Abstract
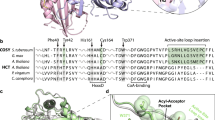
Caffeoyl coenzyme A-O-methyltransferases (CCoAOMTs) which are characterized under class I plant OMTs, methylates CoA thioesters, with an in vitro kinetic preference for caffeoyl CoA. CCoAOMTs exhibit association with lignin biosynthesis by showing a prime role in the synthesis of guaiacyl lignin and providing the substrates for synthesis of syringyl lignin. The sequence analysis of CCoAOMT from Populus trichopora exhibits 58 nucleotide substitutions, where transitions overcome transversions. Validation of homology models of both CCoAOMT1 and 2 isoforms reveals that 92.4% and 96% residues are falling in the most favorable region respectively in the Ramachandran plot, indicating CCoAOMT2 as the more satisfactory model, and the overall quality factor of both isoforms is 98.174. The structural architecture analysis is showing very good packing of residues similar to protein crystal structures data. The active site residues and substrate-product interactions showed that CCoAOMT2 possesses more affinity toward caffeoyl CoA, feruloyl CoA, 5-hydroxy feruloyl CoA and sinapoyl CoA than CCoAOMT1, therefore it exist in a more active conformation. The affinity of CCoAOMT2 with feruloyl CoA is highest among all the affinities of both CCoAOMT isoforms with their substrates and products. This information has potential implications to understand the mechanism of CCoAOMT related enzymatic reactions in Populus trichopora, however the approach will be applicable in prediction of substrates and engineering 3D structures of other enzymes as well.






Similar content being viewed by others
References
Park SH, Kim BG, Lee SH, Lim Y, Cheong Y, Ahn JH (2007) Molecular modeling and site directed mutagenesis of the O-methyltransferase, SOMT-9 Reveal amino acids important for its reaction and regioselectivity. Bull Korean Chem Soc 28:2248–2252
Ibrahim RK, Bruneau A, Bantignies B (1998) Plant O-methyltransferases:molecular analysis, common signature and classification. Plant Mol Biol 36:1–10
Joshi CP, Chiang VL (1998) Conserved sequence motifs in plant S-adenosyl-L-methionine dependent methyltransferases. Plant Mol Biol 37:663–674
Pagadala NS, Arha M, Reddy PS, Kumar R, Sirisha VL, Prashant S, Reddy KJ K, Khan B, Rawal SK, Kishor PBK (2009) Phylogenetic analysis, homology modelling, molecular dynamics and docking studies of caffeoyl–CoA-O-methyl transferase (CCoAOMT 1 and 2) isoforms isolated from subabul (Leucaena leucocephala). J Mol Model 15:203–221
Ferrer J, Zubieta C, Dixon RA, Noel JP (2005) Crystal structures of Alfalfa caffeoyl coenzyme A 3-O-Methyltransferase. Plant Physiol 137:1009–1017
Parvathi K, Chen F, Guo D, Blount JW, Dixon RA (2001) Substrate preferences of O-methyltransferases in alfalfa suggest new pathways for 3-O-methylation of monolignols. Plant J 25:193–202
Vance CP, Kirk TK, Sherwood RT (1980) Lignification as a mechanism of disease resistance. Annu Rev Phytopathol 18:259–288
Lee JY, Lee S, Kim Y (2007) 3D structure of Bacillus halodurans O- methyltransferase, a novel bacterial O-methyltransferase by comparative homology modelling. Bull Korean Chem Soc 28:941–946
Lee YJ, Kim BG, Chong Y, Lim Y, Ahn JH (2008) Cation dependent O-methyltransferases from rice. Planta 227:641–647
Ibdah M, Zhang XH, Schmidt J, Vogt T (2003) A novel Mg2+-dependent O-methyltransferase in the phenylpropanoid metabolism of Mesembryanthemum crystallinum. J Biol Chem 278:43961–43972
Kim BG, Shin KH, Lee Y, Hur HG, Lim Y, Ahn JH (2005) Multiple regiospecific methylations of a flavonoid by plant O-methyltransferases expressed in E. coli. Biotechnol Lett 27:1861–1864
Kim BG, Lee Y, Hur HG, Lim Y, Ahn JH (2006) Flavonoid 3′-O-methyltransferase from rice: cDNA cloning, characterization and functional expression. Phytochemistry 67:387–394
Kim BG, Lee Y, Hur HG, Lim Y, Ahn JH (2006) Production of three O-methylated esculetins with Escherichia coli expressing O-methyltransferase from poplar [Populus deltoides]. Biosci Biotechnol Biochem 70:1269–1272
Kim JH, Kim BG, Park Y, Han JH, Lim Y, Ahn JH (2006) Production of 5, 7-dihydroxy, 3_, 4_, 5_- trimethoxyXavone from 5, 7, 3_, 4_, 5_-pentahydroxyXavone using two O-methyltransferases expressed in Escherichia coli. Agric Chem Biotechnol 49:114–116
Meyermans H, Morrell K, Lapierre C, Pollet B, De Bruyn A, Busson R, Herdewijn P, Devresse B, Van Beeumen J, Marita JM et al. (2000) Modifications in lignin and accumulation of phenolic glycosides in poplar xylem upon down-regulation of caffeoyl coenzyme A O-methyltransferase, an enzyme involved in lignin biosynthesis. J Biol Chem 275:36899–36909
Li L, Osakabe Y, Chandrashekhar PJ, Chiang VL (1999) Secondary xylemspecific expression of caffeoyl-coenzyme A 3-O-Methyltransferase plays an important role in the methylation pathway associated with lignin biosynthesis in loblolly pine. Plant Mol Biol 40:555–565
Martz F, Maury S, Pincon G, Legrand M (1998) cDNA cloning, substrate specificity and expression study of tobacco caffeoyl-CoA 3-O-methyltransferase, a lignin biosynthetic enzyme. Plant Mol Biol 36:427–437
Inoue K, Sewalt VJH, Balance GM, Ni W, Stürzer C, Dixon RA (1998) Developmental expression and substrate specificities of alfalfa caffeic acid 3-O-methyltransferase and caffeoyl coenzyme A 3-O-methyltransferase in relation to lignifications. Plant Physiol 117:761–770
Ye ZH, Kneusel RE, Matern U, Varner JE (1994) An alternative methylation pathway in lignin biosynthesis in Zinnia. Plant Cell 6:1427–1439
Kühnl T, Koch U, Heller W, Wellmann E (1989) Elicitor induced S-adenosyl-1-methionine:caffeoyl-CoA 3-O-methyltransferase from carrot cell suspension cultures. Plant Sci 60:21–25
Pakusch AE, Kneusel RE, Matern U (1989) S-Adenosyl-1-methionine:trans-caffeoyl-coenzyme A 3-O-methyltransferase from elicitor-treated parsley cell suspension cultures. Arch Biochem Biophys 271:488–494
Ye ZH (1997) Association of caffeoyl coenzyme expression with lignifying tissues A 3-O-Methyltra-nsferase in severa1 dicot plants. Plant Physiol 115:1341–1350
Hoffmann L, Maury S, Bergdoll M, Thion L, Erard M, Legrand M (2001) Identification of the enzymatic active site of tobacco caffeoylcoenzyme a O-Methyltransferase by site-directed mutagenesis. J Biol Chem 276:36831–36838
Maury S, Geoffroy P, Legrand M (1999) Tobacco O-Methyltransferases involved in phenylpropanoid metabolism. The different caffeoyl-coenzyme A/5-Hydroxyferuloyl-Coenzyme A 3/5-O-Methyltransfe-rase and Caffeic Acid/5-Hydroxyferulic Acid 3/5-O-Methyltransferase classes have distinct substrate specificities and expression patterns. Plant Physiol 121:215–224
Vidgren J, Svensson LA, Liljas A (1994) Crystal structure of catechol O-methyltransferase. Nature 368:354–357
Zubieta C, He X-Z, Dixon RA, Noel JP (2001) Structures of two natural product methyltransferases reveal the basis for substrate specificity in plant O-methyltransferases. Nat Struct Biol 8:271–279
Zubieta C, Kota P, Ferrer J-L, Dixon RA, Noel JP (2002) Structural basis for the modulation of lignin monomer methylation by caffeic acid/ 5-hydroxyferulic acid 3/5-O-methyltransferase. Plant Cell 14:1265–1277
Sali A, Blundell TL (1993) Comparative protein modelling by satisfaction of spatial restraints. J Mol Biol 234:779–815
Eswar N, Marti-Renom MA, Webb B, Madhusudhan MS, Eramian D, Shen M, Pieper U, Sali A (2006) Comparative protein structure modeling with MODELLER. Current protocols in bioinformatics. Wiley, New York, Supplement 15, 5.6.1-5.6.30
Laskowski RA, MacArthur MW, Moss DS, Thornton JM (1993) PROCHECK: a program to check the stereochemical quality of protein structures. J Appl Crystallogr 26:283–291
Colovos C, Yeates TO (1993) Verification of protein structures: patterns of nonbonded atomic interactions. Protein Sci 2:1511–1519
Pedretti A, Villa L, Vistoli G (2004) VEGA - An open platform to develop chemo-bio-informatics applications, using plug-in architecture and script" programming". J Comput Aided Mater Des 18:167–173
Phillips JC, Braun R, Wang W, Gumbart J, Tajkhorshid E, Villa E, Chipot C, Skeel RD, Kale L, Schulten K (2005) Scalable molecular dynamics with NAMD. J Comput Chem 26:1781–1802
MacKerell AD, Brooks B, Brooks CL, Nilsson L, Roux B, Won Y, Karplus M (1998) CHARMM: the energy function and its parameterization with an overview of the program. In: Schleyer PVR et al. (eds) The encyclopedia of computational chemistry 1. Wiley, Chichester, pp 271–277
MacKerell AD et al. (1998) All-atom empirical potential for molecular modeling and dynamics studies of proteins. J Phys Chem B 102:3586–3616
Willard L, Ranjan A, Zhang H, Monzavi H, Boyko RF, Sykes BD, Wishart DS (2003) VADAR: a web server for quantitative evaluation of protein structure quality. Nucleic Acids Res 31(13):3316–3319
Dundas J, Ouyang Z, Tseng J, Binkowski A, Turpaz Y, Liang J (2006) CASTp: Computed atlas of surface topography of proteins with structural and topographical mapping of functionally annotated residues. Nucleic Acids Res 34:W116–118
Wheeler DL, Barrett T, Benson DA, Wheeler DL, Barrett T, Benson DA, Bryant SH, Canese K, Chetvernin V, Church DM et al. (2006) Database resources of the National Center for Biotechnology Information. Nucleic Acids Res 34:D173–180
Jones G, Willett P, Glen RC (1995) Molecular recognition of receptor sites using a genetic algorithm with a description of desolvation. J Mol Biol 245:43–53
Zhong R, Morrison WH, Himmelsbach DS, Poole FL, Ye ZH (2000) Essential role of caffeoyl coenzyme A O-methyltransferase in lignin biosynthesis in woody poplar plants. Plant Physiol 124:563–577
Zhong R, Morrison WH, Negrel J, Ye ZH (1998) Dual methylation pathways in lignin Biosynthesis. Plant Cell 10:2033–2045
Acknowledgments
Research in VV’s laboratory is supported by XI OBC plan grant of University of Hyderabad. The authors wish to thank Dr. S. C. Ghadi for providing computational facility, Prof. U.M.X. Sangodkar for his valuable suggestions. We also thank Dr. P.V. Bramhachari for helpful discussion.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Phogat, N., Vindal, V., Kumar, V. et al. Sequence analysis, in silico modeling and docking studies of Caffeoyl CoA-O-methyltransferase of Populus trichopora . J Mol Model 16, 1461–1471 (2010). https://doi.org/10.1007/s00894-010-0656-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00894-010-0656-1