Abstract
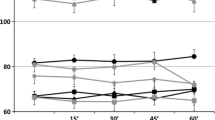
The method of vagus somatosensory evoked potentials (VSEP) was introduced to easily measure the activity of vagus brain stem nuclei. In Alzheimer’s disease, this measure was characterized by longer latencies as compared to controls while amplitudes did not show statistical significant differences at frontal and central recording sites. Therefore, the influence of stimulation and recording parameters on amplitudes of VSEP were systematically examined. In 20 healthy participants, VSEP measurement was done by electrical stimulation of the cutaneous representation of the vagus nerve in the external auditory channel and recording of VSEP over the scalp. The optimum stimulation intensity is 8 mA without perception of pain. There is no effect of stimulation side or gender. Maximum VSEP amplitudes are detected at bipolar recordings comprising the electrode T4 without statistically significant differences of latencies, wave shape and polarity. Thus, recordings of future examinations should be performed at 8 mA including this temporal electrode position. The reason for focussing on brain stem evoked potentials is that recent work has accumulated evidence for this area being involved in early phases of neurodegenerative diseases such as Alzheimer’s disease and Parkinson’s disease. Improved methodological knowledge may facilitate the assessment of this non-invasive and cost-effective method in the early diagnosis of neurodegenerative disorders.


Similar content being viewed by others
References
Braak H, Rüb U, Gai W, Del Tredici K (2003) Idiopathic Parkinson’s disease: possible routes by which vulnerable neuronal types may be subject to neuroinvasion by an unknown pathogen. J Neural Transm 110:517–536
Chiappa KH (1983) Evoked potentials in clinical medicine. Raven Press, New York, p 1938
Del Tredici K, Rüb U, De Vos RA, Bohl JR, Braak H (2002) Where does Parkinson disease pathology begin in the brain? J Neuropathol Exp Neurol 61:413–426
Drechsler F, Neuhauser B (1986) Somatosensory trigeminal evoked potentials in normal subjects and in patients with trigeminal neuralgia before and after thermocoagulation of the Ganglion gasseri. Electromyogr Clin Neurophysiol 26:315–326
Fallgatter AJ, Neuhauser B, Herrmann MJ, Ehlis AC, Wagener A, Scheuerpflug P, Reiners K, Riederer P (2003) Far field potentials from the brain stem after transcutaneous vagus nerve stimulation. J Neural Transm 110:1437–1443
Fallgatter AJ, Ehlis AC, Ringel TM, Herrmann MJ (2005) Age-effect on far field potentials from the brain stem after transcutaneous vagus nerve stimulation. Int J Psychophysiol 56:37–43
Fallgatter AJ, Polak T, Metzger F, Richter MM, Baehne CG, Plichta MM, Scheuerpflug P, Ehlis AC, Fallgatter AJ (2006) Brainstem vagus nuclei evoked potentials—a new diagnostic method in neuropsychiatry? Nervenheilkunde 25:669–673
Fisch U, Schulthess GV (1963) Electromyographic studies on the human stapedial muscle. Acta Otolaryngol 56:287–297
George MS, Nahas Z, Bohning DE, Lomarev M, Denslow S, Ochsenbach R, Ballenger JC (2000) Vagus nerve stimulation: a new form of therapeutic brain stimulation. CNS Spectr 5:43–52
George MS, Nahas Z, Bohning DE, Kozel FA, Anderson B, Chae JH, Lomarev M, Denslow S, Li X, Mu C (2002) Vagus nerve stimulation therapy. A research update. Neurology 59:S56–S61
Grinnell AD (1963) The neurophysiology of audition in bats: intensity and frequency parameters. J Physiol Lond 167:38–66
Hammond EJ, Wilder BJ (1985) Enhanced auditory postauricular evoked responses after corticobulbar lesions. Neurology 35:278–281
Ito S (2002) Visceral region in the rat primary somatosensory cortex identified by vagal evoked potential. J Comp Neurol 444:10–24
Jaspers HH (1958) Report of the committee on methods of clinical examination in electroencephylography. Electroencephalogr Clin Neurophysiol 10:370–371
Jewett DL, Williston JS (1971) Auditory-evoked far fields averaged from the scalp of humans. Brain 94:681–696
Jewett DL, Romano MN, Williston JS (1970) Human auditory evoked potentials: possible brain stem components detected on the scalp. Science 167:1517–1518
Kiess O, Hösel K, Schanze A, Kornhuber J, Foster C, Kraus T (2007) Transkutane Vagusnerv-Stimulation im anterioren Gehörgang führt zur fMRI BOLD-Signal Hyperaktivation eines Vagus-Kerngebietes im Hirnstamm. Nervenarzt 78:276–277
Kraus T, Hösl K, Kiess O, Schanze A, Kornhuber J, Forster C (2007) BOLD fMRI deactivation of limbic and temporal brain structures and mood enhancing effect by transcutaneous vagus nerve stimulation. J Neural Transm 114:1485–1493
Parvizi J, Van Hoesen GW, Damasio A (2001) The selective vulnerability of brain stem nuclei to Alzheimer’s disease. Ann Neurol 49:53–66
Peukert ET, Filler TJ (2002) The nerve supply of the human auricle. Clin Anat 15:35–37
Polak T, Ehlis AC, Langer J, Plichta M, Metzger F, Ringel TM, Fallgatter AJ (2007) Non-invasive measurement of vagus activity in the brainstem—a methodological progress towards earlier diagnosis of dementias? J Neural Transm 110:1437–1443
Riederer P (2002) Psychiatric disorders and Parkinson’s disease: basic concepts. Eur Arch Psych Clin Neurosci 252(Suppl 1):9
Schachter SC (2004) Vagus nerve stimulation: mood and cognitive effects. Epilepsy Behav 5:S56–S59
Sobotta J (1982) Atlas der Anatomie des Menschen. Urban & Schwarzenberg, München, pp 138–139
Stechison MT (1993) The trigeminal evoked potential: Part II. Intraoperative recording of short latency responses. Neurosurgery 33:639–644
Thal DR, Rüb U, Orantes M, Braak H (2002) Phases of A[beta]-deposition in the human brain and its relevance for the development of AD. Neurology 58:1791–1800
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Polak, T., Markulin, F., Ehlis, AC. et al. Far field potentials from brain stem after transcutaneous Vagus nerve stimulation: optimization of stimulation and recording parameters. J Neural Transm 116, 1237–1242 (2009). https://doi.org/10.1007/s00702-009-0282-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00702-009-0282-1