, M.M., Brady, R.O., et al. (1997). alpha-Galactosidase A deficient mice: a model of Fabry disease. Proc. Natl. Acad. Sci. USA 94, 2540–2544.
Peters, C., and Krivit, W. (2000). Hematopoietic cell transplantation for mucopolysaccharidosis IIB (Hunter syndrome). Bone Marrow Transplant. 25, 1097–1099.
Sly, W.S., Vogler, C., Grubb, J.H., Levy, B., Galvin, N., Tan, Y., Nishioka, T., and Tomatsu, S. (2006). Enzyme therapy in mannose receptor-null mucopolysaccharidosis VII mice defines roles for the mannose 6-phosphate and mannose receptors. Proc. Natl. Acad. Sci. USA 103, 15172–15177.
Tomanin, R., and Scarpa, M. (2004). Why do we need new gene therapy viral vectors? Characteristics, limitations and future perspectives of viral vector transduction. Curr. Gene Ther. 4, 357–372.
Tomatsu, S., Orii, K.O., Vogler, C., Nakayama, J., Levy, B., Grubb, J.H., Gutierrez, M.A., Shim, S., Yamaguchi, S., Nishioka, T., et al. (2003). Mouse model of N-acetylgalactosamine-6-sulfate sulfatase deficiency (Galns−/−) produced by targeted disruption of the gene defective in Morquio A disease. Hum. Mol. Genet. 12, 3349–3358.
Vellodi, A., Young, E., Cooper, A., Lidchi, V., Winchester, B., and Wraith, J.E. (1999). Long-term follow-up following bone marrow transplantation for Hunter disease. J. Inherit. Metab. Dis. 22, 638–648.
Voznyi, Y.V., Keulemans, J.L., and van Diggelen, O.P. (2001). A fluorimetric enzyme assay for the diagnosis of MPS II (Hunter disease). J. Inherit. Metab. Dis. 24, 675–680.
Whitley, C.B., McIvor, R.S., Aronovich, E.L., Berry, S.A., Blazar, B.R., Burger, S.R., Kersey, J.H., King, R.A., Faras, A.J., Latchaw, R.E., et al. (1996). Retroviral-mediated transfer of the iduronate-2-sulfatase gene into lymphocytes for treatment of mild Hunter syndrome (mucopolysaccharidosis type II). Hum. Gene Ther. 7, 537–549.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Jung, SC., Park, ES., Choi, E.N. et al. Characterization of a novel mucopolysaccharidosis type II mouse model and recombinant AAV2/8 vector-mediated gene therapy. Mol Cells 30, 13–18 (2010). https://doi.org/10.1007/s10059-010-0083-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10059-010-0083-2